A Rare Case Report of Spindle Cell Ameloblastic Carcinoma Involving the Mandible
Mohammad Asif Kiresur1, Arunodaya Kunche2, Anuradha Ananthaneni3, Bhavana S Bagalad4, Puneeth Horatti Kuberappa5
1 Senior Lecturer, Department of Oral and Maxillofacial Pathology, St. Joseph Dental College, Eluru, Andhra Pradesh, India.
2 Postgraduate Student, Department of Oral and Maxillofacial Pathology, St. Joseph Dental College, Eluru, Andhra Pradesh, India.
3 Professor, Department of Oral and Maxillofacial Pathology, St. Joseph Dental College, Eluru, Andhra Pradesh, India.
4 Senior Lecturer, Department of Oral and Maxillofacial Pathology, St. Joseph Dental College, Eluru, Andhra Pradesh, India.
5 Senior Lecturer, Department of Oral and Maxillofacial Pathology, St. Joseph Dental College, Eluru, Andhra Pradesh, India.
NAME, ADDRESS, E-MAIL ID OF THE CORRESPONDING AUTHOR: Dr. Mohammad Asif Kiresur, Senior Lecturer, Department of Oral and Maxillofacial Pathology, St. Joseph Dental College, Eluru-534004, Andhra Pradesh, India.
E-mail: mdasifkiresur7@gmail.com
Ameloblastic Carcinoma (AC) is uncommon malignant epithelial odontogenic tumour of jaw, with characteristic histologic features and behavior. Clinically, it has aggressive, infiltrative growth pattern with a distinct predilection for mandible. It exhibits histologic features of ameloblastoma and gets dedifferentiated overtime to culminate in carcinoma. Majority of the cases arise denovo (primary) and only few cases arise from a pre-existing ameloblastoma (secondary). Spindle-cell differentiation in ameloblastic carcinoma is rare; Salter described it as a separate entity “low-grade spindle cell ameloblastic carcinoma. Here we report a case of 32-year-old female patient who presented with a swelling present for past six months. It was diagnosed as Spindle cell Ameloblastic Carcinoma (SpAC), after the hemimandibulectomy the patient was under regular follow up for 14 months, no sign of recurrence was seen.
Ameloblastoma, Odontogenic tumour, Spindle cell differentiation
Case Report
A 32-year-old female reported to the outpatient department of St. Joseph Dental College, Eluru, Andhra Pradesh, India, with a chief complaint of swelling on the right side of face present since six months [Table/Fig-1]. The swelling was associated with pain, difficulty in mastication and mouth opening. Patient presented with a history of similar swelling with mobility of teeth in right lower back region two years back for which she was surgically treated. Previous biopsy report confirmed the presence of ameloblastoma.
Diffuse swelling in the middle third of the face.
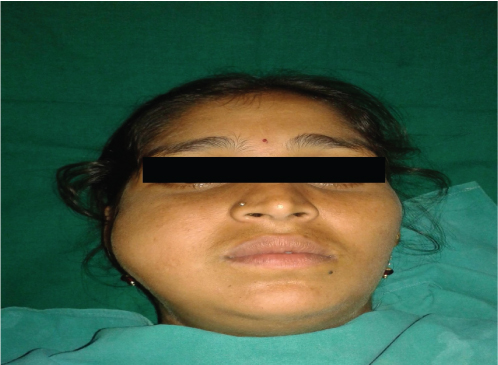
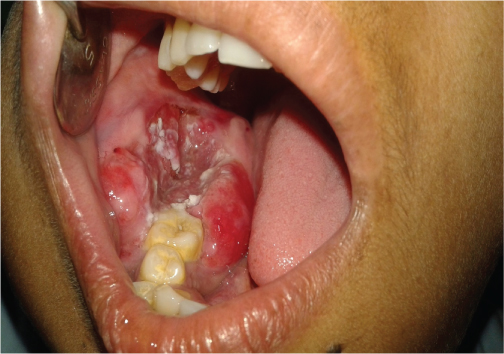
Clinical examination showed diffuse swelling in the middle third of the face which resulted in gross facial asymmetry. On palpation the swelling was smooth, non tender and firm in consistency. Right submandibular lymph nodes were palpable and non-tender. Intraoral examination showed a diffuse swelling with bicortical expansion extending from the distal side of first molar to the retromolar area obliterating both right buccal and lingual vestibule. The lesion was erythaematous and ulcerated [Table/Fig-2].
Erythaematous and ulcerated intraoral swelling.
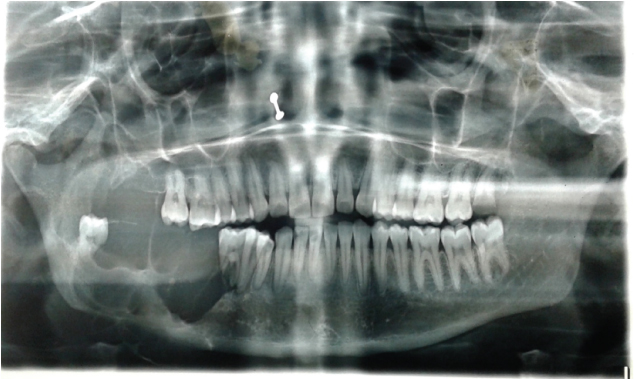
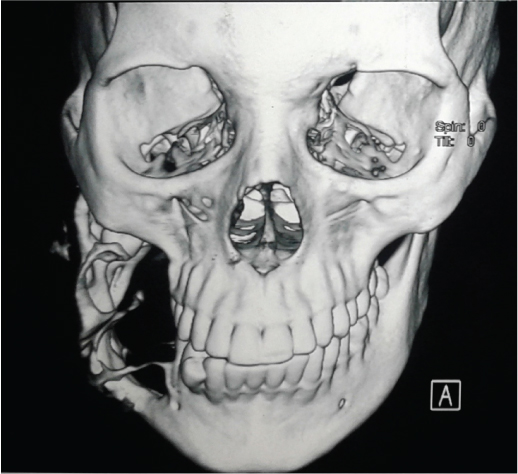
Orthopantomogram (OPG) showed presence of multilocular radiolucency extending from distal aspect of mandibular first molar to ramus of the mandible with displacement of tooth [Table/Fig-3]. The Computed Tomography (CT) scan showed a lobulated, expansile lytic lesion involving body, ramus, and coronoid process of mandible measuring 6x8x7.8 cm in size, with perforation of the cortical plates [Table/Fig-4]. Based on clinical and radiographic features, the lesion was provisionally diagnosed as ameloblastoma (recurrent) incisional biopsy from the lesion was microscopically examined and diagnosed as ameloblastoma. Right hemimandibulectomy was performed.
OPG with presence of multilocular radiolucency extending from distal aspect of mandibular first molar to ramus of the mandible on the right side.
CT scan showing a lobulated expansile lytic lesion involving body, ramus, and coronoid process of mandible.
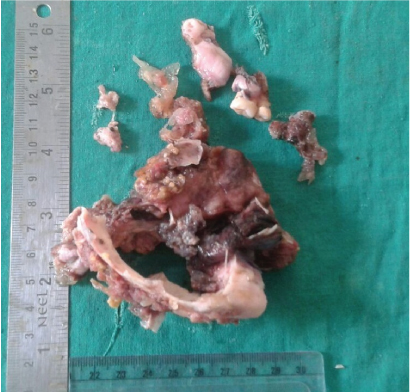
The excised lesion was sent for histopathological analysis. On gross examination, the lingual cortical plate was eroded; the tissue within the mandible appeared to be grayish black with irregular surface and firm in consistency [Table/Fig-5].
Resected part of the mandible showing perforation of cortical bone.
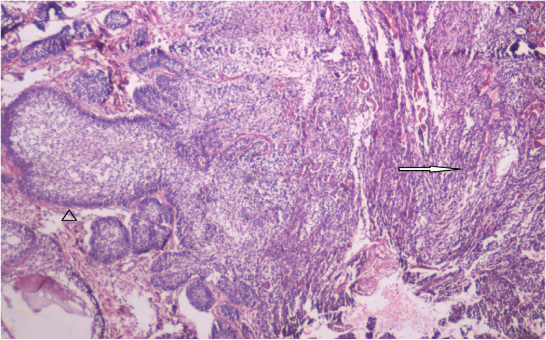
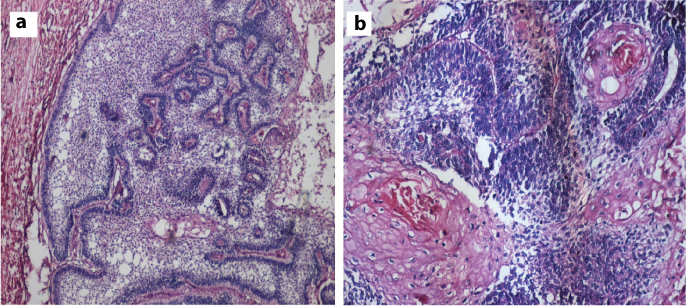
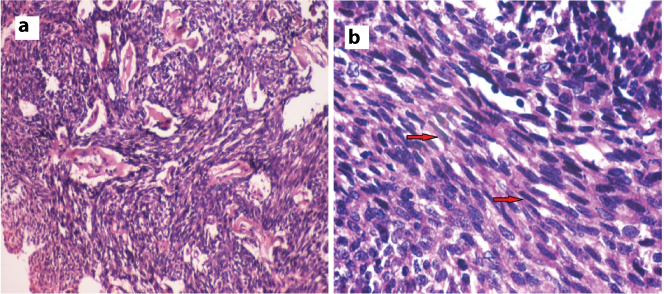
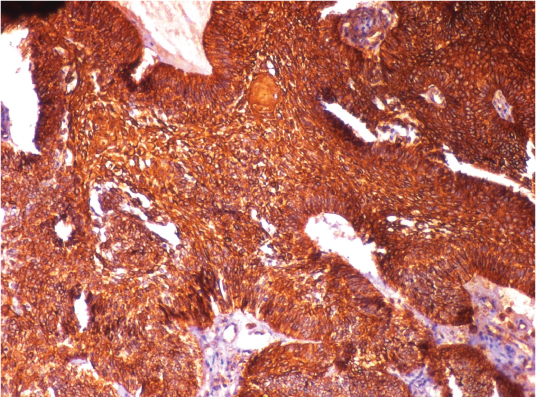
Microscopic examination showed large hypercellular epithelial islands with few cells displaying peripheral tall columnar cells with reverse polarity. Stellate reticulum like cells showed squamous differentiation and keratin pearl formation [Table/Fig-6,7]. Cellular atypia, nuclear hyperchromatism and increased mitosis were observed. Sheets of spindle cells were also observed [Table/Fig-8]. Immunohistochemical (IHC) staining with the cytokeratin-19 showed positivity in the epithelial islands and atypical spindle shaped cells [Table/Fig-9]. Based on the histopathological and IHC findings it was diagnosed as spindle cell variant of AC.
Haematoxylin and eosin stained section shows ameloblastic islands (arrow head) and area of spindle cell differentiation (arrow) (4X magnification).
a) Hypercellular epithelial islands enclosing stellate reticulum (haematoxylin and eosin stain,10X magnification); b) Basal cell hyperplasia wiith keratin pearl formation in stellate reticulum like area (haematoxylin and eosin stain, 40X magnification).
a) Sheets of spindle cells with cellular atypia and nuclear hyperchromatism; b) Atypical spindle shaped cells (arrow).
Immunohistochemical staining positive with cytokeratin-19 for ameloblastic islands and spindle shaped cells.
Discussion
AC is a rare odontogenic tumour that shows the histological features of ameloblastoma with cellular atypia in the absence of metastasis [1]. According to Elzay RP and Slootweg PJ the term AC can be used to label a lesion that shows histologic features of both ameloblastoma and carcinoma [2,3]. AC can arise from odontogenic cysts, ameloblastoma, odontogenic epithelial rests, salivary gland epithelium and epithelium entrapped along embryonic fusion sites [4–6]. AC demonstrate two different variants depending on the differentiation: 1) De-differentiated (secondary) variant characterized by lesions that initially demonstrate the morphology of an ameloblastoma, but which de-differentiate in the course of time. De-differentiation can be spontaneous or related with surgical procedures or radiaotherapy: 2) Less-differentiated (primary) variant is comprised of histologic features of an ameloblastoma associated with less differentiated areas. As the patient was surgically treated for ameloblastoma two years back this case can be considered as de-differentiated or secondary in origin [7].
AC is commonly seen in the posterior part of the mandible, the unusual locations reported are maxilla, nasal cavity and anterior skull. The age of occurrence of SpAC is third to seventh decade [Table/Fig-10] [3,7–16]. Cases reported by Lau SK et al., and Ismail SB et al., were seen in 23 and 21-year-old individuals respectively [9,11]. In the present case a 32-years-old female presented with swelling of the right mandible associated with pain, difficulty in mastication and mouth opening, the cervical lymph nodes were palpable and non tender.
| Author | Age/Gender | Location | Treatment | Recurrence/Follow up |
|---|
| Slootweg PJ et al., [3] | 75/Male | Mandible | Chemotherapy and radiation | Dead, one year after treatment |
| Nagai N et al., [7] | 50/Male | Mandible | Surgery | NA |
| Infante-Cossio P et al., [8] | 69/Female | Maxilla | Surgery and radiation | Alive, five years after surgery |
| Lau SK et al., [9] | 23/Male | Mandible | Surgery | Alive, five years after surgery |
| Kawauchi S et al., [10] | 67/Male | Mandible | Surgery | Dead, one year and 10 months after surgery |
| Ismail SB et al., [11] | 21/Female | Mandible | Surgery with resection of lymph node | Absent |
| Jindal C et al., [12] | 60/Female | Mandible | Surgery | Absent |
| Kamath VV et al., [13] | 75/Male | Mandible | Surgery | Patient died two weeks after surgery |
| Ansari HA et al., [14] | 60/Male | Maxilla | Surgery followed by radiotherapy | NA |
| Mc Naught MJ et al., [15] | 53/Male | Mandible | Surgery | Absent/Alive |
| Matsushita Y et al., [16] | 69/Male | Mandible | Surgery followed by radiotherapy | Absent/Alive 23 months of follow up |
| Present report | 35/Female | Mandible | Surgery | Absent/Alive 14 months follow up |
AC shows multilocular radiolucency with ill-defined borders and perforation of cortical bone, invasion into the soft tissue, a feature that is not commonly seen in an ameloblastoma. In most cases, radiographs show ill-defined radiolucency, focal areas of radiopacity can be seen in radiolucent area [1]. In the present case OPG showed multilocular radiolucency involving right side of the mandible extending upto the retro molar area. The second molar was missing, possibly it was extracted during the treatment of ameloblastoma.
Histologically, ACs have characters of both a benign ameloblastoma and carcinoma. The growth pattern can be follicular or plexiform, or combination of both. The peripheral tall columnar cells are arranged in a palisaded pattern, reversal of nuclear polarity and hyperchromatism may be evident. Basaloid cells with predominant nuclear hyperchromatism may dominate in the center most areas of the tumour islands, loss of ameloblastic differentiation, spindling, more than two mitotic figures per high-power field can be seen [1]. The cases reported by Jindal C et al., and Kamath VV et al., showed spindle cell associated with cystic epithelium [12,13].
Ansari HA et al., [14] reported a case of SpAC with presence of nests of tumour cells arranged in cribriform pattern with luminal eosinophilic hyaline-like material, which resembled adenoid cystic carcinoma.
In the present case, histopathologic examination showed few characteristic ameloblastic follicles. Stellate reticulum like area showed squamous differentiation and keratin pearl formation.
Numerous areas of necrosis were present, pseudosarcomatous areas were predominated by sheets of spindle cells. The spindle cells showed nuclear hyperchromatism and pleomorphism, scattered mitotic figures, two to five mitotic figures were present in the high power field. IHC stain showed positivity for cytokeratin 19 in the epithelial islands and in the spindle shaped cell area; positive reaction with cytokeratin 19 confirmed the epithelial origin of the spindle cells. Similar IHC findings were seen in cases reported by Kawauchi S et al., [10] and Ismail SB et al., [11]. Thus, it was diagnosed as SpAC.
Resection with complete removal of the tumour is the treatment of choice. Radiotherapy with regular follow up is advised when the resection is impossible. Prognosis is very poor and metastasis can occur to lungs, few years after the treatment [1,3]. It should be treated aggressively as early as possible with regular follow up. In the present case right hemi-mandibulectomy was done with complete excision of lesion and the patient was followed for 14 months and periodic follow up was advised.
Conclusion
SpAC is a rare odontogenic epithelial neoplasm of the jaw that shows combination of histological features of an ameloblastoma with features of cytological atypia, with a poor prognosis. Due to lack of literature, the biological behavior, recurrence and metastatatic nature of this lesion is not known. This case report described a SpAC of mandible that originated from a pre-existing ameloblastoma with prompt treatment and regular follow up.
[1]. Barnes L, Eveson JW, Reichart P, Sidransky D, World Health Organization Classification of TumoursPathology and Genetics of Head and Neck Tumours 2005 LyonIARC Press:287-93.Odontogenic carcinomas [Google Scholar]
[2]. Elzay RP, Primary intraosseous carcinoma of the jaws. Review and update of odontogenic carcinomasOral Surg Oral Med Oral Pathol 1982 54:299-303. [Google Scholar]
[3]. Slootweg PJ, Muller H, Malignant ameloblastoma or ameloblastic carcinomaOral Surg Oral Med Oral Pathol 1984 57:168-76. [Google Scholar]
[4]. Corio RL, Goldblatt LI, Edwards PA, Hartman KS, Ameloblastic carcinoma: A clinicopathologic study and assessment of eight casesOral Surg Oral Med Oral Pathol 1987 64:570-76. [Google Scholar]
[5]. Yoon HJ, Hong SP, Lee JI, Lee SS, Hong SD, Ameloblastic Carcinoma: An analysis of 6 cases with review of the literatureOral Surg Oral Med Oral Pathol Oral Radiol Endod 2009 108:904-13. [Google Scholar]
[6]. Yoshioka Y, Toratani S, Ogawa I, Okamoto T, Ameloblastic carcinoma, secondary type, of the mandible: A case reportJ Oral Maxillofac Surg 2013 71:58-62. [Google Scholar]
[7]. Nagai N, Takeshita N, Nagatsuka H, Inoue M, Nishijima K, Nojima T, Ameloblastic carcinoma: Case report and reviewJ Oral Pathol Med 1991 20:460-63. [Google Scholar]
[8]. Infante-Cossio P, Hernandez-Guisado JM, Fernandez-Machin P, Garcia-Perla A, Rollon-Mayordomo A, Gutierrez-Perez JL, Ameloblastic carcinoma of the maxilla: A report of 3 casesJ Craniomaxillofac Surg 1998 26:159-62. [Google Scholar]
[9]. Lau SK, Tideman H, Wu PC, Ameloblastic carcinoma of the jaws. A report of 2 casesOral Surg Oral Med Oral Pathol Oral Radiol Endod 2004 85:78-81. [Google Scholar]
[10]. Kawauchi S, Hayatsu Y, Takahashi M, Furuya T, Oga A, Niwa S, Spindle-cell ameloblastic carcinoma: A case report with immunohistochemical, ultrastructural, and comparative genomic hybridization analysesOncol Rep 2003 10:31-34. [Google Scholar]
[11]. Ismail SB, Zain RB, Yaacob HB, Abraham MT, Ameloblastic carcinoma (spindle cell variant)Pathology 2009 41:292-95. [Google Scholar]
[12]. Jindal C, Palaskar S, Kaur H, Shankari M, Low-grade spindle-cell ameloblastic carcinoma: Report of an unusual case with immunohistochemical findings and review of the literatureCurr Oncol 2010 17:52-57. [Google Scholar]
[13]. Kamath VV, Satelur K, Yerlagudda K, Spindle cell variant of ameloblastic carcinoma arising from an unicystic amelobastoma: Report of a rare caseDent Res J 2012 9:328-33. [Google Scholar]
[14]. Ansari HA, Ray PS, Khan N, Khan AH, Spindle-cell ameloblastic carcinoma of the maxilla with adenoid cystic carcinoma - like areas: A new variant?Indian J Pathol Microbiol 2015 58:513-15. [Google Scholar]
[15]. McNaught MJ, Turella SJ, Fallah DM, Demsar WJ, Spindle cell variant of ameloblastic carcinoma: A case report and review of literatureMil Med 2015 180(5):614-17. [Google Scholar]
[16]. Matsushita Y, Fujita S, Yanamoto S, Yamada S, Rokutanda S, Yamashita K, Spindle cell variant of ameloblastic carcinoma: A case report and literature reviewOral Surg Oral Med Oral Pathol Oral Radiol 2016 121(3):54-61. [Google Scholar]