Blood transfusion reduces morbidity in many clinical diseases and conditions but there is a potential risk of TTIs associated with it. This makes vigilant screening of blood a mandatory process. Trends and prevalence of infections among healthy individuals may be monitored and estimated by screening of TTIs among blood donors [1].
In India, the main sources of blood for transfusion are voluntary donors and replacement donors. Replacement donors are the relatives or close friends of a patient. Voluntary donors are preferred over replacement donors because replacement donor may tend to hide some history with the intention of benefitting their patient, out of ignorance about the fact that they are putting someone’s life in danger. The prevalence of HIV, hepatitis viruses and other blood borne infections is lowest among voluntary unpaid blood donors and highest among family or family replacement donors who give blood only when it is required by a member of the patient’s family or community [2].
Present study was conducted to determine the prevalence of TTIs among the blood donors, which can further help in determination of the population subset to be focused on for enhancing blood donor pool and to compare the prevalence with similar other studies done in India and other parts of the world.
Materials and Methods
This retrospective cross-sectional study was conducted at Blood Bank of G.B Pant Hospital, Andaman & Nicobar Islands institute of Medical Sciences, Port Blair, India. The study was conducted on all voluntary and replacement donors for a period of three years from April 2013 to March 2016. Voluntary blood donation was conducted in the blood bank as well as various blood donation camps.
Pre donation screening
Donors were made to go through strict donor selection criteria which includes thorough history taking about past illness, medical treatments, high risk behaviour, time since last donation if any, etc., along with brief physical examination. Donor was asked to go through a questionnaire which aimed at both donor and recipient’s blood safety.
Though every unit of blood taken was screened for HCV, HBsAg, HIV, syphilis & malaria, but the questionnaire focussed on avoiding the window period, in which the infection, if present, was undetectable. It also aimed to avoid donors who were at risk of carrying a latent infection. Also, for the donor’s safety, various factors like time since last meal, sleep, presence of any chronic illness, general well being, etc., were taken into consideration. In physical examination, vitals like temperature, blood pressure, pulse & respiratory rate were recorded. According to regulations, haemoglobin levels had to be above 12.5 gm%, weight above 45kg, age between 18 to 60 years. For females, history of menstruation, last pregnancy/delivery and history of lactation was taken. Donor contact information in the form of phone number or e-mail was noted. A written consent was taken from every donor.
TTIs testing
After a donor passed pre-donation screening, blood unit was collected in a single, double, triple or Saline Adenine Glucose Mannitol (SAGM) bag, according to the requirements of various blood components. Every unit of blood was screened for anti-HCV, HBsAg, Anti- HIV, Syphilis antibody & Malaria parasite antigen. Rapid tests were done by RAPID kits approved & Licensed by National Aids Control Society (NACO). Test protocol and test interpretation was according to guidelines laid by NACO. Those samples which were tested with rapid kit were again tested with enzyme-linked immunosorbent assay (ELISA) for HIV, HbSAg and HCV. Thus all the samples were tested with ELISA once. For syphilis, Rapid Plasma Reagin (RPR) was done. All the positive donors were informed telephonically, by post or by e-mail. Strict confidentiality was maintained for the positive results. The positive donors were referred to Integrated Testing and Counselling Centre (ICTC) except for malaria.
Results
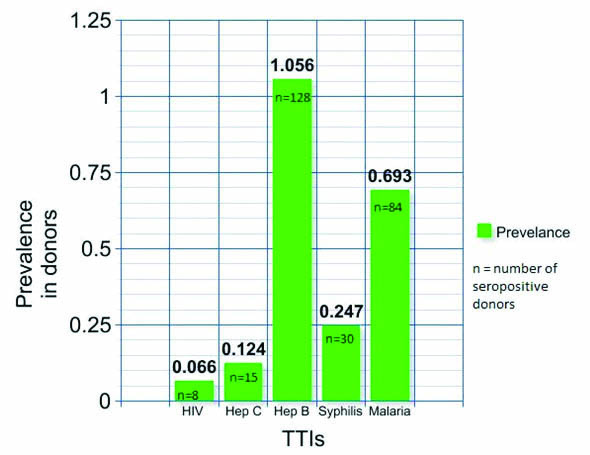
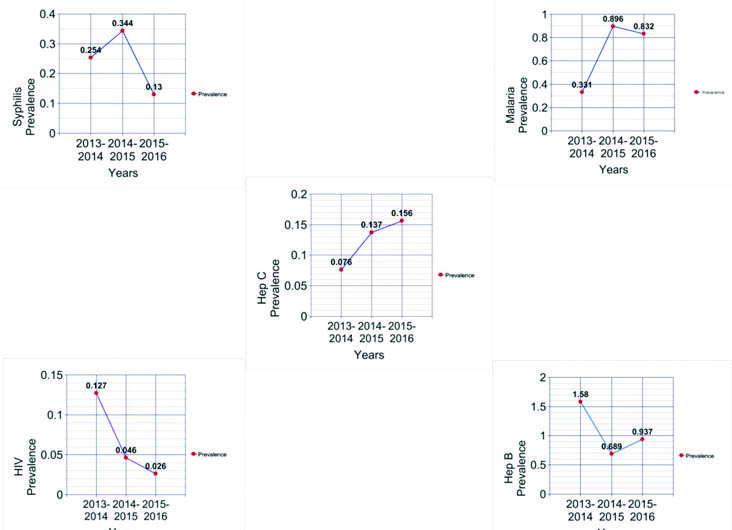
During the study period, a total of 12,118 donors were screened for TTIs. Majority of the screened donors belonged to 25-40 age group. Among them, 11756 (97.01%) were males and 362 (2.99%) were females. A total of 9409 (77.64%) were voluntary donors and 2709 (22.35%) were replacement donors. Out of 12118 screened donors, overall 265 (2.186%) were seropositive cases [Table/Fig-1]. Overall prevalence of Hepatitis B was found to be the highest followed by malaria, syphilis, hepatitis C and HIV respectively. In our study, the prevalence of TTIs showed a decreasing trend of HIV whereas an increasing trend of hepatitis C [Table/Fig-2].
Prevalence of HIV, Hep B, Hep C, syphilis and malaria among blood donors.
Trends of prevalence of TTIs among blood donors.
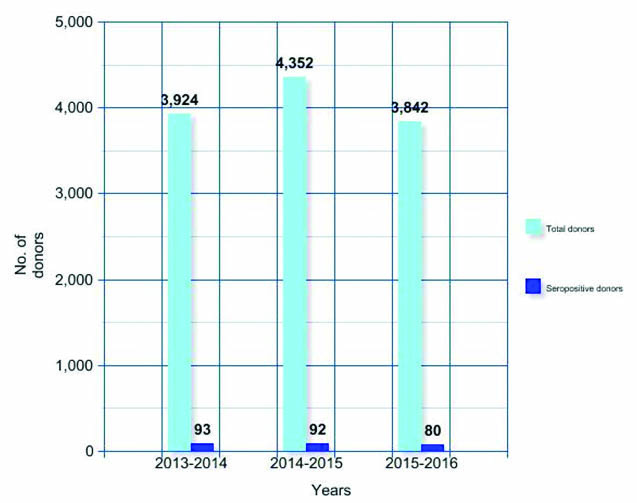
Total number of donors per year was 3924, 4352 and 3842 from April 2013 to March 2016 respectively. Yearly prevalence of seroreactivity was 2.370% (93), 2.113% (92) and 2.082% (80) for three consecutive years, from April 2013 to March 2016 [Table/Fig-3,4].
Yearly seroprevalence of TTIs.
| TTIs | 2013-2014 | 2013-2014 | 2014-2015 | 2014-2015 | 2015-2016 | 2015-2016 |
|---|
| No. Of positive | Preva-lence | No. Of positive | Preva-lence | No. of positive | Preva-lence |
| HIV positive | 5 | 0.127% | 2 | 0.046% | 1 | 0.026% |
| Anti-HCV positive | 3 | 0.076% | 6 | 0.137% | 6 | 0.156% |
| HBsAg positive | 62 | 1.580% | 30 | 0.689% | 36 | 0.937% |
| Syphilis positive | 10 | 0.254% | 15 | 0.344% | 5 | 0.130% |
| MP- Antigen positive | 13 | 0.331% | 39 | 0.896% | 32 | 0.832% |
| Total Seropositive donors | 93 | 2.370% | 92 | 2.113% | 80 | 2.082% |
Yearly distribution of total blood donors and total seropositive donors.
Discussion
Blood transfusion is an essential component of health care which potentially saves millions of lives every year. Every country needs to meet its requirements for blood and blood products and ensure that blood supplies are free from HIV, hepatitis viruses and other life threatening infections that can be transmitted through unsafe transfusion. WHO recommends that, at minimum, all blood for transfusion should be screened for HIV, hepatitis B, hepatitis C and Syphilis. Blood safety is integral to the WHO HIV/AIDS plan to combat the spread of HIV infection and to the achievement of the health-related Millennium Development Goals to reduce child mortality, improve maternal health, combat HIV and develop global partnership for development [3].
WHO promotes voluntary over replacement donation. In our study, 77.64% were voluntary and 22.35% were replacement donors. Patel et al., from western Ahmedabad reported voluntary blood donors as 95.56% [4], but a study from Karnataka and from Haryana reported it as 58% and 31.4% respectively [5,6]. People should be encouraged to become voluntary blood donors for which blood camps are conducted.
In our study, males (97.01%) outnumbered females (2.99%). This is similar to other studies done in India by Pallavi P et al., in which 97.84% were males [7], by Patel PA et al., in which 85% were males [4], by Arora D et al., in Southern Haryana [6], by Singh K et al., in Coastal Karnataka [8], by Pahuja et al., and Singh B et al., in Delhi [9,10], noting more than 90% of the male donors.
WHO obtained data through WHO Global Database on Blood Safety (GDBS) for the year 2013 which were reported by 156 countries. To give a more complete overview of the global situation, data for the year 2012 was used from 14 countries and data for the year 2011 from 9 countries, where current data were not available. Overall, responses received from 179 countries cover 98.6% of the world’s population. This fact sheet showed prevalence of HIV as 0.003% (0.001%-0.040%) in high-income countries, 0.020% (0.020%-0.340%) in middle-income countries and 1.080% (0.560%-2.690%) in low income countries [11]. In our study, the prevalence of HIV was 0.066%. The fact sheet showed prevalence of hepatitis B virus as 0.030% (0.008%-0.180%) in high-income countries, 0.910% (0.280%-2.460%) in middle-income countries and 3.700% (3.340%-8.470%) in low-income countries [11]. In our study, the prevalence was 1.056%. The fact sheet showed prevalence of hepatitis C virus as 0.020% (0.003%-0.160%) in high-income countries, 0.320% (0.090%-0.690%) in middle-income countries and 1.030% (0.670%-1.800%) in low-income countries [11]. In our study, the prevalence was 0.124%.
Mondal R et al., observed that cumulative seroprevalence of HIV, HBV, HCV and syphilis were 0.42%, 1.24%, 0.62% and 0.65% respectively; with solitary malaria-infected donor in a sub-Himalayan rural tertiary care centre in Darjeeling, India [12]. In our study, the prevalence of HIV, HBV, HCV and syphilis were 0.066%, 1.056%, 0.124% and 0.247%.
Nigam JS et al., conducted a study in Uttar Pradesh, India and observed that out of the 4128 screened donors, overall 93 (2.25%) were seroreactive cases [13]. In our study, seroreactive cases were 2.18%.
In a study conducted by Motayo BO et al., in Nigeria, high prevalence rates of 6.2%, 10% and 1.5% were observed for HIV, HBV and HCV respectively and there was 0% prevalence of Treponema pallidum antibodies [14]. The prevalence in our study of Andaman and Nicobar Islands differs from this study done in Nigeria.
In a study done by Bhattacharya H on Nicobarese tribe of Andaman and Nicobar islands, the prevalence of HBsAg reduced down to 7.4% in 2015 after vaccination in this tribe [15]. The prevalence of HBsAg in our study differs from this high prevalence in Nicobarese tribe because donor screening questionnaire is done for these donors; and the positive cases either don’t come for voluntary donation or they are not allowed to donate after knowing their hepatitis status. High endemicity of Hepatitis B virus (11.4%) was also observed in rural areas of Vietnam by Viet L et al., [16].
In a study done by Li C et al., in China on 43,66,283 donors, the prevalence of HIV, HBsAg, HCV and syphilis was 0.08%, 0.86%, 0.51% and 0.47%, respectively [17]. The results are comparable to our study [Table/Fig-5].
Comparison of sero-prevalence of present study with other studies [11-17].
| Study done by | Place of study | Prevalence of HIV | Prevalence of HbsAg | Prevalence of HCV | Prevalence of Syphilis | Prevalence of Malaria |
|---|
| Present study | Andaman & Nicobar | 0.066 | 1.056 | 0.124 | 0.247 | 0.693 |
| WHO global database on blood safety [11] | High income countries | 0.003 | 0.030 | 0.020 | - | - |
| Middle income countries | 0.020 | 0.910 | 0.320 | - | - |
| Low income countries | 1.080 | 3.700 | 1.030 | - | - |
| Mondal R [12] | Darjeeling, India | 0.42 | 1.24 | 0.62 | 0.65 | 0 |
| Nigam JS [13] | Uttar Pradesh, India | 0.05 | 0.97 | 0.63 | 0.46 | 0.15 |
| Motayo BO [14] | Nigeria | 6.2 | 10 | 1.5 | 0 | 0 |
| Bhattacharya [15] | Nicobar, India | Study not done on HIV | 7.4 | Study not done on HCV | Study not done on syphilis | Study not done on malaria |
| Viet L [16] | Vietnam | Study not done on HIV | 11.4 | 0.17 | Study not done on syphilis | Study not done on malaria |
| Li C [17] | China | 0.08 | 0.86 | 0.51 | 0.47 | - |
Nucleic acid Amplification Techniques (NAT) in western countries have reduced the risk of TTIs to a major extent [9]. Low cost-effectiveness of this technique makes implementation in economically restricted countries difficult.
Limitation
We recognize that our study had certain limitations which are worth mentioning. The ideal condition to conduct a prevalence study is to sample the general population. As blood donors are specifically selected based on extensive questionnaire and a physical examination, and apparently healthy individuals with lowest risk of TTIs are allowed to donate blood, our study does not represent general population. Further, the donor pool was predominantly composed of male population. With merely 2.99% of female donors, the prevalence cannot be generalized to female population of this region. Advanced methods like NAT are needed for detection of window period infections where antibodies are still undetectable but viral DNA can be picked up by NAT. These techniques are not available in our blood bank.
Conclusion
A higher prevalence of diseases which are transmissible through blood in developing countries causes the risk of TTIs more than that in developed countries. Lack of access to safe blood places unacceptable burden on the health sector of developing countries. This urges the use of more sensitive donor screening tests such as Nucleic acid testing even more desirable in the developing countries. There is a need for constant encouragement of voluntary blood donation, strict donor selection criteria and sensitive screening tests to minimize TTIs in Indian setting.