Respiratory Symptoms as Prominent Manifestation of Brucellosis: A Case Series
Stamatis S Papadatos1, George Bazoukis2, Georgios Deligiannis3, Stefanos Mylonas4, Christos Zissis5
1 Faculty, Department of Internal Medicine, Athens School of Medicine, National and Kapodistrian University of Athens, Sotiria General Hospital, Athens, Greece.
2 Faculty, Department of Internal Medicine, Elpis General Hospital, Athens, Attica, Greece.
3 Faculty, Department of Cardiology, General Hospital of Karditsa, Karditsa, Thessaly, Greece.
4 Faculty, Department of Internal Medicine, General Hospital of Trikala, Trikala, Thessaly, Greece.
5 Faculty, Department of Internal Medicine, General Hospital of Trikala, Trikala, Thessaly, Greece.
NAME, ADDRESS, E-MAIL ID OF THE CORRESPONDING AUTHOR: Dr. Stamatis S Papadatos, Department of Internal Medicine, Athens School of Medicine, National and Kapodistrian University of Athens, Sotiria General Hospital, Athens-421 00, Greece.
E-mail: stamspap@gmail.com
Brucellosis is a zoonotic infection primarily affecting the reticuoendothelial system: spleen, liver and bone marrow. Despite the fact that multisystem infection in brucellosis is usually reported, pulmonary involvement is considered to be very rare. We report four cases (three male and one female, farm workers) of pulmonary brucellosis who presented with signs and symptoms of lower respiratory infection. All of them underwent thorough clinical examination and laboratory tests before the diagnosis of pulmonary brucellosis was confirmed. All patients were hospitalized for at least eight days and given appropriate antibiotic therapy. Follow up after 12-24 months showed no lasting effects from the infection and laboratory tests were found within the normal limits.
Brucella, Pneumonia, Pulmonary brucellosis, Zoonotic infection
Introduction
Brucellosis is a zoonotic bacterial infection caused by the genus Brucella. These small aerobic intracellular coccobacilli, which are Gram-negative and non spore forming, shed in the urine, milk, placenta and other fluids of animals, mainly domestic. Humans are accidental hosts [1]. Transmission is generally achieved either through direct contact with contaminated animals, or through ingestion of unpasteurized dairy products and inhalation of infectious aerosol particles, suggesting that Brucellae enter the human body through skin or mucous discontinuities [2]. Symptoms include fatigue, malaise, anorexia and body aches. Fever is the most common sign [3]. Pulmonary involvement rarely occurs as a result of inhalation of infected aerosol or hematogenous spread and pulmonary manifestations including pleural effusions and pneumonias, can be found in up to 16% of complicated cases [4].
Case Series
Case 1
A 55-year-old shepherd was admitted to the Emergency Department (ED) by a general practitioner due to persistent fever, along with productive cough and fatigue that started eight days ago and did not respond to empiric antibiotic therapy with cefuroxime axetil. Physical examination was unremarkable and laboratory exams revealed mild leukopenia, elevated C-Reactive Protein (CRP) and slightly high Erythrocyte Sedimentation Rate (ESR) [Table/Fig-1]. Chest x-ray was negative for lobar pneumonia but ground glass opacity was found in high resolution Computerized Tomography (CT) scan. The diagnosis was confirmed by positive Wright agglutination and detection of IgM antibodies by ELISA. Initiation of anti-brucella treatment with doxycycline 100 mg P.O, twice daily and rifampicin 600 mg/day P.O, resulted in significant clinical improvement on day three. The patient remained asymptomatic and on day 10 and he was discharged. Follow-up at three months and 12 months was unremarkable.
The characteristics, clinical and laboratory findings of each patient.
| No | Sex | Age | Possible Exposure | Prominent Symptoms and Signs | Other Symptoms | Lung CT Scan | WBC(103/mm3) | PLT(103/mm3) | CRP(mg/l) | ESR(mm/h) | Other Lab finding | Cultures | WRIGHT test | ELISAIgM |
|---|
| blood | sputum |
|---|
| 1 | M | 55 | Shepherd | Fever, productive cough | Fatigue | Ground glass opacity | 4.0 | 172 | 9.4 | 32 | None | – | – | + 1/1280 | 3,8 (+) |
| 2 | M | 49 | Farmer | Low-grade-fever, non productive cough, bronchospasm | Pain in joints, anorexia | Pulmonary nodules | 6.8 | 220 | 15.7 | 50 | SGOT=68SGPT=70(U/L)Sacroiliitis | + | – | +1/640 | 4.2 (+) |
| 3 | F | 68 | Shepherd | Fever, paroxysmal cough | Fatigue, pain in joints | Basal right side pneumonia, thoracic lemphadenopathy | 4.1 | 102 | 8.3 | 38 | None | – | – | +1/320 | 3 (+) |
| 4 | M | 72 | Farmer | Fever, dyspnoea, hypoxemia | Anorexia | tree-in-bud sign | 9.9 | 198 | 11.2 | 49 | None | – | – | +1/320 | 3.2 (+) |
WBC: White Blood Cells, PLT: Platelets, CRP: C-reactive protein, ESR: Erythrocyte Sedimentation Rate, SGOT: Serum Glutamil Oxaloacetic Transaminase, SGPT: Serum Glutamic Pyruvic Transaminase, (+): positive, (-): negative, M: Male, F: Female
Case 2
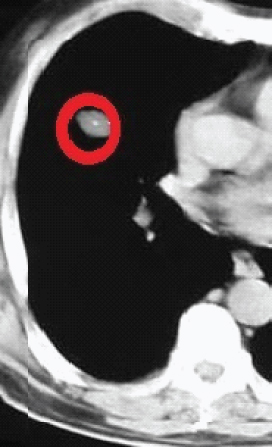
A 49-year-old, never-smoker farmer presented to the ED with low grade fever and fatigue for more than 15 days. He had been treated for brucellosis with bone involvement 12 months before the onset of the new symptoms. He complained of pain in joints and non productive cough as well. The auscultation revealed true bronchospasm wheezes in the left hemithorax. Inflammatory markers were high [CRP: 15.7 mg/l (0-1mg/l), ESR: 50 mm/hr (<25 mm/hr)], the liver biochemistry was abnormal [Serum Glutamil Oxaloacetic Transaminase (SGOT): 68 U/L (10-30 U/l), Serum Glutamic Pyruvic Transaminase (SGPT): 70 (10-30 U/l)] [Table/Fig-1] and pulmonary nodules were observed in the CT scan [Table/Fig-2]. Two out of three blood cultures BACTEC Plus Aerobic/F and Anaerobic/F [Becton Dickinson, Allschwil, Switzerland], incubated for 10 days were positive for Brucella species. The Wright agglutination test and the ELISA IgM antibodies set the diagnosis. The patient was treated for brucellosis relapse with six week doxycycline (200 mg/day, orally) plus rifampicin (900 mg/day, orally) combined with gentamicin (5 mg/kg, intramuscularly) for the initial two weeks. The patient was lost to follow up.
Pulmonary nodule in the chest CT scan of a patient (hemithorax) with pulmonary brucellosis was a non specific finding.
Case 3
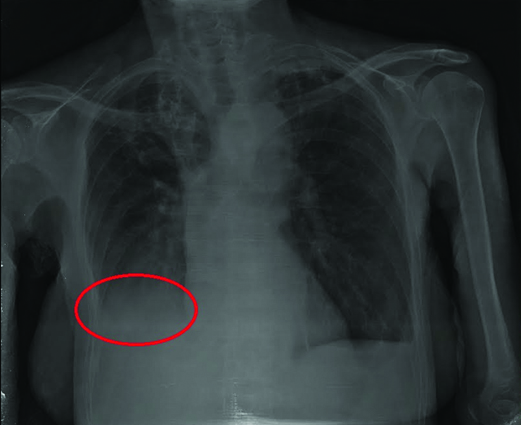
A 68-year-old hypertensive female who lived in a rural area and had received anti Brucella treatment 17 years ago was hospitalized for fever, paroxysmal cough and joint pain without signs of arthritis. CRP was elevated but ESR was within the normal range. Leukocytes and platelets were slightly low [Table/Fig-1]. Chest x-ray was consistent with lobar, basal right side pneumonia and showed ground glass opacities [Table/Fig-3]. The CT scan revealed thoracic lymphadenopathy as well. Having positive Wright agglutination test and ELISA IgM antibodies, the patient was treated for brucella reinfection with doxycycline 200 mg/day P.O. and rifampicin 600 mg/day P.O. for six weeks. Semiannual clinical and paraclinical follow up revealed total resolution of the infection.
Basal pneumonia in the chest x-ray of a woman with pulmonary Brucellosis.
Case 4
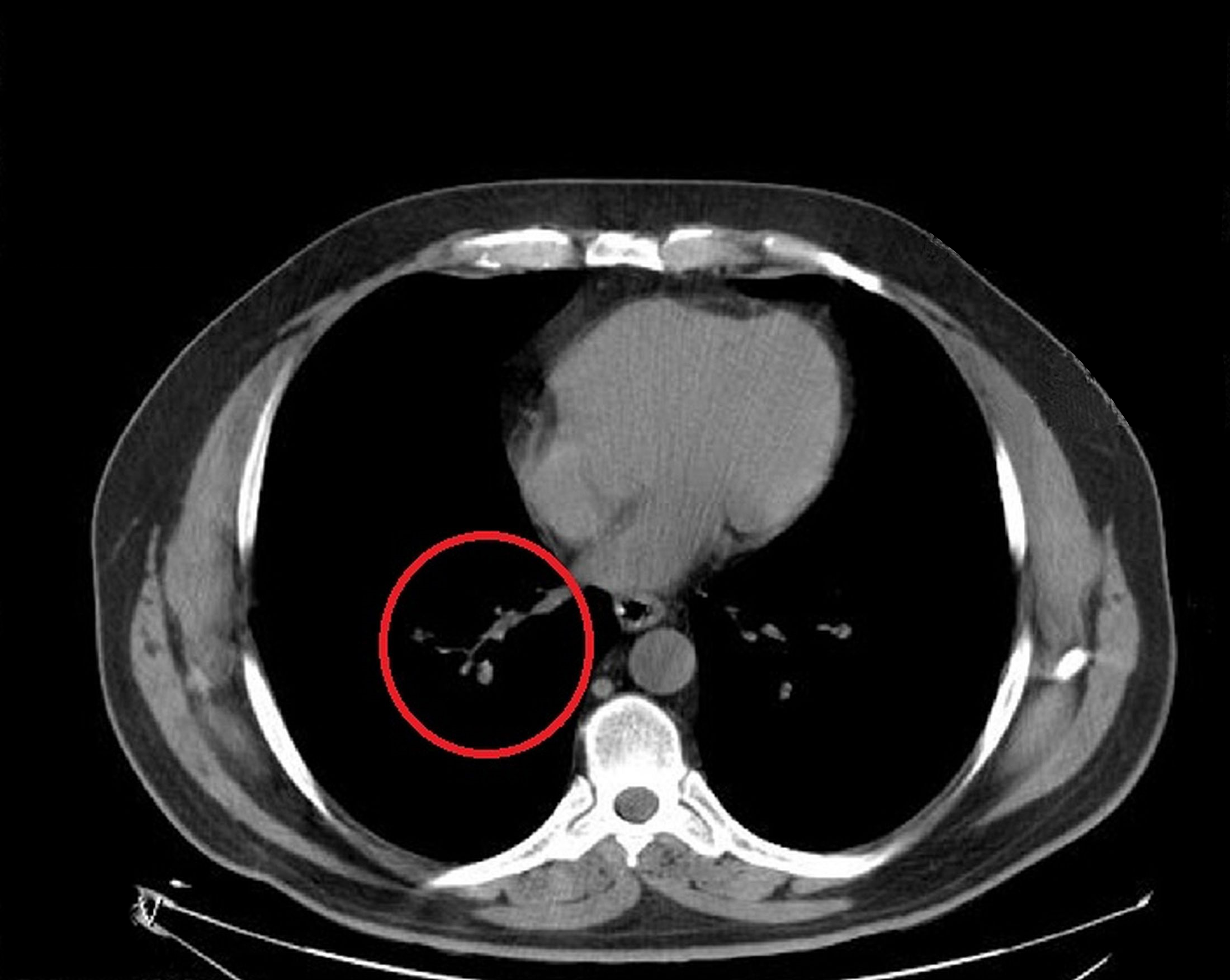
A 72-year-old male farmer presented to the ED complaining about gradually established with dyspnoea, fever and productive cough. He had past medical history of chronic ischemic heart disease. He was hypoxemic and, therefore, he underwent a lung CT scan which revealed a tree-in-bud sign [Table/Fig-4]. The patient had been treated for Brucellosis 21 years ago. Sputum and blood cultures were negative but Wright agglutination test as well as Elisa IgM antibodies set the diagnosis for Brucella reinfection with pulmonary involvement [Table/Fig-1]; the patient received triple antibiotic therapy with intravenous gentamicin 5 mg/kg for seven days and then with doxycycline 100 mg P.O. twice daily and rifampicin 600 mg/day P.O. for 45 days. The patient was followed up for two years and no sign of relapse was detected.
Chest CT scan of a patient with pulmonary Brucellosis [Case 4]. Note the tree-in-bud sign.
Discussion
The most common and virulent species of Brucella that infect human are Brucella melitensis (typically associated with sheep and goats) and Brucella abortus (associated with cattle) [5]. Human brucellosis generally occurs through direct exposure to the pathogen. All of our patients had direct contact with domestic animals or their raw products before the onset of the symptoms. Any organ may be affected. One of our patients had additional liver involvement while another one had previously been treated for osteoarticular brucellosis. Pulmonary involvement in brucellosis may occur through inhalation and therefore, the immune system associated with the lungs play an important role in mediating the initial recognition of infection [6]. Nonetheless, Brucella species are intracellular pathogens and, therefore, they manage to avoid the cellular immune response and establish infections for prolonged periods [7]. The onset of symptoms may be acute or insidious and almost always they are non specific [8]. The general symptoms of our patients were non specific as well. The initiation of symptoms for one of our patients was considered to be acute and, therefore, we assumed that he suffered an acute, first ever pulmonary brucellosis. As far as laboratory tests are concerned, thrombocytopenia and leukopenia are thought to be strongly associated with brucellosis [9], yet only two of our patients had an abnormal total blood count. There are few reports of respiratory involvements and a wide spectrum of lung disease has been reported [10]; empyema [11], pleural effusion [12], solitary nodules [13], interstitial pneumonia [14] and even pneumothorax [15] and pulmonary embolism [16] have all been reported and they can even be the sole symptom of brucellosis. The clinical manifestations of our four patients were consistent with lower respiratory infection. Due to prolonged and non specific symptoms, tuberculosis, granulomatous diseases and tumors should not be neglected in the differential diagnosis of pulmonary Brucellosis [8]. Isolation of Brucella spp. from any clinical specimen or detection of agglutinating antibodies combined with detection of non agglutinating antibodies or PCR confirm the diagnosis [5].
Conclusion
Brucellosis is a systemic infection, signs and symptoms of which are non specific. Keeping in mind that brucellosis is a good mimic, clinicians should always consider it in any patient with personal history of exposure to a possible source of infection.
[1]. Pappas G, Papadimitriou P, Akritidis N, Christou L, Tsianos EV, The new global map of human brucellosisThe Lancet Infectious Diseases 2006 6:91-99. [Google Scholar]
[2]. Pappas G, Akritidis N, Bosilkovski M, Tsianos E, BrucellosisN Engl J Med 2005 352:2325-36. [Google Scholar]
[3]. Franco MP, Mulder M, Gilman RH, Smits HL, Human brucellosisThe Lancet Infectious Diseases 2007 7:775-86. [Google Scholar]
[4]. Hatipoglu CA, Bilgin G, Tulek N, Kosar U, Pulmonary involvement in brucellosisThe Journal of Infection 2005 51:116-19. [Google Scholar]
[5]. Corbel M, Elberg SS, Cosivi O, Brucellosis in Humans and Animals 2006 GenevaWorld Health Organisation [Google Scholar]
[6]. Archambaud C, Salcedo SP, Lelouard H, Devilard E, de Bovis B, Van Rooijen N, Contrasting roles of macrophages and dendritic cells in controlling initial pulmonary Brucella infectionEuropean Journal of Immunology 2010 40:3458-71. [Google Scholar]
[7]. Skendros P, Boura P, Immunity to brucellosisRevue Scientifique Et Technique 2013 32:137-47. [Google Scholar]
[8]. Takahashi H, Tanaka S, Yoshida K, Hoshino H, Sasaki H, Takahashi K, An unusual case of brucellosis in Japan: difficulties in the differential diagnosis from pulmonary tuberculosisInternal Medicine 1996 35:310-14. [Google Scholar]
[9]. Bouley AJ, Biggs HM, Stoddard RA, Morrissey AB, Bartlett JA, Afwamba IA, Brucellosis among hospitalized febrile patients in northern TanzaniaThe American Journal of Tropical Medicine and Hygiene 2012 87:1105-11. [Google Scholar]
[10]. Pappas G, Bosilkovski M, Akritidis N, Mastora M, Krteva L, Tsianos E, Brucellosis and the respiratory systemClin Infect Dis 2003 37(7):e95-99. [Google Scholar]
[11]. Apostolova E, Papadopoulos V, Leptidou-Kerestetzi T, Branchial cyst empyema due to Brucella melitensis infection as a form of focal BrucellosisThe Journal of Infection 2002 44:271 [Google Scholar]
[12]. Eruz ED, Birengel S, Azap A, Bozkurt GY, A case of brucellosis presenting with multiple hypodense splenic lesions and bilateral pleural effusionsCase Reports in Medicine 2011 2011:614546 [Google Scholar]
[13]. Cobo F, Aliaga L, de Cueto M, Cueto A, de La Rosa M, Solitary lung nodules produced by Brucella melitensisEnfermedades Infecciosas Y Microbiologia Clinica 2001 19:452-53. [Google Scholar]
[14]. Solera J, Solis Garcia, Del Pozo J, Treatment of pulmonary brucellosis: a systematic reviewExpert Review of Anti-Infective Therapy 2017 15:33-42. [Google Scholar]
[15]. Patel PJ, Al-Suhaibani H, Al-Aska AK, Kolawole TM, Al-Kassimi FA, The chest radiograph in brucellosisClinical Radiology 1988 39:39-41. [Google Scholar]
[16]. Sen T, Cagli K, Golbasi Z, Cagli K, Thrombus-in-transit entrapped in a patent foramen ovale: a complication of brucellosisTurk Kardiyol Dern Ars 2011 39(6):487-90. [Google Scholar]