Good anatomic outcomes have been achieved with transvaginal synthetic materials in pelvic organ prolapse, but mesh-related complications are a major health concern. Therefore, developing new synthetic or biological materials with increased tissue strength and decreased complications are major challenges. This paper presents preliminary pilot series of Posterior Intravaginal Slingoplasty (PIVS) procedure performed with mixed material graft containing both autologous tissue and a synthetic material. Six women with symptomatic Utero-Vaginal Prolapse (UVP) ≥ stage 2 underwent the PIVS procedure. Pelvic floor symptoms were assessed with the Pelvic Organ Prolapse Quantification (POP-Q) system, Pelvic Floor Distress Inventory (PFDI), and the Pelvic Floor Impact Questionnaire (PFIQ) both before the procedure and 36 months after the surgery. There were no mesh-related complications or recurrence and all the patients had good functional and anatomic outcomes. These preliminary results are promising, but further long-term studies with a larger series are needed.
Introduction
UVP is a common health issue, which is associated with a variety of urinary, bowel, sexual, and social problems [1]. The cardinal and utero-sacral ligaments mainly support the uterus and upper vagina. Defects in these ligaments result in the loss of apical support and the occurrence of UVP [1]. The incidence of POP will increase in the coming decades as the elderly population increases [2]. The traditional abdominal route in surgery is associated with long operation times, high costs, and surgical risk [2]. These problems have led surgeons to seek minimally invasive procedures or to modify existing procedures, especially in patients who are poor candidates for surgery due to medical co-morbidities [2].
PIVS was first reported by Petros PE as a minimally invasive procedure for the treatment of vaginal vault prolapse [3]. Traditional operating procedures for UVP have a long learning curve. In contrast, PIVS is a minimally invasive procedure with a short learning curve, a short operation time, and requires mostly regional anaesthesia [4]. High cure rates have been reported with the procedure, with success rates varying from 75% to 98% in short-term follow up studies [3,5,6]. Recurrence rates have ranged from 0.3% to 8.8% [7,8]. Recently, Copobianco G et al., reported a 93.18% success rate for the PIVS procedure based on the results of a nine year follow up [9].
In 2008, the Food and Drug Administration (FDA) issued a public health notification, followed by an update in 2011 regarding concerns about increased rates of complications associated with the use of vaginal mesh [10,11]. Although, the success rate with the PIVS procedure is comparable to that of many operative techniques for UVP, but, because of mesh related complications many surgeons are reluctant to perform this procedure [6-13]. Tape erosion, pelvic infection, dyspareunia, pelvic pain, and sexual dysfunction are the most commonly reported morbidities associated with PIVS [6-13].
New synthetic or biological materials with increased tissue strength and decreased complications are essential to optimize surgical results. We designed a mixed material graft composed of a polypropylene mesh combined with vaginal tissue. By combining natural and synthetic tissue, we aimed to decrease mesh-related complications without reducing the tissue strength. Six women with symptomatic UVP ≥ stage 2 who were poor candidates for abdominal surgery (morbid obesity, prior two or more abdominal surgery, cardiac or respiratory diseases) underwent the PIVS procedure with the mixed material graft. This paper presents the anatomic and functional outcomes of this PIVS case series.
Cases
Six patients underwent the PIVS procedure with the mixed material graft at Kocaeli University, Medicine School, Department of Obstetrics and Gynaecology. The study was performed in accordance with the principles of the Declaration of Helsinki, and it was approved by the Local Ethics Committee of the board. The patients were all informed about the operation procedure, and they were given detailed information about the possible risks and complications. All the patients filled a consent form.
Six women with symptomatic UVP presented to the clinic. The prolapse was staged by the POP-Q classification system of the International Continence Society [14]. All the patients had some degree of cysto-rectocele and uterine prolapse ≥ stage 2 and all patients were poor candidates for abdominal surgery. The patients were aged between 30 and 64 years. None of the patients had urinary or anal incontinence. Information on patient characteristics, such as age, parity, type of delivery, co-morbid medical problems, sexual activity, desire for uterus preservation, and the presence of urinary and anal incontinence, was also obtained. The patients’ characteristic findings are presented in [Table/Fig-1]. All the patients underwent PIVS and anterior–posterior colporraphy.
Preoperative patient characteristics.
| Characteristics | Cases |
|---|
| 1 | 2 | 3 | 4 | 5 | 6 |
|---|
| Age (years) | 51 | 37 | 63 | 64 | 30 | 56 |
| Gravidity | 7 | 6 | 10 | 9 | 2 | 5 |
| Parity | 4 | 4 | 7 | 7 | 2 | 5 |
| Abortion | 3 | 2 | 3 | 2 | 0 | 0 |
| POP-Q Stage of UP | 3 | 2 | 4 | 3 | 2 | 4 |
| Cystocele Stage | 1 | 2 | 2 | 2 | 1 | 2 |
| Rectocele Stage | 2 | 1 | 2 | 1 | 2 | 2 |
| Menopausal Status | M | R | M | M | R | M |
| Type of Delivery | VD | VD | VD | VD | VD | VD |
(VD: vaginal delivery, M:menopause, R:reproductive age, UP:uterine prolapse)
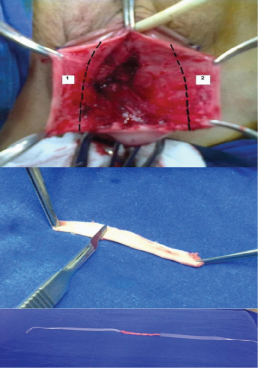
Mixed material tape preparation for PIVS: Full-thickness 5 cm long and 8 mm wide strips of vaginal tissue were extracted bilaterally from the vagina during the cystocele operation [Table/Fig-2a]. A scalpel was used to scrape the outer surface of the vaginal epithelium [Table/Fig-2b]. Each 5 cm long vaginal tape was then sutured at the end with No. 3/0 polypropylene sutures to produce a 10 cm long vaginal tape. Lastly, 8 mm wide monofilament polypropylene tape was sutured to each side of the 10 cm long vaginal tissue with No. 3/0 polypropylene [Table/Fig-2c]. Thus, mixed material graft was obtained, with vaginal tissue in the center and mesh arms at the sides.
Preperation of mixed material graft containing both autologous vaginal tissue and a synthetic material. a) The extraction of excess vaginal tissue of ~5 cm from each side during the cystocele operation, b) Preparation of the vaginal tissue, showing the outer surface of the vaginal epithelium being scraped with a scalpel, c) The integration of the full-thickness vaginal tissue into the polypropylene tape (The polypropylene tapes were sutured with No. 3/0 polypropylene to the newly formed (~10 cm) vaginal tissue on each side). (Images up to down)
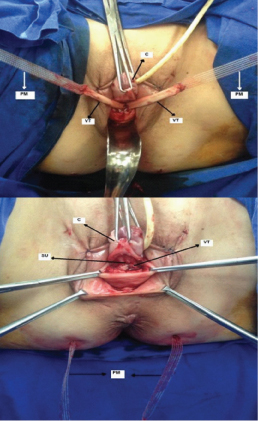
PIVS surgery: The mixed material graft was used in all the PIVS procedures, which were performed as described by Petros PE [3] by the same operator. After ensuring the tape remained flat, the center part of the newly formed tape (vaginal tape) was sutured securely with interrupted No. 3/0 polypropylene sutures to the utero-sacral ligaments [Table/Fig-3a]. The mesh arm of the new tape was then attached to the tip of the needle and pulled back out of the buttock skin incision. The same procedure was done on the other side. So, the vaginal tissue-based tape with the polypropylene mesh arms was then pulled through the buttock incisions [Table/Fig-3b].
The application of mixed material graft in the PIVS procedure. a) Suturing the mixed material graft from the center of the vaginal tissue to the utero-sacral ligaments, b) The vaginal tissue-based tape with the polypropylene mesh arms was then pulled through the buttock incisions. (Images up to down) (PM: polypropylene mesh, C: cervix, VT: vaginal tissue, SU: sacrouterine ligament).
Intraoperative complications were recorded. A complete blood count was obtained 24 hour after the surgery. If the haemoglobin level decrease was >2 gm/dl, it was accepted as blood loss. All the patients were examined both before and after the operation. To decrease bias, a specialist blinded to the surgery evaluated the anatomic outcomes of the patients before and after the surgery. Pelvic floor symptoms were assessed using the PFDI and the PFIQ [14] before and 36 months after the surgery. Surgical outcomes of UVP were assessed by the POP-Q classification and the 9 points were evaluated to assess the anatomic results [Table/Fig-4]. A cure was defined as postoperative POP-Q stage 0 based on point C. Improvement was defined as stage I. Failure was accepted if the postoperative POP-Q was stage II or greater. Patient satisfaction with the surgery was assessed on a 3-point Likert scale.
Pelvic organ prolapse quantification scores before and after the operation according to cases.
| Ap | Aa | Bp | Ba | C | D | GH | PB | TVL | * |
|---|
| B/A | B/A | B/A | B/A | B/A | B/A | B/A | B/A | B/A | ** |
|---|
| 1 | +1/-3 | +2/-3 | +2/-3 | +3/-3 | +4/-4 | +4/-6 | 3/3 | 1.5/2 | 4/6 | |
| 2 | +2/-3 | +2/-3 | +3/-3 | +4/-3 | +2/-6 | +2/-8 | 3/3 | 2/2 | 6/8 | |
| 3 | +3/-3 | +3/-3 | +4/-3 | +5/-3 | +8/-7 | +8/-6 | 4/3.5 | 1.5/2 | 4/8 | |
| 4 | +2/-3 | +1/-3 | +3/-3 | +3/-3 | +2/-4 | +2/-6 | 3/3 | 2.5/3 | 4/6 | |
| 5 | +2/-3 | +2/-3 | +4/-3 | +3/-3 | +2/-6 | +2/-8 | 4/3.5 | 2/2 | 6/8 | |
| 6 | +2/-3 | +2/-3 | +2/-3 | +3/-3 | +2/-4 | +3/-6 | 3/3 | 2/2 | 4/6 | |
* Aa: Anterior vaginal wall 3 cm proximal to the hymen, Ap: Posterior vaginal wall 3 cm proximal to the hymen, Ba: Most dital position of the remaining upper anterior vaginal wall, Bp: Most dital position of the remaining upper posterior vaginal wall, C: Most distal edge of cervix, D: Posterior fornix, GH: Genital hiatus, PB: Perineal body, TVL:Total vaginal lenght
**B: Before operation; A: After operation
The postoperative course: The postoperative course of all the patients was uneventful. None of the patients had intraoperative bladder, bowel, or rectal injuries, intraoperative blood loss, urinary retention, or urinary infections due to the surgery. All the patients had stage 0 apical support 36 months after the surgery. There were no tape-related complications, such as erosion, extrusion, shrinkage. The PFDI and PFIQ scores before the procedure and 36 months after the operation are presented in [Table/Fig-5]. Both the PFDI and PFIQ scores decreased significantly (p<0.05). Based on a 3-point Likert scale, all the patients were highly satisfied with the surgery.
Pelvic Floor Distress Inventory (PFDI) and Pelvic Floor Impact Questionnaire (PFIQ) scores before and 32 months after the operation.
| Case | Pelvic Floor Distress Inventory |
|---|
| Urinary Distress Inventory | Pelvic Organ Prolapse Distress Inventory | Colorectal-Anal Distress Inventory | Total |
|---|
| B | A | B | A | B | A | B | A* |
|---|
| 1 | 50 | 16.6 | 62.5 | 4.16 | 9.375 | 6.25 | 121.875 | 27.01 |
| 2 | 37.5 | 16.6 | 33.3 | 0 | 0 | 15.6 | 70.8 | 32.2 |
| 3 | 45.8 | 12.5 | 20.8 | 0 | 3.12 | 0 | 69.72 | 12.5 |
| 4 | 45.8 | 8.3 | 33.3 | 4.16 | 9.37 | 0 | 88.47 | 12.46 |
| 5 | 16.6 | 12.5 | 37.5 | 4.16 | 6.25 | 3.125 | 60.35 | 19.785 |
| 6 | 45.8 | 12.5 | 54.1 | 4.16 | 0 | 0 | 99.9 | 16.66 |
| Pelvic Floor Impact Questionnaire |
| Urinary Impact Questionnaire | Pelvic Organ Prolapse Impact Questionnaire | Colorectal-Anal Impact Questionnaire | Total |
| B | A | B | A | B | A | B | A |
| 1 | 9.5 | 4.7 | 47.6 | 0 | 0 | 0 | 57.1 | 4.7 |
| 2 | 42.8 | 9.5 | 28.5 | 9.5 | 4.7 | 19.04 | 76 | 38.04 |
| 3 | 52.3 | 23.8 | 23.8 | 9.5 | 4.7 | 0 | 80.8 | 33.3 |
| 4 | 57.14 | 9.52 | 42.8 | 14.2 | 0 | 0 | 99.94 | 23.72 |
| 5 | 38 | 23.8 | 33.3 | 9.5 | 0 | 0 | 71.3 | 33.3 |
| 6 | 23.8 | 9.5 | 28.5 | 9.5 | 0 | 0 | 52.3 | 19 |
(*B: Before operation; A: After operation)
Discussion
Selecting the type of surgery is one of the major challenges faced by surgeons. Surgery decisions in POP depend on the patient’s desires, type, and severity of the POP [2]. PIVS has some benefits, such as a shorter operation time with regional anaesthesia and a shorter recovery time and hospitalization, compared to traditional approaches [4]. Apart from the anatomical support, success of vaginal surgery depends on functional outcomes as quality of life, genitourinary symptoms and sexual life [15]. PIVS is a minimal invasive procedure with comparable functional and anatomical outcomes [16]. However, mesh-related complications discourage many surgeons from performing this procedure [10,11]. Mesh-related complications after PIVS have been reported in 0%–17% of cases in the literature [6-13]. Therefore, new tape materials are needed to optimize surgical outcomes.
The use of endogenous tissue in reconstructive surgery is an attractive idea. However, its long-term durability varies and are conflicting in the literature [17]. Raz S et al., reported that urinary incontinence due to intrinsic sphincteric damage was successfully treated with a sling constructed from the vaginal wall tissue [17]. However, the use of full-thickness vaginal tissue combined with mesh arms in reconstructive surgery for POP was not described previously. To our knowledge, this paper is the first such presentation.
The anatomic success of PIVS is well known, but mesh-related complications in the vagina and surrounding tissue are recognized problems with this procedure. To address these problems, materials with optimal tissue strength and a minimum risk of erosion are needed. Vaginal tissue part of mixed material graft covers the vagina and rectum without harming them, while the mesh arms pull uterus strongly. The postoperative course was uneventful in the patients, and the new tape provided good anatomical support 36 months after the surgery.
Conclusion
These preliminary results are the first case series of PIVS patients treated with a combination of autologous tissue and synthetic mesh. These results are promising, with good anatomical and functional outcomes. However, further evaluations with a larger series are needed to determine the optimal biomechanical properties with long-term use.
(VD: vaginal delivery, M:menopause, R:reproductive age, UP:uterine prolapse)
* Aa: Anterior vaginal wall 3 cm proximal to the hymen, Ap: Posterior vaginal wall 3 cm proximal to the hymen, Ba: Most dital position of the remaining upper anterior vaginal wall, Bp: Most dital position of the remaining upper posterior vaginal wall, C: Most distal edge of cervix, D: Posterior fornix, GH: Genital hiatus, PB: Perineal body, TVL:Total vaginal lenght
**B: Before operation; A: After operation
(*B: Before operation; A: After operation)
[1]. Ashton-Miller J, Delancey JO, Functional anatomy of the female pelvic floorAnn NY Acad Sci 2007 1101:266-96. [Google Scholar]
[2]. Siddiqui NY, Edenfield AL, Clinical challenges in the management of vaginal prolapseInt J Womens Health 2014 6:83-94. [Google Scholar]
[3]. Petros PE, New ambulatory surgical methods using an anatomical classification of urinary dysfunction improve stress, urge and abnormal emptyingInt Urogynecol J Pelvic Floor Dysfunct 1997 8:270-77. [Google Scholar]
[4]. Nyyssönen V, Talvensaari-Mattila A, Santala M, Posterior ıntravaginal slingplasty versus unilateral sacrospinous ligament fixation in treatment of vaginal vault prolapseISRN Obstet Gynecol 2013 2013:958670 [Google Scholar]
[5]. Maher C, Feiner B, Baessler K, Adams EJ, Hagen S, Glazener CM, Surgical management of pelvic organ prolapse in womenCochrane Database Syst Rev 2010 14:CD004014 [Google Scholar]
[6]. Nyyssönen V, Talvensaari-Mattila A, Santala M, Intravaginal slingplasty sling is associated with increased risk of vaginal erosionActa Obstet Gynecol Scand 2009 88:1222-26. [Google Scholar]
[7]. Luck AM, Steele AC, Leong FC, McLennan MT, Short-term efficacy and complications of posterior intravaginal slingplastyInt Urogynecol J Pelvic Floor Dysfunct 2008 19:795-99. [Google Scholar]
[8]. Chen HY, Ho M, Chang YY, Hung YC, Chen WC, Risk factors for surgical failure after posterior intravaginal slingplasty: a case seriesEur J Obstet Gynecol Reprod Biol 2011 155:106-09. [Google Scholar]
[9]. Capobianco G, Donolo E, Wenger JM, Madonia M, Cosmi E, Antimi L, Efficacy and 9 years’ follow-up of posterior intravaginal slingplasty for genital prolapseJ Obstet Gynaecol Res 2014 40:219-23. [Google Scholar]
[10]. Chermansky CJ, Winters JC, Complications of vaginal mesh surgeryCurr Opin Urol 2012 22:287-91. [Google Scholar]
[11]. US Food and Drug Administration Update on serious complications associated with transvaginal placement of surgical mesh for pelvic organ prolapse. 2011. http://www.fda.gov/medicaldevices/safety/alertsandnotices/ucm262435.htm [Google Scholar]
[12]. Cosma S, Preti M, Mitidieri M, Petruzzelli P, Possavino F, Menato G, Posterior intravaginal slingplasty: efficacy and complications in a continuous series of 118 casesInt Urogynecol J 2011 22:611-19. [Google Scholar]
[13]. Lee YS, Han DH, Lee JY, Kim JC, Choo MS, Lee KS, Anatomical and functional outcomes of posterior intravaginal slingplasty for the treatment of vaginal vault or uterine prolapse: a prospective, multicenter studyKorean J Urol 2010 51:187-92. [Google Scholar]
[14]. Barber MD, Walters MD, Bump RC, Short forms of two condition-specific quality-of-life questionnaires for women with pelvic floor disorders (PFDI-20 - PFIQ-7)Am J Obstet Gynecol 2005 193:103-13. [Google Scholar]
[15]. Ulrich D, Dwyer P, Rosamilia A, Lim Y, Lee J, The effect of vaginal pelvic organ prolapse surgery on sexual functionNeurourol Urodyn 2015 34(4):316-21. [Google Scholar]
[16]. Ilhan TT, Sivaslioglu A, Ilhan T, Uçar MG, Dolen I, Comparison of the efficiency of Posterior Intravaginal Sling (PIVS) procedure in older and younger groupsJ Clin Diagn Res 2016 10(7):QC05-07. [Google Scholar]
[17]. Raz S, Siegel AL, Short JL, Snyder JA, Vaginal wall slingJ Urol 1989 141:43-46. [Google Scholar]