Rheumatoid Arthritis (RA) is a chronic systemic inflammatory disorder of unknown aetiology that may affect many tissues and organs but principally attacks the joints symmetrically producing a non-suppurative proliferative and inflammatory synovitis. Regarding clinical course of the disease, vast majority of the patients exhibit a pattern of persistent and progressive disease activity that waxe and wane in intensity over time. In clinical practice, there is general agreement that rheumatoid inflammation should be controlled as soon as possible, as completely as possible, and that control should be maintained for as long as possible consistent with patient safety [1]. As the goal of treatment is to control of rheumatoid inflammation, it is clear that the management of RA should include systematic and regular quantitative evaluation of disease activity. So an accurate, valid and reliable method for assessment of disease activity is one of the most important issues for guiding the treatment protocol during management of the RA patients. Several composite indices have been developed to assess clinical disease activity like SDAI (Simplified Disease Activity Index), CDAI (Clinical Disease Activity Index), DAS 28 and DAS 28-3 score (Modified Disease Activity Score). Among these DAS 28-3 (using three variables- tender joint count, swollen joint count and ESR) score system is the most commonly used parameter now-a-days to measure the disease activity as it is extensively validated and clinically interpretable [2] but it is mainly dependent on clinical findings. In this era of evidence based clinical practice, addition of laboratory based investigations like some haematological parameters including Haemoglobin (Hb) level, Platelet count and Mean Platelet Volume (MPV) which are found to be altered in this chronic inflammatory disease can further improve the assessment status of disease activity.
In the previous years, various authors have studied the association between disease activity of RA and these haematological parameters separately. Most of them found inverse relationship between disease activity and Hb level and thrombocytosis with high disease activity [3–7]. But data regarding MPV is not conclusive. However, evidences have shown an important role of MPV as a marker of inflammation and disease activity [8]. But there is hardly any study which provided the association between these three haematological parameters altogether and markers of disease activity in RA on the basis of reliable index like DAS 28-3 score specially in Eastern region of India.
The current study intended to find out the association of DAS 28-3 score of RA with platelet count, MPV and Hb level so that an assessment of disease activity of RA can be done by these haematological parameters in addition to conventional serological markers so that a better management of the patients of RA can be done in the population of underprivileged society.
Materials and Methods
After obtaining approval from Institutional ethics committee, this hospital based cross-sectional study was carried out in the Department of Pathology, R.G. Kar Medical College, Kolkata in collaboration with the Rheumatology clinic of the same institute over a period of one year. Among the patients attending RA clinic, newly diagnosed cases of RA according to “The 2010 ACR-EULAR criteria for classification of Rheumatoid Arthritis” (ACR- American College of Rheumatology; EULAR- European League Against Rheumatism) were included [9]. Patients suffering from malignancies, aplastic anemia, chronic renal failure, diabetes mellitus, hypertension, arterial or venous thrombosis, other systemic inflammatory diseases like inflammatory bowel disease, psoriasis, Systemic lupus erythaematosus, bleeding disorders or RA patients receiving anti-platelet or anti-coagulant drugs, steroids, DMARDs (Disease-Modifying Anti-Rheumatic Drugs) were excluded from the study because the haematological parameters in relation to our study may be altered in the above mentioned diseases and systemic disorders irrespective of disease activity of RA [8].
Along with the patients, age and sex matched healthy controls comprising healthy relatives of the patients who volunteered to participate were screened clinically and by laboratory investigations were also included.
After obtaining written informed consent, a detailed history was taken and relevant clinical examination of the patients was done at rheumatology clinic of the Institution. DAS 28-3 score of the cases based on tender joint count, swollen joint count and ESR (Erythrocyte sedimentation rate) was calculated by computer with internet connection (www.das-score.nl) [10].
Laboratory tests included estimation of Hb, ESR, platelet count, MPV, Fasting Blood Sugar (FBS), serum urea and creatinine, RF (rheumatoid factor), CRP (C-Reactive protein) and anti-CCP (Anti-cyclic citrullinated peptide) antibody. Blood samples were drawn by clean aseptic venepuncture using 23G needle and were collected in 3 types of vial: 1) vial with EDTA (Ethylenediaminetetra-acetic acid) anti-coagulant for Hb, ESR, platelet count and MPV estimation; 2) vial with Fluoride-oxalate anti-coagulant for estimation of FBS level; and 3) vial without any anti-coagulant for determination of urea, creatinine, CRP, RF and Anti-CCP antibody level. The blood samples were analysed between 1-3 hours of collection by automated cell counter (Sysmex KX-21) based on impedance technology. Hb level, platelet count and MPV of the cases and controls were measured by the automated cell counter. Platelet count and morphology was further evaluated by examining the blood films stained by Leishman’s stain under binocular light microscope (Olympus CH20i). ESR was measured by conventional Westergren method.
Tests for Anti-CCP antibody were done with the INOVA QUANTA Lite® CCP3 IgG ELISA kit. CRP levels were analysed with a Cobas Integra System (Roche diagnostics, Switzerland). RF was estimated using latex agglutination method.
Statistical analysis was performed using SPSS version 16.0 software. All analyses were two-tailed and differences were considered statistically significant when p <0.05. For continuous variables, the mean and standard deviation were calculated; unpaired t-test was used to compare variance between groups of patients with low to moderate disease activity and patients with high disease activity.
Results
Total 80 patients of RA (Female=60; Male=20) were selected by simple random sampling as per inclusion and exclusion criteria. The sample size was calculated from the formula: n= [{(Zα/2)2 X p X (1-p)} ÷d2] where n is the sample size, Zα/2 is a constant of value 1.96 considering 95% confidence interval, p is the estimated proportion of high disease activity of RA cases is taken as 16% and d is the precision which is taken as 8.
A total of 48 patients (F: M=41:7) had high disease activity (DAS 28-3>5.1) (Group A) and 32 (F: M=19:13) had low to moderate disease activity (DAS 28-3 ≤5.1) (Group B). Along with them, 80 age and sex matched healthy subjects were selected as control group (Group C). Observations regarding various study variables are represented in [Table/Fig-1,2].
Study variables based on Platelet count, MPV and Hb level of RA patients with low to moderate disease activity and high disease activity compared to healthy control.
| Study parameter | Control population (Group C) | Low to moderate disease activity (Group B) | High disease activity(Group A) |
|---|
| Platelet count (lac/cmm) (mean±SD*) | 2.07±0.44 | 2.17±0.69p=0.38 | 4.53±1.22p<0.001 |
| MPV (fl) (mean±SD) | 9.4±1.39 | 10.19±1.09p=0.004 | 11.86±1.13p<0.001 |
| Hb in male (g/dl) (mean±SD) | 13.31±0.87 | 12.25±1.0p=0.003 | 10.05±1.58p<0.001 |
| Hb in female (g/dl) (mean±SD) | 12.01±0.83 | 11.91±1.21p=0.67 | 10.12±1.84p<0.001 |
*SD= Standard Deviation
Comparison of study variables based on Platelet count, MPV and Hb level between RA patients with low to moderate disease activity and high disease activity.
| Study parameters | Low to moderate disease activity (Group B) | High disease activity(Group A) | p-value |
|---|
| Platelet count (lac/cmm) (mean±SD*) | 2.17±0.69 | 4.53±1.22 | <0.001 |
| MPV (fl) (mean±SD) | 10.19±1.09 | 11.86±1.13 | <0.001 |
| Hb in male (g/dl) (mean±SD) | 12.25±1.0 | 10.05±1.58 | 0.001 |
| Hb in female (g/dl) (mean±SD) | 11.91±1.21 | 10.12±1.84 | <0.001 |
*SD= Standard Deviation
The mean (±SD) age of disease onset in this study was 37.73 (±9.45) years and ranged from 21 years to 60 years.
The Hb levels (Mean±SD) of male patients with low to moderate disease activity and with high disease activity were 12.25±1.0 g/dl and 10.05±1.58 g/dl, respectively. The difference between these two groups regarding Hb level was statistically highly significant (p=0.001). Comparing with the control population (comprising of 20 healthy male subjects) having Hb level (mean±SD) of 13.31±0.87 g/dl, both the group had significantly lower Hb level.
The Hb levels (Mean±SD) of female patients with low to moderate disease activity and with high disease activity were 11.91±1.21 g/dl and 10.12±1.84 g/dl, respectively. The difference between these two groups regarding Hb level was statistically highly significant (p=0.0003). However, comparing with the control population (comprising 60 healthy females) having Hb level (mean±SD) of 12.01±0.83 g/dl, the female patients with high disease activity had statistically significant low Hb level (p<0.001).
From the above observations, it is quite clear that patients with high disease activity (Group-A) had significantly lower Hb level than the patients with low to moderate disease activity (Group-B) both in male and female groups.
The platelet counts (mean±SD) of age and sex matched control population, patients with low to moderate disease activity and patients with high disease activity were 2.07±0.44 lac/cmm, 2.17±0.69 lac/cmm and 4.53±1.22 lac/cmm, respectively. On statistical analysis, it was found that there was no significant difference between the platelet count of control group (Group-C) and that of patients with low to moderate disease activity (Group-B). However, patients with high disease activity (Group-A) had a significantly higher platelet count comparing with the control group (p<0.001) and with the group of patients having low to moderate disease activity (p<0.001).
This study also dealt with the MPV of RA patients. The MPV (mean±SD) of age and sex matched control population, patients with low to moderate disease activity and patients with high disease activity were 9.4±1.39 fl, 10.19±1.09 fl and 11.86±1.13 fl, respectively. On statistical analysis, it was found that the patients with high disease activity had significantly higher MPV compared to patients with low to moderate disease activity (p<0.001).
Discussion
The concept of disease activity is essential in rheumatology, for guiding the treatment and influencing the outcome in RA [11]. This study was focused on finding an association between different haematological parameters (Hb, Platelet count and MPV) and DAS 28-3 score (Modified Disease Activity Score) so that these evidence-based and cost-effective parameters can be used to assess disease activity and thus, can improve clinical management of RA.
The study clearly proved that patients with high disease activity had significantly lower Hb level than the patients with low to moderate disease activity both in male and female groups which is consistent with previous studies done by Jeffrey MR., Agrawal S et al., and Borah DJ et al., [12–14].
Regarding platelet count, this study showed that platelet count increases with increase in disease activity in RA patients. This result is supported by the study done by Milovanovic M et al., where they found elevated platelet count in patients with active RA [15]. In RA, thrombopoietin level is increased as acute phase reactant [16]. Apart from thrombopoietin, different cytokines like IL-6, IL-1β, and IL 4 are also positively correlated with high disease activity and they also account for pathologic thrombocytosis in RA [17,18].
This study also found that patients with high DAS 28-3 score had higher MPV levels. This result is consistent with the inference drawn by previous studies done by Milovanovic M et al., and Yazici et al., where they found a positive correlation between MPV and disease activity [15,19].
This increase in MPV can be explained by the fact that different cytokines (e.g., IL-6, thrombopoietin etc.,) cause increased platelet production in active RA as a part of inflammatory process and these newly formed young platelets are larger [Table/Fig-3] and more active causing increased risk of atherosclerosis and cardio-vascular disease in RA patients with high disease activity [20].
Peripheral blood smear of a Rheumatoid Arthritis patient showing many platelets; one of them is large platelet (arrow) (Leishman’s stain, oil immersion field, 100X)
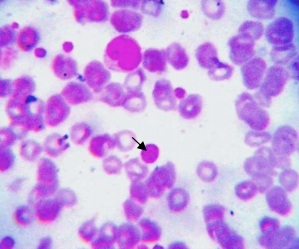
In contrast to this, the studies done by Kisacik et al., and Gasparyan et al., demonstrated decreased MPV in patients with active RA [21,22]. This controversial result might be due to presence of some confounding factors yet to be discovered and also due to presence of additional challenge relating to methodological issues. Due to these conflicting reports, further studies on large scale are needed to clarify the discrepancies.
Limitation
There were certain limitations to this study. It was a cross-sectional and hospital based study. Other measures of disease activity like IL2, TNFα, or HLA-DRB1 alleles, which have shown good association with disease activity, could not be done in our settings. Further longitudinal studies involving larger sample sizes would provide insight into these findings specially regarding MPV in future.
Conclusion
This hospital-based cross-sectional study demonstrated lower Hb level in RA patients with high disease activity compared to patients with low to moderate disease activity both in male and female groups. Regarding platelet count and MPV, this study demonstrated that patients with high disease activity have statistically significant higher platelet count and MPV compared with the patients with low to moderate disease activity. So, from Hb level, platelet count and MPV, we can predict disease activity in RA patients which can guide us for proper and aggressive management to prevent further disease progression of RA.
*SD= Standard Deviation
*SD= Standard Deviation