Infectious granulomatous dermatitis is seen in a limited number of skin infections. Granulomatous lesions of skin often presents as a diagnostic challenge even to the most experienced dermatopathologist. It forms a common and intriguing problem in day to day routine practice. Arrival at an exact and appropriate diagnosis is mandatory for successful treatment. Histopathological examination remains a time tested tool for establishing a correct diagnosis in various diseases of organ system of the body [1].
Clinical lesions of skin often reveal surprising underlying pathology, since similar histological finding is produced by several causes or vice versa [2]. Hence, all the skin lesions diagnosed clinically should undergo histopathological examination with routine haematoxylin and eosin along with other special stains that might help in identifying the type and aetiological agent of the granuloma [3].
Granuloma can be defined as any focal chronic inflammation consisting of clusters of epithelioid cells surrounded by lymphocytes and plasma cells [4]. Completely developed granulomas shows sheets of epithelioid histiocytes and giant cells, however, subtle lesion containing a few epithelioid histiocytes also can be considered as granulomatous lesion [5]. There is difficult and ambiguity of satisfactorily classifying granulomatous reaction [6]. Previous literature has made an attempt to classify granulomatous lesions of skin on the basis of pathophysiology, aetiology, immunology and morphological findings [5]. Hence, in the present study, we have made an attempt to classify the granulomatous lesions of skin based on its aetiology and morphology of the granuloma.
Six histological types of granulomas can be identified on the basis of constituent cells and other changes within the granulomas: Tuberculoid; Sarcoidal; Necrobiotic; Suppurative: Foriegn body: Mixed inflammatory [7,8].
Materials and Methods
All the skin biopsies with the granulomatous reaction diagnosed histopathologically were reviewed and assessed in detail. Complete clinical information and relevant history were recorded. In each of the case, the Haematoxylin and Eosin (H&E) stained paraffin sections along with relevant special stains like Periodic acid Schiff (PAS), Zeihl-Neelsen (ZN), Grocott, Gram, Giemsa, Fite faraco stain etc. were done whenever required and studied under the light microscope.
Inclusion criteria: All type of skin biopsies diagnosed histopathologically, to have granulomatous lesion.
Exclusion criteria: Inadequate and poorly preserved skin biopsies were excluded.
Results
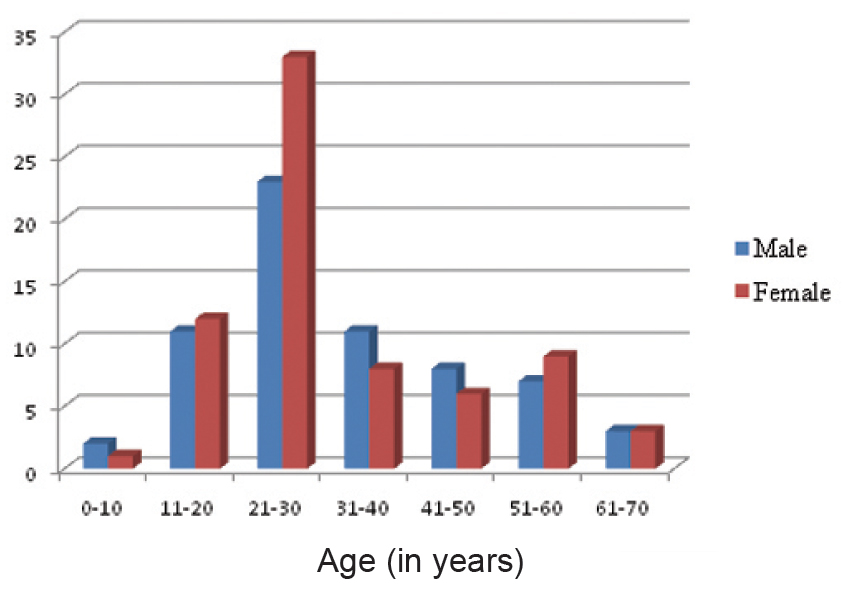
Histopathological sections of 137 granulomatous lesions were analysed. We observed that females (52.77%) were more prone for granulomatous lesions as compared to males (47.4%) [Table/Fig-1]. Out of total 137 cases, maximum numbers of biopsies were from head and neck region accounting for 41.6% of cases. 25 cases (18.24%) were from lower limb, 10 cases (7.5%) were from upper limb and 5 cases (3.6%) from chest, abdomen and back. Anatomical sites were not mentioned in the case records reviewed retrospectively in 29.1% (40) of the cases.
Distribution of granulomatous skin lesions based on age and gender.
Out of the total 137 cases, 109 (79.56%) showed tuberculoid granulomas, 12 cases (8.75%) revealed foreign body granulomas, seven cases (5.1%) each of suppurative and necrobiotic type and one case (0.7%) each of histiocytic and mixed inflammatory type of granuloma. Details of histopathological features of granulomatous lesions of skin are listed in [Table/Fig-2,3].
Histopathological diagnosis of granulomatous lesions of skin.
| Sr. No | Histopathological diagnosis | Number of cases | % of cases |
|---|
| 1 | Leprosy | 91 | 66.4% |
| 2 | Lupus Vulgaris | 11 | 8% |
| 3 | Scrofuloderma | 7 | 5.1% |
| 4 | Fungal infection | 13 | 9.5% |
| 5 | Actinomycosis | 7 | 5.1% |
| 6 | Milkers nodule | 5 | 3.6% |
| 7 | Parasitic granuloma | 1 | 0.7% |
| 8 | Syphilis | 2 | 1.5% |
| Total | 137 | 100% |
Histomorphological features of granulomatous lesions of skin.
| Sr. No | Histopathological Diagnosis | Location of granuloma | Epidermal change |
|---|
| 1. | Leprosy (91 cases) | | |
| a. | Tuberculoid Leprosy (30 cases) | Upper dermis | Atrophic |
| b. | BTL (48 cases) | Upper dermis | Atrophic |
| c. | BL (6 cases) | Upper & mid dermis | Atrophic |
| d. | LL (5 cases) | Upper & mid dermis Gernz Zone | Atrophic |
| e. | Histioid leprosy (2 cases) | Upper & mid dermis | Atrophic |
| 2. | Lupus Vulgaris (11 cases) | Upper dermis -5 casesUpper & mid dermis-6 cases | Acanthosis -5 casesUnremarkable – 6 cases |
| 3. | Scrofuloderma (7cases) | Upper & mid dermis | Acanthosis - 4 casesUnremarkable – 3 cases |
| 4. | Fungal infections (13 cases) | Mid dermis | Acanthosis-5 casesPsudoepitheliomatousHyperplasia-6 casesUnremarkable-2 cases |
| 5. | Actinomycosis (7 cases) | Mid dermis | Acanthosis |
| 6. | Milkers Nodule (5 cases) | Mid dermis | Koilocytic change |
| 7. | Parasitic granuloma (1 cases) | Mid dermis | Acanthosis, Hyperkeratosis |
| 8. | Syphilis (2 cases) | Mid dermis | Acanthosis – 1 casePsudoepitheliomatousHyperplasia-1 case |
*BTL – borderline tuberculoid leprosy; BL – borderline leprosy; LL – lepromatous leprosy
The 109 cases of tuberculoid granuloma were further subclassified based on histomorphology and aetiology. Maximum number of cases of tuberculoid type of granulomas were leprosy accounting for 66.4% of cases [Table/Fig-4,5 and 6], followed by lupus vulgaris 8% of cases [Table/Fig-7,8] and Scrofuloderma accounting for 5.1% of cases [Table/Fig-9]. On ZN staining, tuberculoid granulomas exhibited 16 cases that were AFB positive [Table/Fig-10]. The leprosy cases were further subdivided as per Ridley Jopling’s classification [9] as shown in [Table/Fig-3].
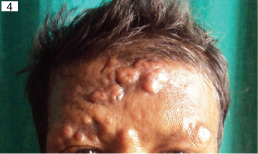
Clinical picture of multiple nodules over a face in case of leprosy.
Microphotograph of skin showing granuloma around adnexa and nerve bundles (arrow) in case of tuberculoid leprosy (H&E, x400).
Microphotograph of skin showing subepidermal clear zone (arrow) and aggregation of foamy macrophages in case of lepromatous leprosy (H&E, x400).
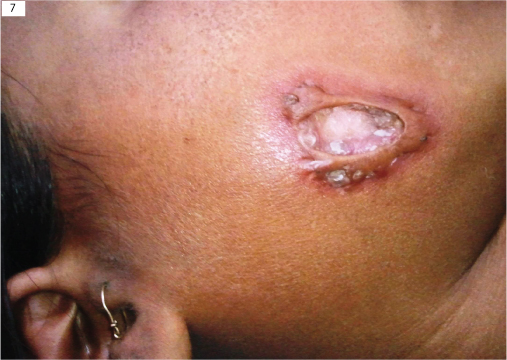
Clinical picture of ulcerative nodule over a face in case of lupus vulgaris.
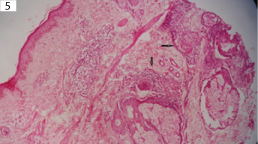
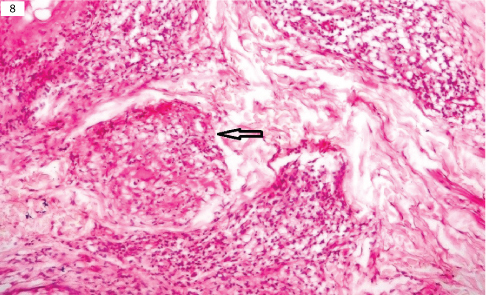
Microphotograph of lupus vugaris showing well formed granulomas (arrow) in upper dermis (H&E, x400).
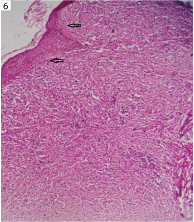
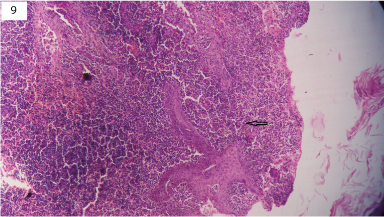
Microphotograph of scrofuloderma showing ulcerated epithelium and underlying abscess with well formed granulomas and langhan’s type of giant cell (arrow) (H&E, x400).
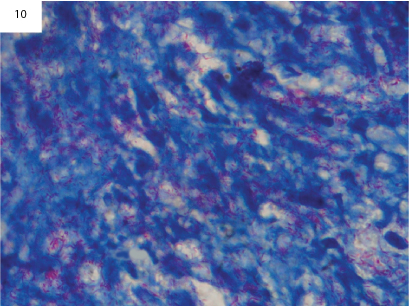
Microphotograph of acid fast bacilli in lepromatous leprosy (Zeihl-Neelsen stain, x1000).
Another uncommon infectious granulomatous lesions noted in our study were fungal infections (chromoblastomycosis, mucormycosis and aspergillosis) actinomycosis, Milker’s nodule and parasitic granuloma.
Thirteen cases of fungal infections were identified. Few of cases were demonstrated on H&E stain and majority of cases required special stains like PAS and GMS to identify the fungus. All these cases of fungal infection were later confirmed by the culture. Among the fungal infection seven cases of chromoblastomycosis [Table/Fig-11], four cases of aspergillosis, and one case each of rhinosporidiosis [Table/Fig-12] and mucormycosis [Table/Fig-13] were seen. Histopathologically, majority of fungal infections presented as suppurative/mixed inflammatory type of granuloma.
Microphotograph of Chromoblastomycosis showing pigment spores (arrow) which are round, brown thick walled sclerotic bodies (H&E, x1000).
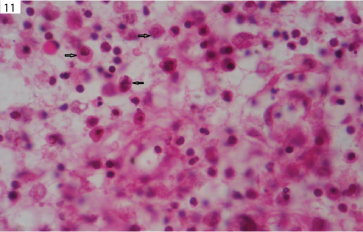
Microphotograph of rhinosporidiosis showing its wall and spores (arrow) (&E, x100).
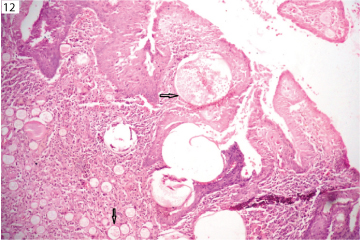
Microphotograph of mucormycosis showing broad aseptate fungus with irregular branching (arrow) (H&E, x1000).
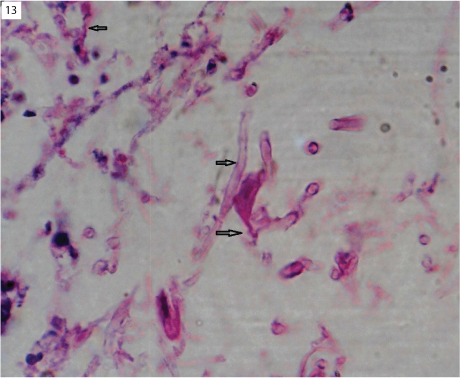
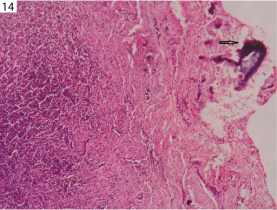
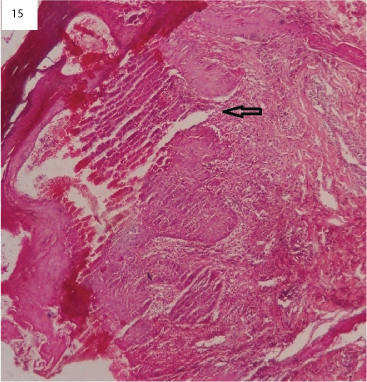
In our study, seven cases of actinomycosis were found and histopathologically showed suppurative granulomas with actinomycotic colonies. Actinomycotic colonies were seen on H&E stain [Table/Fig-14], and later were confirmed by Grams staining. Five cases of Milker's nodule were identified. Milker's nodule is a viral infection caused by pox virus. Histopathologically, epidermis show koilocytic change with dermis showing mixed inflammatory type of granuloma [Table/Fig-15], in all the five cases histopathological diagnosis had correlated with clinical diagnosis. One case of parasitic granulomas was noted in the present study and histologically showed mixed inflammatory type of granuloma, containing parasite embedded in eosinophilic material. The parasite showed thick, laminated cuticle with longitudinal ridges and transverse striae as shown in [Table/Fig-16]. Two cases of syphilis were encountered in our study which were diagnosed on histopathology and were confirmed by serology.
Microphotograph showing mixed inflammatory granuloma with actinomycotic colonies (arrow) (H&E, x40).
Microphotograph of Milker’s nodule showing reticulated vesicles (arrow) affecting the epidermis with the dermis showing granulomas and mononuclear infiltrate (H&E, x40).
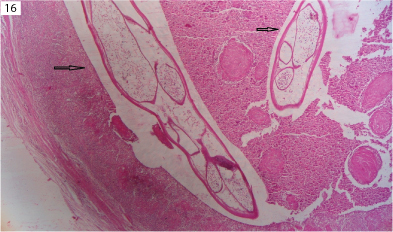
Microphotograph of dirofilaria parasite (arrow) surrounded by granulomas, giant cell and lymphocyte (H&E, x40).
Discussion
Since early nineteenth century, infectious granulomatous dermatitis has been a topic of interest till date. It poses a common and intriguing problem in day to day practice to both dermatologist and histopathologist. Arrival to a proper diagnosis is mandatory for appropriate treatment. Histopathology remains a gold standard for establishing an accurate diagnosis [10].
In the present study, age group of the patients was between 6-70 years with mean age being 29.25 years. Above finding is in concordance with Dhar’s [1] and MNU Zafar’s study [10]. Our study shows that female had higher predilection for granulomatous lesions than males as seen in earlier study of MNU Zafar’s [10], while Dhar’s study [1] found male preponderance.
In our study, the prevalence of leprosy was high. We found 66.4% of all dermatology patients with granulomatous disease having leprosy. Among the Leprosy cases BTL formed the largest subgroup of Ridley and Jopling classification accounting for 52.74% (48/91) of the cases followed by Tuberculoid Leprosy (TL) in 32.96% (30/91) of the cases.
This finding is alarming to all concerned health care providers dealing with respect to prevention, control and management of leprosy. The exact reason for high prevalence of leprosy could not be identified, but probably due to poverty, overcrowding, lack of personal hygiene, education or ignorance about the disease.
Also, our centre being one of the few hospitals where free camps are conducted for skin lesions and biopsies are carried out have contributed to the higher prevalence of leprosy.
Histopathologically, BTL and TL show epithelioid granulomas with langhans’ and foreign body giant cells and needs to be differentiated from sarcoidosis and noncaseating tuberculoid granuloma [11,12]. In these cases, Fite Faracco stain is of no use because of sparse bacilli [13]. In the present study, only four cases of BT and none of TL were positive for lepra bacilli. However, location of granulomas around neurovascular bundle, erector pili muscle and adnexa in combination with clinical pictures were helpful. Borderline leprosy (BL) and lepromatous leprosy (LL) are characterized by histiocytic granulomas and are strongly positive for lepra bacilli, thus having no difficulty in appropriate diagnosis. In the present study five cases of BL, five cases of LL and two cases of histioid leprosy were positive for lepra bacilli. Histopathological examination in leprosy serves dual purpose: establishment of diagnosis and assignment of the disease to one of the seven recognized types which are essential for therapeutic purpose [1].
In our study, leprosy (91/137) accounts for 66.4% of granulomatous lesions of skin. Our finding is in concordance with Dhar S, Bal A et al., Singh R et al., and discordance with Zafar MNU et al., [1,14,15,10]. Borderline tuberculoid leprosy was the most common subtype of leprosy encountered in our study and similar findings were noted by Bal A et al., and Gautam K et al., [14,16].
In a study done by Nayak SV et al., showed positive for lepra bacilli with fite faracco stain in 25 cases (44.64%) out of 56 cases [17]. Harish S premi et al., showed positivity for lepra bacilli in 9 cases (25.74%) out of 35 cases [18]. In our study, 11 cases (12.8%) showed positivity for lepra bacilli. The probable decrease in the percentage of positive cases for lepra bacilli using fite faracco stain in our study may be due to more number of cases of leprosy (91 cases) compared to above studies. Moreover, the positivity of fite faracco stain depends on bacterial load/index. Hence, the major recent advancement in laboratory diagnosis, include molecular techniques such as PCR (Polymerase Chain Reaction) and reverse PCR in identification of causative organism in clinical specimens. Since the molecular techniques are costly, it has not yet been approved as a routine clinical test in the diagnosis of leprosy that needs greater attention.
Another common infectious granulomatous lesion of skin noted in our study was cutaneous tuberculosis in 13.1% of cases (18/137). Cutaneous tuberculosis represents reinfection and reactivation of tuberculosis [19]. Cutaneous tuberculosis is still prevalent in the developing countries accounting for 0.4% of patients with skin diseases [20]. However, in India incidence has fallen from 2% to 0.15% [21].
In the present, study 18 cases of cutaneous tuberculosis were found out of which lupus vulgaris accounts for 8% of cases followed by scrofuloderma in 5.1% of cases. Histologically caseation necrosis in an epithelioid granuloma is diagnostic, but its absence does not rule out diagnosis of tuberculosis [22]. Demonstration of acid fast bacilli by ZN stain and Auramine rhodamine stain is specific; however, they are not detected with ease and literature reports 13-15% positivity in lupus vulgaris and upto 50% positivity in srofuloderma [23,24]. The accurate diagnosis can be done by PCR by dectecting the mycobacterial DNA in the tissues [3]. In the present study, none of the case of lupus vulgaris and scrofuloderma revealed acid fast bacilli by ZN stain and Auramine rhodamine stain. Though ZN stain and Auramine Rhodamine was negative the diagnosis of lupus vulgaris and scrofuloderma was made, due to the presence of caseous necrosis, raised ESR and other relevant clinical findings.
In the present study, lupus vulgaris was the commonest cutaneous tuberculosis. Our finding is in consistent with Zafar MNU et al., [10] Khan Y et al., Singh G and Kumar B et al., who also found lupus vulgaris as the commonest cutaneous tuberculosis [10,25–27]. None of the case of cutaneous tuberculosis revealed AFB positive. According to Singh R et al., and Veena S et al., [15,28]. AFB was found in 11.5% and 6.45% of cases, respectively.
In order to provide accurate diagnosis of granulomatous lesions of skin there should be cooperation between clinician and pathologist which is very important in the field of dermatology for the greatest benefit from the biopsy. The clinician should provide detailed clinical history which includes age, sex, site, type and colour of skin lesions with list of differential clinical diagnosis [29].
Limitation
The study lacks information regarding the post treatment follow-up of the patient and also, the inter observer variation of clinician and histopathologist is not considered in our study.
However, as in our study, the data have been collected from various records and the above mentioned limitations were beyond our control.
Conclusion
Infectious granulomatous lesions of skin account for significant mortality and morbidity with leprosy and tuberculosis as the leading cause in the present days. Hence, early and accurate diagnosis can be done with combined approach of both clinician and pathologist for identification of aetiology and appropriate therapy and outcome of specific diseases.
*BTL – borderline tuberculoid leprosy; BL – borderline leprosy; LL – lepromatous leprosy