Comparative Evaluation of Antibacterial and Adhesive Properties of Chitosan Modified Glass Ionomer Cement and Conventional Glass Ionomer Cement: an In vitro Study
Arpan Debnath1, Srivastava Bagepalli Kesavappa2, Gyanendra Pratap Singh3, Shruthi Eshwar4, Vipin Jain5, Madhuniranjan Swamy6, Punith Shetty7
1 Postgraduate Student, Department of Public Health Dentistry, KLE Society’s Institute of Dental Sciences, Bengaluru, Karnataka, India.
2 Head, Department of Public Health Dentistry, KLE Society’s Institute of Dental Sciences, Bengaluru, Karnataka, India.
3 Postgraduate Student, Department of Conservative Dentistry, Krishnadevaraya Institute of Dental Sciences, Bengaluru, Karnataka, India.
4 Reader, Department of Public Health Dentistry, KLE Society’s Institute of Dental Sciences, Bengaluru, Karnataka, India.
5 Senior Lecturer, Department of Public Health Dentistry, KLE Society’s Institute of Dental Sciences, Bengaluru, Karnataka, India.
6 Senior Lecturer, Department of Public Health Dentistry, KLE Society’s Institute of Dental Sciences, Bengaluru, Karnataka, India.
7 Postgraduate Student, Department of Public Health Dentistry, Krishnadevaraya Institute of Dental Sciences, Bengaluru, Karnataka, India.
NAME, ADDRESS, E-MAIL ID OF THE CORRESPONDING AUTHOR: Dr. Arpan Debnath, 2nd Floor Site no 7, 4th Cross, 3rd Main, Basweswara Layout, Nagshettyhalli, P.O-Sanjaynagar, Bengaluru-560094, Karnataka, India.
E-mail: arpan.dnath@gmail.com
Introduction
Recent studies have reported the incorporation of modifiers with conventional Glass Ionomer Cement (GIC) to enhance its antibacterial effect. One such modification being the addition of Chitosan (CH). However, these modifiers might affect the physical properties of the restorations.
Aim
The aim of the present study was to investigate the effect of modifying the liquid phase of conventional GIC with 10% v/v CH on the antibacterial properties and adhesion to enamel in comparison to conventional GIC.
Materials and Methods
The liquid of commercially available restorative GIC (Fuji IX, GC Industrial Corporation, Japan) was modified with 10% v/v CH solution (Everest Biotech, Bengaluru). GIC powders were mixed with the unmodified and the CH-modified liquids at the desired powder/liquid ratio. Fourier Transform Infrared Spectroscopy (FTIR) was used to determine the setting reaction between the conventional and CH modified GIC liquid with GIC powder. For evaluation of the antibacterial properties, biofilms of Streptococcus mutans (S. mutans) were formed on the GIC discs and characterized by Scanning Electron Microscope (SEM). For the characterization of the adhesive properties, the unmodified and CH-modified GICs were bonded to the enamel surface and the microshear bond strength (μSBs) was evaluated. Student’s paired t-test was used to compare the micro-shear bond strength between CH modified GIC and unmodified GIC.
Results
Modification with 10% v/v CH solution improved the antibacterial properties of GIC against S. mutans in terms of resistance to biofilm formation which was assessed using SEM. Microshear bond strength of CH modified GIC was 85.40 MPa compared to 46.94 MPa of conventional GIC. This difference was found to be statistically significant.
Conclusion
Modifying the liquid phase of a conventional GIC with 10% v/v CH significantly improves the antibacterial property of GIC as well as its adhesion to enamel.
Antibacterial properties, Biocompatible, Streptococcus mutans
Introduction
Ever since the introduction of GIC in 1972 by Wilson and Kent, it’s been used as excellent dental restorative materials, having many benefits like chemical adhesion to enamel and dentin with antibacterial properties due to release of fluoride [1]. Because of biocompatible nature, GICs have become the restorative material of choice in minimal intervention approaches, such as Atraumatic Restorative Treatment (ART) [2].
Conventional GICs have been formulated specifically for the ART, however, it was seen that the antibacterial effect of conventional GICs is not efficient in affecting biofilm formation and cell viability [3,4]. To enhance its antibacterial action, chlorhexidine was added to conventional GIC, which in turn affected its mechanical and physical properties [5,6].
Thus, there was a need for an alternative biocompatible additive which has a potential for enhancing the antibacterial effect of GIC without compromising its physical properties.
CH is a natural linear bio-polyaminosaccharide formed by alkaline deacetylation of chitin, which occurs naturally in the shells of crabs and shrimps and it is considered to be one of the most widely distributed biopolymers [7,8]. It is a weak base and is insoluble in water and organic solvents; however, it is soluble in dilute aqueous acidic solutions such as acetic acid [9].
In addition to its unique biological characteristics, such as biocompatibility, mucoadhesion, it has got a wide spectrum of antibacterial and antibiofilm properties against Gram-positive and Gram-negative bacteria [10,11].
Earlier studies have shown improved mechanical properties of modified GIC when 10% v/v CH was added to the liquid phase [12]. However, bond strength to enamel has never been assessed, although bonding to enamel is a more reliable factor in case of GIC due to its chemical nature [13]. Even assessment of the antibacterial nature of CH modified GIC under controlled conditions is still unknown.
Thus, the aim of this study was to investigate the effect of modifying the liquid phase of conventional glass ionomer restorative material with 10% v/v CH on the antibacterial properties and adhesion to enamel in comparison to conventional GIC.
Materials and Methods
A randomized concurrent parallel experimental design was undertaken to evaluate the antibacterial and microshear properties of CH modified GIC and conventional GIC, which was conducted in KLE Institute of Dental Sciences, Bengaluru, Karnataka, India, for a period of one month (from 17.08.2015 to 18.09.2015). Sample size calculation was done using G power software (3.1.9.2 version) with level of significance set at 5% and power of the test at 80%. Eight GIC discs each for both modified and unmodified GICs was used to assess the antibacterial properties through SEM. Forty recently extracted sound human molars within a week of start of study was used after approval from Institutional Review Board for the assessment of microshear bond strength. All the teeth were vital at the time of surgical removal and were extracted for periodontal reasons (clinically Grade III mobility). Teeth with caries, restorations, cracks, fractures or other structural defects were excluded from the study. Ethical approval for the study was granted by the Ethical and Research Committee of the Institution.
Modification of Glass Ionomer with Chitosan
Almost 1.8 ml of glacial acetic acid was made up to 100 ml with distilled water in a 100 ml standard flask to get 0.3 N acetic acid. About 20 mg of CH (Everest Biotech, Bengaluru) was weighed separately and dissolved in 0.3 N acetic acid and made up to 100 ml with the same acetic acid in a 100 ml standard flask to get 0.2 mg / ml Chitosan solution. Then, 0.1 ml of 0.2 mg/ml of CH solution was added to 0.9 ml of GIC liquid (Fuji IX, GC Industrial Corporation, Japan) to get 10% v/v CH modified glass ionomer solution [14].
Fourier Transform Infrared Spectrometry Analysis (FTIR)
FTIR is a technique which is used to obtain an infrared spectrum of absorption or emission of a solid, liquid or gas. An FTIR spectrometer simultaneously collects high spectral resolution data over a wide spectral range. This confers a significant advantage over a dispersive spectrometer which measures intensity over a narrow range of wavelengths at a time [15].
FTIR analysis was carried out to determine the setting reaction between the conventional and CH modified GIC liquid with GIC powder (Fuji IX, GC Industrial Corporation, Japan). The GIC powder was mixed with the conventional and Chitosan modified GIC liquid at specified powder/liquid ratio of 3.6 g/1.0 g and allowed to set. The set cement was incubated at 37°C and 100% humidity for 24 hours. Afterwards, the set cement was crushed into powder and FTIR analysis was carried out [14].
Assessment of Antimicrobial Property
Specimen preparation and formation of biofilm: Eight unmodified and modified GIC discs each, measuring 7 mm diameter and 2 mm thickness were prepared using split-teflon mold and the specimen was stored at 37°C for 24 hours and 100% humidity before sterilization. The GIC discs were then sterilized using ethylene oxide gas. For formation of the biofilm, S. mutans bacteria (MTCC no. 497 strain) were used. The bacteria were cultured in 3 ml of Brain Heart Infusion (BHI) broth (Oxoid Microbiology product, Bengaluru) and were incubated overnight at 37°C in 5% CO2 incubator. The GIC discs were then placed in the well plate, inoculated with 1 ml of the inoculation medium for each well. These were then incubated at 37°C in 5% CO2 for three days to allow biofilm to form. After three days the discs were prepared and viewed using SEM (Team EDS Analysis System, USA) at x3000 magnification.
Assessment of Microshear Bond Strength
Sample collection: Forty freshly extracted human molars were used for this study. Immediately after extraction, the teeth were rinsed under running water and stored in specimen bottles filled with purified filtered water. These teeth were randomly divided into two groups (n=20).
Specimen preparation: Samples were prepared for microshear bond strength analysis by removing the occlusal enamel perpendicular to the long axis of the tooth with microtome under water lubrication. A 600 grit size silicon carbide paper was used for finishing the surface. The enamel surface was treated with conditioner for 10 seconds using cotton pellet, then rinsed thoroughly with water and gently dried without desiccation. The GIC powder (Fuji IX, GC Industrial Corporation, Japan) was mixed with the conventional and CH modified GIC liquid at specified powder/liquid ratio of 3.6g/1.0g. Prefabricated Poly Tetra Fluoro Ethylene (PTFE) molds were placed over the enamel surface and both modified and unmodified GICs were condensed in the hollow PTFE molds (1 mm internal diameter) [Table/Fig-1]. After initial setting of 15 minutes, the restored teeth were incubated at 37°C and 100% humidity for 24 hour. Each specimen with acrylic mold was attached to the jig of universal testing machine (Dong Guan Precision Instrument Co. Ltd) [Table/Fig-2]. Shear force at cross head speed of 1 mm/min was applied at the enamel-cement junction until fracture occurred and microshear bond strength was calculated at that point [12].
Prefabricated PTFE mold with GIC condensed into hollow mold.

Universal testing machine and specimen.

Statistical Analysis
All the numerical data were expressed as means and standard deviations. Analysis was carried out using SPSS (IBM SPSS Statistics Version 24.0). Paired t-tests were done to compare the microshear bond strength between the two groups. The p-values less than 0.05 were considered statistically significant. Antibacterial properties of the two materials were assessed using relative density of the microbiological colonies formed on the surface by SEM.
Results
FTIR Analysis
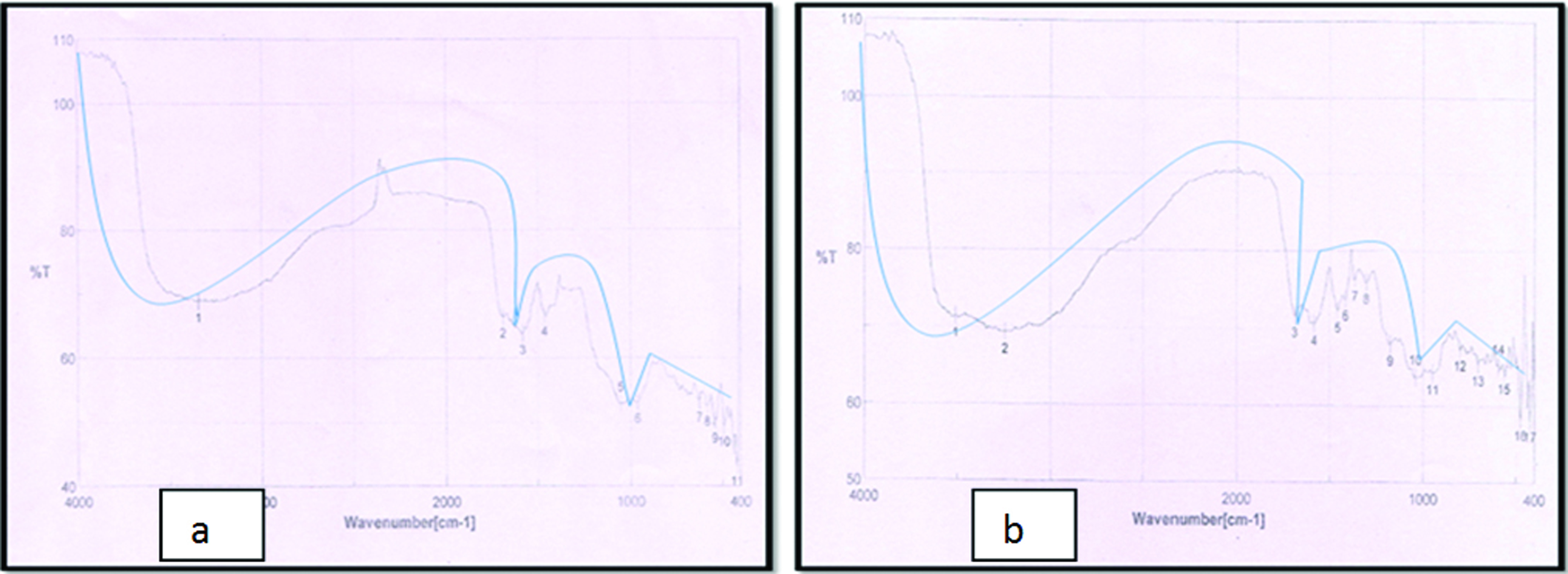
FTIR analysis of conventional GIC [Table/Fig-3a] showed the predominant functional groups were hydroxyl (OH) and carboxyl (C=O) groups and in case of CH modified GIC [Table/Fig-3b], major reaction took place between the primary amino (NH2) group of CH and the hydroxyl (OH) and carboxyl (C=O) groups of GIC at the wavelength range of 3200-3600 cm-.1 and 1670-1820 cm-.1 respectively.
Infrared spectrum of: a) GIC matrix; b) Chitosan modified GIC matrix.
Characterization of Antibacterial Properties
The SEM images of three days old biofilm on unmodified and CH modified GIC spherical discs are shown in [Table/Fig-4]. At x3000 magnification, conventional GIC discs show a thick, uniform and dense bacterial coverage [Table/Fig-4a], whereas, in case of 10% CH modified GIC, sparse collection of coccal bacterial forms can be seen with very minimal surface cracks [Table/Fig-4b].
SEM images of biofilm formation on conventional GIC and chitosan modified GIC at X3,000 magnification: a) The unmodified GIC specimens showed thick, uniform, dense and heavy biofilm coverage where the underlying GIC surface is hardly seen; b) Thinner and less dense of the biofilm coverage was seen 10 v/v% chitosan modified GIC.

Characterization of Adhesion to Enamel
Modification of GIC liquid with 10% v/v CH solution significantly increased the mean microshear bond strength of CH modified GIC (85.40 Mpa) compared to conventional GIC (46.94 Mpa) with standard deviation of 3.6 and 4 respectively [Table/Fig-5]. The difference between two groups was found to be statistically significant.
Shows the mean microshear bond strength of two groups of chitosan modified GIC and conventional GIC.
| GROUPS | MEAN+SD (MPa) | p-value |
|---|
| Chitosan GIC | 85.40+3.6 | <0.05 |
| Conventional GIC | 46.94 + 4.0 |
Paired t-test used
Statistical significance <0.05
Discussion
GICs are used in dentistry for restorative and preventive applications because of its unique properties. These applications motivated various modifications in conventional GICs so as to enhance the antibacterial and/or physical properties without adversely affecting the chemical adhesion to enamel and dentin [16,17].
CH has been used as a natural biocompatible linear biopolyaminosaccharide which has been proven that it has a potent antibacterial effect against oral biofilms, specifically S. mutans, thus, paving the way for its use as a preventive and therapeutic agent to control dental caries [18,19]. CH in the present study was used with acetic acid to modify the GIC liquid and the pH values of the modified liquids were kept in the acidic range (approximately 1). CH solubility in acidic environment is explicated by the protonation of the free amino groups (NH2) to NH3+ [12]. This is because CH can be considered a strong base as it possesses primary amino groups with a pKa value of 6.3. The presence of the amino groups indicates that pH substantially alters the charged state and properties of CS. At low pH these amines get protonated and become positively charged and that makes CS a water soluble cationic polyelectrolyte [20]. An infrared spectroscopic study was carried out to determine the setting reaction between the CH modified and conventional GIC. The study revealed that the reaction had taken place between amino (-NH2) group of CH and the functional group (OH group and C=O group) of GIC. The final matrix showed no additional peaks or downs representing that the CH was thoroughly mixed in the GIC matrix. Since CH possess hydroxyl and acetamide groups, they bind to hydroxyl group of powder particles and carboxylic groups of polyacrylic acid by hydrogen bonding. This interaction reduces interfacial tension among GIC components, thereby improving mechanical performance. But with increasing Chitosan concentration, CH molecules segregate and interact with one another rather than with GIC components resulting in poor mechanical performance [21,22].
The first aim of the study was to investigate the effect of modification of GIC liquid with CH on the bonding of GIC to enamel. It is important that any modification of GIC should not affect the bonding ability to enamel and/dentin. Previous studies have shown no difference in microshear bond strength between CH modified GIC and conventional GIC with respect to dentin. Both adhesive bonding failures and partial cohesive failures were seen between dentin and GIC [12]. But the bonding of GIC to enamel forms a major reliable factor for establishing the bond strength of the material [23]. This bonding of GIC to enamel was never assessed in previous studies. In the present study, microshear bond strength was assessed between the GIC and the enamel. The mean microshear bond strength of CH modified GIC is 84% more than conventional GIC [Table/Fig-5], which is in contrast to the result which assessed the adhesion of CH modified GIC to dentin [12]. This can be explained due to the polar and ionic attraction between the carboxylic groups of polyacrylic acid and hard tooth surfaces. Since CH is a high chelating agent, more polyacrylic acid chains would diffuse into the enamel to displace the phosphate and calcium ions from hydroxyapatite crystals.
The second aim was to investigate the affect of 10% v/v CH modified GIC on the S. mutans (MTCC no 497). The SEM images showed thick biofilm formation in conventional GIC but a significant reduction was seen in the SEM images of CH modified GIC which could be attributed to the CH’s bactericidal and/or bacteriostatic effect. Although, the antimicrobial action mechanism is not fully understood, CH acts on the cellular wall of microorganism by modifying the electric potential of the cellular membrane. It was claimed that the protonated amino groups of chitosan could bind to anionic groups of microorganisms, resulting in agglutination of bacterial cells [24]. The probable mechanisms which might explain the antibacterial activity of CH-GIC are CH may promote displacement of Ca2+ of the anionic sites of the membrane resulting in cell damages and interaction between positive load of CH and negative load of microbial cell wall, which might lead to rupture and loss of important intracellular constituent. Further, chitosan with low molecular weight and viscosity could possibly penetrate inside the bacterial cell to be linked to bacterial DNA thereby, inhibiting transcription and consequently translation [12].
Study conducted by Petri D et al., [21] showed that the amount of fluoride released from CH modified GIC was much higher than the normal GICs and the antibacterial properties was also higher in the CH modified GIC according to the study conducted by Ibrahim M et al., which was in accordance to the present study [12]. This was because amalgamation of acidic solutions of CH in the polyacrylic acid liquid of GIC at v/v ratio of 10% enhanced the antibacterial properties of conventional GIC against S. mutans and also expressively increased the bond strength of GIC to enamel [12].
Limitation
In the present study for how long the antibacterial effect against S. mutans will last with aging and maturation of modified GIC was not assessed and also the antibacterial effect on other biofilm models involved in dental caries and gum diseases was not assessed.
Conclusion
Based on the results of the current study and other related studies modification of GIC with CH could be of potential clinical significance in preventive dentistry owing to its proven antibacterial and mechanical reinforcement effects and its ability to release potential bioactive molecules and growth factors. More in vivo clinical trials will have to be conducted involving CH modified GIC for it to have a prominent role in the field of preventive dentistry in the coming years.
[1]. Almuhaiza M, Glass ionomer cement in restorative dentistry: a critical appraisalJ Contemp Dent Pract 2016 17(4):331-36. [Google Scholar]
[2]. Yip H, Smales R, Ngo H, Selection of restorative materials for the atraumatic restorative treatment (ART) approach: a reviewSpec Care Dentist 2001 21(6):216-21. [Google Scholar]
[3]. Eick S, Glockmann E, Brandl B, Pfister W, Adherence of Streptococcus mutans to various restorative materials in continuous flow systemJ Oral Rehabil 2004 31:278-85. [Google Scholar]
[4]. Forss H, Näse L, Seppä L, Fluoride concentration, mutans streptococci and lactobacilli in plaque from old glass ionomer fillingsCaries Res 1995 29(1):50-53. [Google Scholar]
[5]. Botelho MG, Inhibitory effects on selected oral bacteria of antibacterial agents incorporated in a glass ionomer cementCaries Res 2003 37:108-14. [Google Scholar]
[6]. Palmer G, Jones FH, Billington RW, Pearson G.J, Chlorohexidine release from an experimental glass ionomer cementBiomaterials 2004 25:5423-31. [Google Scholar]
[7]. Muzzarelli RAA, Chitin 1977 New YorkPergamon Press [Google Scholar]
[8]. Roberts GAF, Chitin chemistry 1992 HoundmillsMacMillan Press [Google Scholar]
[9]. Chandy T, Sharma CP, Chitosan–as a biomaterialBiomater Artif Cells Artif Organs 1990 18:01-24. [Google Scholar]
[10]. Hu Y, Du Y, Yang J, Tang Y, Li J, Wang X, Self aggregation and antibacterial activity of N-acylated chitosanPolymer 2007 48(11):3098-106. [Google Scholar]
[11]. Xia W, Liu P, Zhang J, Chen J, Biologic activities of chitosan and chitooligosaccharidesFood Hydrocolloid 2011 25(2):170-79. [Google Scholar]
[12]. Ibrahim M, Neo J, Esguerra RJ, Fawzy AS, Characterization of antibacterial and adhesion properties of chitosan modified glass ionomer cementJ of Biomater Appl 2015 30(4):409-19. [Google Scholar]
[13]. Craig RG, Powers JM, Wahtaha JC, Dental materials: Properties and manipulation 2004 8th edSt. LouisMosby [Google Scholar]
[14]. Karthick A, Kavitha M, Evaluation of microshear bond strength of chitosan modified GICWorld J of Med Sci 2014 10(2):169-73. [Google Scholar]
[15]. Griffiths P, Haseth J, Fourier transform infrared spectrometry 2007 2nd edJohn Willey and Sons Publication [Google Scholar]
[16]. Cepowicz E, Kolada G, Antibacterial activity of selected glass ionomer cementsPostepy Hig Med Dosw 2014 68:23-28. [Google Scholar]
[17]. Wakeel A, Elkassas D, Bonding of contemporary glass ionomer cements to different tooth substrates; microshear bond strength and scanning electron microscope studyEur J Dent 2015 9(2):176-82. [Google Scholar]
[18]. Hayashi Y, Ohara N, Ganno T, Chewing chitosan containing gum effectively inhibits the growth of cariogenic bacteriaArch Oral Biol 2007 52:290-94. [Google Scholar]
[19]. Hayashi Y, Ohara N, Ganno T, Chitosan-containing gum chewing accelerates antibacterial effect with an increase in salivary secretionJ Dentistry 2007 35:871-74. [Google Scholar]
[20]. Pillai C, Paul W, Chitin and chitosan polymers: Chemistry, solubility and fiber formationProgress in Polymer Science 2009 April 34:641-78. [Google Scholar]
[21]. Petri D, Donega J, Benassi AM, Bocangel JA, Preliminary study on chitosan modified glass ionomer restorativesJ of Dent Mater 2007 23(8):1004-10. [Google Scholar]
[22]. Sailaja GS, Velayudhan S, Sunny MC, Sreenivasan N, Varma H.K, Ramesh P, Hydroxyapatite filled chitosan polyacrylic acid polyelectrolyte complexesJ Mater Sci 2003 38(17):3653-62. [Google Scholar]
[23]. Cortes O, Godoy F, Boj JR, Bond strength of resin-reinforced glass ionomer cements after enamel etchingAm J Dent 1993 6:299-301. [Google Scholar]
[24]. Carvalho M, Stamford T, Chitosan as an oral antimicrobial agentCommunicating Current Research and Technological Advances 2011 :542-44. [Google Scholar]