Sertoli-Leydig Cell Tumour (SLCT) is included under sex-cord stromal tumour of testis or ovary. Ovarian pathology is an extremely rare entity constituting less than 0.5% of all ovarian neoplasms. Majority of the cases present at younger age group, i.e., 2nd and 3rd decade of life. The clinical presentation depends on either mass effect or excess hormone production. Virillization symptoms are the most common presentation and depend upon the quantity of androgen production. Most of the patients have a unilateral mass without extraovarian spread and present at stage-I. Treatment and prognosis depends on the degree of the differentiation and the stage of the disease. Here, we report a case of SLCT of the ovary in a young female. The patient initially presented with pain abdomen due to mass effect and was treated with conservative surgery followed by adjuvant chemotherapy due to the presence of poorly differentiated pathology with heterologous elements.
Chemotherapy, Conservative surgery, Diagnosis, Sex-cord stromal tumour
Case Report
A 27-year-old female married since one year, presented with left sided pain in abdomen without any symptoms of virillization. Per-abdomen examination showed a non-tender hard mass approximately 10 x 10 cm size at left iliac fossa. The patient was clinically suspected as teratoma or other germ cell tumours of the left ovary.
Abdominal ultrasonography showed a well marginated solid mass size 11.9 x 8.6 cm at left adnexa. Contrast enhanced CT scan showed a mild heterogeneous enhancing lobulated solid mass of size approximately 8x10 cm in the left adnexa. Serum markers: LDH, beta-HCG, AFP and CA-125 were within normal limits. Preoperative serum inhibin test was not done due to absence of any symptoms of virillization. Left salpingo-ophorectomy was performed. There was no lymph node enlargement on intraoperative findings and also there was absence of any omental seedling. Peritoneal fluid washing was done and cytology was negative for malignancy.
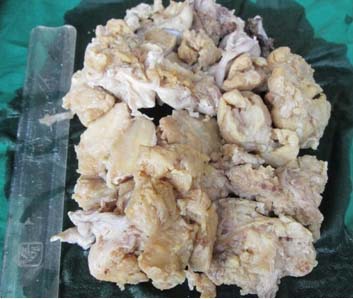
Grossly, specimen showed a well encapsulated tan coloured solid firm mass with smooth external surface measuring 12 x 6 x 4 cm. Cut section showed mostly solid with small cystic areas, small foci of necrosis but no haemorrhage [Table/Fig-1].
Gross photograph of ovarian tumour showing mostly solid areas with few small cystic spaces and necrosis.
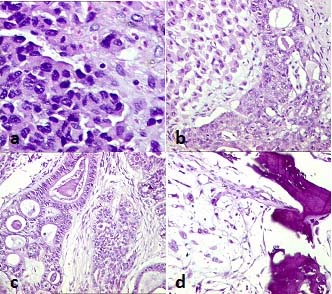
Microscopically, tumour tissue arranged in sheets and anastomosing trabecular pattern [Table/Fig-2a]. The cells were columnar to round and polygonal with scant to moderate amount of eosinophilic cytoplasm. Nuclear chromatin varied from vesicular in columnar cells to hyperchromatic in polygonal cells. Mitotic figures were few. In some foci, there were heterologous elements of two basic types like endodermal and mesodermal or mesenchymal. Stroma comprised of spindled hyperchromatic cells with few nests of round cells having abundant eosinophilic to foamy cytoplasm and centrally placed vesicular nuclei [Table/Fig-2b]. Acinar spaces of variable size lined by tall columanr mucin secreting cells of intestinal type were the heterologous endodermal element. Mesodermal element was represented by presence of irregularly shaped bony spicules with entrapped osteocytes [Table/Fig-2c&d].
(a) Nests of poorly differentiated sertoli cells (H&E stain x400); (b) Sheets of Leydig cells (H&E stain x400); (c & d) Heterologous elements-endodermal and mesenchymal (H&E stain x200).
Immunohistochemistry showed positivity for inhibin, calretinine and AE1AE3 [Table/Fig-3a-c], and negativity for cytokeratin conforming the diagnosis of SLCT. The case was pathologically diagnosed at stage-I disease with poorly differentiated histology. After surgery, serum inhibin, androgen, testosterone, estrogen levels were within normal limit.
Immunohistochemistry positive for a) inhibin; (40X); b) calretinin (40X); and c) AE1AE3 (10X).

Patient received three cycles of BEP (bleomycin: 15 units/m2 day1, etoposide: 100 mg/m2 day1-5, cisplatin: 25 mg/m2 day1-5) chemotherapy regimen (every three weekly) due to the presence of the heterologous elements on microscopic examination and tolerated well.
Discussion
The cells derived from stromal component of ovary and testis is responsible for origin of SCST. SLCT is included as a subtype among SCSTs. Ovarian SLCT is an extremely rare entity constituting 0.5% of all ovarian neoplasms [1]. Here, we reported a case of SLCT in a young woman and treated with unilateral salpingo-oophrectomy followed by adjuvant chemotherapy.
WHO defines SLCTs as the tumour composed of sertoli, leydig cell in varying proportions and in case of intermediate and poorly differentiated forms primitive gonadal stroma and sometimes heterologous elements [2].
Grossly, tumours are well encapsulated, solid, firm, lobulated, yellow or tan coloured with smooth external surface. Cut section shows greasy/fleshy areas with straw coloured fluids and cystic spaces, but necrosis and haemorrhage areas presents in poorly differentiated tumours [3]. Present case showed mostly solid with small cystic areas and necrosis without haemorrhage or papillary excrescences.
Microscopically, it is diagnosed by various degrees of differentiations of tubules lined by sertoli cells and by intervening nests of leydig cells, and includes well, intermediate and poorly differentiated forms [3]. Mitotic figures rarely exist in well/intermediate differentiated tumours and are abundant in poorly differentiated forms. 20% cases are associated with heterologous elements, both endodermal (gastric and/or intestinal type of mucin secreting epithelium) and mesodermal (immature cartilage, bone, and skeletal muscle) [4]. Endodermal elements are mostly present in intermediate differentiated forms, whereas, mesodermal elements are found in poorly differentiated tumours. Intermediate and poorly differentiated types form a continuum and infrequently varying degrees of differentiation can be present in the same tumour, as in the present case. SLCT of intermediate differentiation is diagnosed by detection of anastomosing cords and trabeculae of columnar cells.
In poorly differentiated SLCT sertoli cells are arranged in sheets or nests with foci of leydig cells and may exhibit mesodermal elements. Few foci of intestinal type epithelium [Table/Fig-2c], immature cartilage and bony spicules were detected in the present case [Table/Fig-2d]. Inhibin, calretin and AE1AE3 are positive and EMA is negative on immnuhistochemistry [5]. Immature teratoma is a differential diagnosis due to presence of heterologous elements. But it was ruled out due to absence of mature ectodermal tissue especially neural elements, neuroepithelial tubules, presence of sertoli cells, leydig cells and immunohistochemistry positive for inhibin, calretinine, AE1AE3.
Nearly 75% cases occur during 10-30 years of age group. Mostly patients present with symptoms of virillization due to excess androgen production like virilism, hirsutism, amenorrhoea, acne, alopecia, breast atrophy, clitoromegaly and hoarseness of voice. Presentation due to excess estrogen production is rare. Few patients present with dull aching abdominal pain, with swelling due to mass effect [1]. Symptoms of only pain abdomen and mass, as in the present case, creates difficulty in the investigation towards the diagnosis of SLCT and its management particularly in young women [6].
SLCTs may be purely solid, purely cystic or mixed. Ultrasonographically, it is solid appearance with intramural cystic component [6]. Staging and degree of differentiation are important prognostic factors [1]. Locoregional recurrence is rare in well differentiated early stage tumour. Metastasis may occur in poorly differentiated histology or advanced stage disease. Five-year overall survival is 100% in well differentiated form, 80% in moderate and poorly differentiated forms, 95% in stage-I disease and zero percent in stage-III/IV patients [1,7].
Surgery is the initial treatment of choice [8]. Conservative surgery should be done in young patients with stage-I disease. Unilateral salpingo-oophorectomy is the surgery of choice. Adjuvant chemotherapy should be considered in stage-I patients with risk factors (intermediate or poorly differentiated histology, heterologous elements, increased mitotic activity, rupture or spillage of the tumour), advanced disease and metastatic disease [1]. Stage-II disease onwards should be treated with radical hysterectomy with surgical staging followed by chemotherapy. Fertility sparing surgery may be considered in well differentiated stage-II disease [9].
Chemotherapy regimen used is: BEP, CAP (Cyclophosphamide, Adriamycin, Cisplatin) and PVB (Cisplatin, Vinblastin, Bleomycin) [10]. The present case was treated with unilateral salpingo-oophorectomy followed by three cycles of BEP chemotherapy.
Conclusion
Clinical presentation of pain abdomen with lower abdominal mass without symptoms of virillization and radiological finding of ovarian mass in a young woman is an important issue for the pretreatment diagnosis of SLCTs. Pretreatment serum markers like inhibin, androgen, testosterone and estrogen should be done in solid ovarian mass to rule out of SLCT. In patients desiring fertility appropriate management is an important issue. There is no standard protocol regarding management, especially chemotherapy and needs further evaluation.
[1]. Young RH, Scully RE, Ovarian Sertoli-Leydig cell tumours. A clinicopathological analysis of 207 casesAm J Surg Pathol 1985 9(8):543-69. [Google Scholar]
[2]. Dietrich JE, Kaplan A, Lopez H, Jaffee I, A case of poorly differentiated Sertoli-Leydig tumour of the ovaryJ Pediatr Adolesc Gynecol 2004 17:49-52. [Google Scholar]
[3]. Nouriani M, Felix JC, Dubeau L, Histogenesis and histopathological characteristics of Sertoli-Leydig cell tumorsCME J Gynecol Oncol 2002 7(2):114-20. [Google Scholar]
[4]. Mathur SR, Bhatla N, Rao IS, Manoj KS, Sertoli-Leydig cell tumor with heterologous gastrointestinal epithelium: A case reportIndian J Pathol Microbiol 2003 46(1):91-93. [Google Scholar]
[5]. McCluggage WG, Young RH, Immunohistochemistry as a diagnostic aid in the evaluation of ovarian tumorsSeminars in Diagnostic Pathology 2005 22(1):3-32. [Google Scholar]
[6]. Abu-Zaid A, Azzam A, Alghuneim LA, Metawee MT, Amin T, Al-Hussain TO, Poorly differentiated ovarian sertoli-leydig cell tumor in a 16-year-old single woman: a case report and literature reviewCase Reports in Obstetrics and Gynecology 2013 2013:1-6. [Google Scholar]
[7]. Sigismondi C, Gadducci A, Lorusso D, Candiani M, Breda E, Raspagliesi F, Ovarian Sertoli-Leydig cell tumors. A retrospective MITO studyGynecol Oncol 2012 125(3):673-76. [Google Scholar]
[8]. Weng CS, Chen MY, Wang TY, Tsai HW, Hung YC, Yu KJ, Sertoli-Leydig cell tumors of the ovary: a Taiwanese gynecologic oncology group studyTaiwan J Obstet Gynecol 2013 52(1):66-70. [Google Scholar]
[9]. Gui T, Cao D, Shen K, Yang J, Zhang Y, Yu Q, A clinicopathological analysis of 40 cases of ovarian Sertoli-Leydig cell tumorsGynecologic Oncology 2012 127(2):384-89. [Google Scholar]
[10]. Sachdeva P, Arora R, Dubey C, Sukhija A, Daga M, Singh DK, Sertoli-Leydig cell tumor: a rare ovarian neoplasm. Case report and review of literatureGynecol Endocrinol 2008 24(4):230-34. [Google Scholar]