Enhanced External Counter Pulsation (EECP) treatment is a non-invasive, mechanical, out-patient treatment for patients with chronic stable angina and heart failure who are symptomatic in spite of optimal medical management and conventional re-vascularization strategies such as Percutaneous Transluminal Coronary Angioplasty (PTCA) or Coronary Artery Bypass Surgery (CABG) [Table/Fig-1] [1–3]. EECP treatment is provided by Electrocardiogram (ECG) synchronised, cyclic inflation and deflation of cuffs in the lower extremities, which causes shear stress on vascular endothelium similar to that of a moderate to severe intensity aerobic exercise. This shear stress works on myocardial supply side by decreasing ischemia by opening dormant collaterals and promoting angi-ogenesis [4–8]. It reduces myocardial oxygen demand by improving peripheral endothelial function mediated through increase in the plasma nitric oxide and decrease in endothelin level [9–11]. This mechanism of improving myocardial supply and decreasing the myocardial demand has been shown to improve the patients clinical Canadian Cardiovascular Society (CCS) angina class, New York Heart Association (NYHA) shortness of breath symptoms, decrease their dependency on nitroglycerine for frequent chest pain, ability to do more work without exercise induced ischemia and improved quality of life [12–14]. The effect of EECP on central blood pressure and arterial stiffness in patients with Left Ventricular (LV) dysfunction immediately post-EECP has not been studied. We tested our hypothesis in patients with normal brachial systolic pressure and decreased brachial systolic blood pressure with associated LV dysfunction, on which EECP treatment may have differential effect. This differential effect in systolic pressure may be mainly driven by improvement in LV function. This favorable vascular effect may be the reason for improvement in clinical symptoms in Coronary Ar-tery Disease (CAD) patients with LV dysfunction.
Materials and Methods
A total of 72 consecutive patients who underwent EECP treatment for symptomatic CAD with LV dysfunction and had completed the full course of 35 days of EECP sessions were enrolled in the study from 2014 to 2016. These patients recruited for the study were divided into two groups based on their baseline brachial systolic pressure with cut-off pressure of 100mmHg. In the first group patients had baseline brachial systolic pressure >100mmHg and in the second group the patients had baseline brachial systolic pressure ≤100mmHg [14]. The study was approved by the Chettinad Academy of Research and Education Institutional Human Ethic Committee and written informed consent was obtained from all patients. The majority of the patients in the study groups were male (n=61). The study was conducted at a single centre, Chettinad Hospital and Research Centre, Chennai, Tamil Nadu, India.
To be eligible for the study the patients had to meet the following criteria. Patients who were stable with proven CAD with LV dysfunction, symptomatic with angina or shortness of breath, refractory to medical management and not a candidate for or refused conventional revascularization procedure.
Patients were excluded if they have any one of these following: Acute coronary syndrome within 1 month period, PTCA or CABG within 3 months, deep vein thrombosis, aortic aneurysm, any history of aneurysm repair, severe peripheral arterial disease, significant valvular heart disease, arrhythmia that would interfere with ECG gating, hypertrophic obstructive cardiomyopathy, severe aortic regurgitation, any bleeding diathesis, severe pulmonary hypertension and uncontrolled hypertension (systolic pressure > 180mmHg and diastolic pressure > 100mmHg). Pregnant women, patient taking any ayurvedic preparation or who were enrolled in cardiac rehabilitation program were also excluded.
Patient’s functional class was measured by CCS for angina and NYHA class for heart failure by physicians. All patient were scheduled for 35 one hourly sessions of EECP treatment over 6 weeks. Every patient’s time schedule for taking one hour EECP session was fixed and strictly followed. Time changes were not permitted. Patients were instructed not to take any food, coffee or caffeine drinks or to smoke for at least two hours prior receiving EECP treatment. All cardiac medications were kept unchanged during the 35 days treatment period. Patients were instructed not to take any sublingual nitrate for at least 2 hour prior to EECP treatment.
Enhanced External Counter Pulsation therapy: This therapy system is an electro-mechanical system consisting of a specialized treatment table with integrated valve regulatory system, attached to specialized two sets of three cuffs and a treatment console. The console comprises of air compressor and touch screen panel to control inflation and deflation of the cuffs in synchronisation with the patients ECG. The cuffs inflate sequentially from calves to upper thigh in early diastolic phase and deflate simultaneously at the end of diastolic phase. This results in increased retrograde blood flow during diastole thereby augmenting central aortic blood pressure and flow. This is called diastolic augmentation [15]. At the end of diastole simultaneous cuff deflation results in decreased systemic peripheral vascular resistance and decrease myocardial oxygen demand. The cuff pressure can be increased from 80 to 300mmHg. The central diastolic augmentation can be roughly estimated by using finger Plethysmography. The pressure is adjusted in the cuffs to reach the Diastolic Augmentation (DA) ratio of more than one, which has been shown to have a favorable haemodynamic effect. The treatment is given on an out-patient day care basis, one-hour session per day, six days per week over six weeks for a total of 35 hours.
Brachial Cuff Blood Pressure: In all the patients, brachial blood pressure was taken 15 minutes prior to starting EECP treatment, in the left arm in sitting position in a quiet room. A standard calibrated mercury sphygmomanometer was used for measuring brachial blood pressure. To determine the pre-EECP blood pressure, the first 7 days (1st-7th session) measured blood pressure prior to starting EECP was averaged and recorded as pre-EECP blood pressure. Similarly to determine the post-EECP blood pressure the last 7 days (29th-35th session) measured blood pressure prior to starting EECP was averaged and recorded as post-EECP blood pressure. Averaging of blood pressure was done to eliminate the rise in the blood pressure during the first couple of days due to anxiety and to reduce any natural variations in blood pressure. We have also taken the blood pressure only prior to starting EECP even for post-EECP blood pressure recording to eliminate the acute drop in blood pressure, which usually happens during one hour EECP treatment. This acute temporary reduction in blood pressure may interfere with our study analysis and interpretations.
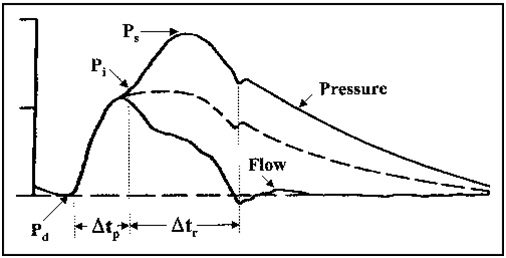
Pulse Wave Form Analysis: Central aortic systolic pressure and arterial stiffness parameters were derived non-invasively by using the SphygmoCor system (Atcor Medical Sydney, Australia). This system uses a peripheral pressure wave form measured at the wrist in the radial artery by using application tonometry, after 20 sequential good quality waveforms were acquired, these waves are averaged and a validated generalised transfer function was used to derive corresponding central aortic wave form with high accuracy. Some of these indexes derived from this generated central aortic pressure wave form are mentioned in the [Table/Fig-2]. The (Ps-Pd) is augmented systolic pressure wave amplitude, composed of forward traveling wave with amplitude (Pi-Pd) and reflected wave arriving from periphery with amplitude (Ps-Pi). The forward travelling wave is generated by LV ejection and this un-augmented pressure wave is related to central elastic artery stiffness and the reflected pressure wave depends on the stiffness of the entire arterial tree. The increased amplitude of the central systolic pressure due to reflected wave coming from the periphery is called Augmentation Pressure (AP) shown as (Ps-Pi). Augmentation Index (AIx) is derived from the AP and central pulse pressure expressed as in percentage shown as (Ps-Pi)/(Ps-Pd). AIx is overall reflection of the stiffness of entire vascular system including the central elastic artery, muscular artery and to lesser extent the arterioles. Since AIx is influenced by heart rate it is expressed as index normalized for heart rate of 75 beats/min (AIx @ 75). Left ventricular Ejection Duration (ED) is the time duration between start of the systolic pressure waveform (i.e., minimum diastolic pressure) Pd to the dicrotic notch (Closure of aortic valve).
Derived ascending aortic pressure wave form.
Pd - minimum diastolic pressure, Ps - peak systolic pressure, Pi - an inflection point that indicates the beginning upstroke of the reflected pressure wave, Δtp - the round trip travel time of forward wave of ascending aorta to a major reflec-tion sit and back, Δtr is the systolic duration of reflected wave, the broken wave is the pressure wave if there is no reflection wave from periphery.
After brachial blood pressure measurement was completed, a SphygmoCor diagnostic test was performed by applying the tonometer in the radial artery on the first day of EECP and the last day after completion of 35 sessions of EECP. Three consecutive measurements were done in sitting position by the same operator. Two measurements which had close values were chosen and the average of these 2 measurements was taken as final measurement. The third reading was discarded. Care was taken that in all measurement the SphygmoCor quality control operator index was achieved more than 95%.
Echocardiography: All patients’ pre and post- ECHO was done within one week before starting the treatment and within one week of completion of the 35 sessions of the EECP treatment. The ECHO physician was blinded and had no knowledge about patient identity or whether it was pre- or post -EECP measurement to prevent any bias in visual estimation of EF.
Statistical Analysis
The primary analyses of cardiac parameters were performed with the intention to treat the study population. Two treatment groups were identified within the study population, one group with patients >100 brachial systolic pressure and the other with ≤100 brachial systolic pressure All statistical analysis was done using Statistical Package For The Social Sciences (SPSS 21). The change in all cardiac parameters before and after the EECP treatment was assessed using parties sample t-test. Statistical significance was set at p<0.05.
Results
All the 72 patients tolerated the EECP treatment very well and completed the full course of 35 sessions of EECP treatment without any clinical events. The baseline characteristics of the patients are shown in the [Table/Fig-3]. Most of the patients were male (87.5%), with high prevalence of diabetes mellitus (57%) and hypertension (47%) with history of myocardial infarction present in 38% of the patients. A total of 63% of the patients had severe triple vessel disease and 25% in whom CABG had been already attempted and 44% were in symptomatic heart failure. Haemodynamic results are presented in the [Table/Fig-4].
| Demographic Parameter | Baseline Value |
|---|
| Age | 58 Yrs |
| Male | 87.5% |
| Baseline systolic pressure | 115.97±11.84 |
| Baseline heart rate | 74.96±12.17 |
| Diabetes mellitus | 41/57% |
| Hypertension | 34/47% |
| Myocardial infarction | 27/38% |
| Prior CABG | 18/25% |
| Heart failure, no | 32/44% |
| Ejection fraction % mean | 43.97±15.97 |
| Triple vessel disease | 45/63% |
CABG- Coronary artery bypass graft
The tabular column shows the pre- and post- EECP changes in both the groups assessed using the paired sample t-test.
| Parameter | PRE-EECP | POST-EECP | p-value |
|---|
| Systolic >100mmHg(N=58) | Systolic <100mmHg(N=14) | Systolic >100mmHg(N=58) | Systolic <100mmHg(N=14) | Systolic >100mmHg | Systolic <100mmHg |
|---|
| B sys. pre | 121.10 ±11.84 | 94.71 ±6.35 | 114.66 ±13.63 | 103.86 ±11.52 | 0.001 | 0.001 |
| A sys.pre | 109.48 ±10.57 | 87.86 ±5.66 | 104.36 ±11.55 | 95.64 ±11.55 | 0.001 | 0.01 |
| SHR | 75.95±12.20 | 70.86±11.58 | 73.20 ±10.57 | 68.29 ±10.95 | 0.05 | 0.17 |
| APP | 32.88 ± 8.49 | 20.64 ±6.02 | 29.52 ±9.59 | 25.93 ±7.99 | 0.008 | 0.009 |
| AP | 7.57±4.26 | 4.79 ±2.94 | 6.29 ±4.0 | 6.14 ±8.84 | 0.017 | 0.109 |
| AIX | 22.47 ±7.20 | 20.43 ±7.93 | 19.48 ±6.93 | 18.43 ±9.05 | 0.001 | 0.345 |
| EF | 46.40 ±15.88 | 33.93 ±12.39 | 50.05 ±13.20 | 45.71 ±13.43 | 0.001 | 0.001 |
Brachial Systolic Pressure and Central Pressure Components: In the first group of mean baseline brachial systolic pressure pre-EECP of 121.10±11.84 mmHg reduced to 114.66±13.63mmHg after completion of 35 sessions of EECP (p< 0.001). Similarly central aortic systolic pressure re-duced from 109.48±10.57 mmHg to 104.36±11.55mmHg (p < 0.001), heart rate reduced from 75.95±12.2 to 73.20±10.57 (p< 0.05), aortic pulse pressure reduced from 32.88±8.49mmHg to 29.52±9.59 mmHg (p< 0.008), AP reduced from 7.57±4.26mmHg to 6.29±4.0mmHg (p< 0.01) and AIx reduced from 22.47±7.20% to 19.48±6.93% (p< 0.001).
In the second group with mean baseline brachial systolic pressure pre-EECP of 94.71± 6.35 mmHg increased to 103.86±11.52 mmHg (p< 0.001), similarly central aortic systolic pressure increased from 87.86±5.66mmHg to 95.64±11.55mmHg (p< 0.01), heart rate reduced from 70.86±11.58 to 68.29±10.95 (p<0.17), aortic pulse pressure increased from 20.64±6.02mmHg to 25.93± 7.99mmHg (p> 0.009), AP increased from 4.79±2.94mmHg to 6.14±8.84mmHg (p< 0.10) and AIx reduced from 20.43±7.93% to 18.43±9.05% (p< 0.35).
Both the groups responded to EECP differentially based on their, brachial systolic pressure. When the baseline brachial blood pressure was higher with the mean of 121mmHg, EECP reduced the blood pressure both peripheral and central with similar effect on aortic pulse pressure, augmentation AP and AIx. In the second group with lower brachial systolic pressure with mean of 94mmHg, EECP seems to restore both the peripheral and central systolic pressure towards normal along with aortic pulse pressure but with no change in AP and AIx. The heart rate seems to reduce in both the groups but reached significance only in the group with normal systolic pressure.
Left Ventricular Ejection Fraction: In the first group the baseline mean LVEF increased from 46.40±15.88% pre-EECP to 50.05±13.20% post-EECP (p< 0.001), which is an 8% increase in the LV function. Similarly, in the second group the baseline mean EF was lower than the first group and it increased from 33.93±12.39% pre- EECP to 45.71±13.43% post- EECP (p< 0.001) representing an average 35% increase in the LV function.
Functional Class: In the study groups of total 72 patients undergone EECP treatment 40%/56% were treated for their CCS class angina symptoms and 32%/44% were treated for their NY-HA class heart failure symptoms. Pre-EECP 33% with angina symptoms were in the CCS Class III-IV and 78% with heart failure symptoms were in NYHA Class III-IV. All the patients improved at least one functional class post-EECP in both NYHA and CCS class.
Discussion
The clinical benefit derived from the 35 sessions of EECP treatment is due to dual mechanism of action. The central mechanism is by stimulating angiogenesis and in peripheral mechanism by decreasing the stiffness of both elastic and muscular arteries. In our current study, in patients with symptomatic CAD with LV dysfunction we evaluated the effect of EECP in a group of 72 patients with normal (n=58) and reduced systolic pressure (n=14). As expected, patients with reduced systolic pressure had a low mean baseline EF (33.93 ±12.39%) when compared to patients with normal systolic pressure with mean baseline EF of 46.40 ±15.88%.
Campbell in his paper has clearly demonstrated the differential effect of EECP in brachial blood pressure. In patients with normal systolic blood pressure and hypertensive patients EECP decreases the systolic as well as the diastolic pressure, but in patients with low systolic and diastolic pressure EECP increases both the systolic and diastolic pressure [16]. However the mechanism of why there is differential effect is not well understood.
In another study, Nichols et al., have shown that EECP treatment, in patients with refractory angina, causes a significant reduction in both peripheral and central systolic pressure along with pulse pressure. They also have shown reduction in aortic AP and AIx, both these parameter are the indices of arterial stiffness [17]. They postulated that the decrease in central and peripheral systolic pressure is due to decrease in arterial stiffness, which is mediated by the shear stress caused by increase in both diastolic and systolic blood flow by repeated inflation and deflation of the pneumatic cuffs over the lower extremities, resulting in release of nitric oxide. Both these studies confirmed the reduction of peripheral pressure and central pressure observed in CAD patients is due to reduction in arterial stiffness mediated by improvement in endothelial function. These positive changes in vascular function can significantly reduce the LV load and myocardial oxygen demand and may be the contributing factor for the improvement of clinical symptoms. This relationship between endothelial function and clinical response is shown in another study which used Reactive Hyperemia- Peripheral Artery Tonometry (RH-PAT). In this study, RH-PAT index was significantly higher only in patients who experienced decrease in angina symptoms after completion of EECP treatment [18]. This corroborates the direct relation of endothelia improvement and clinical benefit achieved in symptomatic CAD patients.
Our study confirms the similar benefit in our first group with baseline mean systolic pressure of 121.10 ±11.84mmHg. EECP reduced brachial systolic pressure by 6.44mmHg, central systolic pressure by 5.12mmHg, central pulse pressure by 3.36mmHg and arterial stiffness. There is also 8% increase in the LVEF.
The important finding in this study is in patients with low brachial systolic pressure <100mmHg and low baseline LVEF. Since EECP is expected to cause peripheral vasodilatation due to increase in nitric oxide level in plasma and improvement in vascular endothelial function there is an apprehension this may cause further reduction in systolic pressure. This will be counterproductive in patient with heart failure with compromised LV function as further reduction in systolic pressure may lead to hypotension and poor tissue perfusion. First time our study has demonstrated in this group, EECP was shown to significantly increase the central systolic pressure, pulse pressure and brachial systolic pressure. However, there is no change in arterial stiffness. AP and AIX did not show any significant reduction.
Absence of vascular response similar to that seen in the first group may be due to intra-vascular fluid overload normally observed in patients with moderate to severe LV dysfunction or may be ex-plained by maximal vasodilators prescribed due to severe ischaemic symptoms. These may be the possible explanation for absence of vascular vasodilatation response. The other possible explanation is LVEF improved to 35% in this group, whereas the increase is only 8% in the other group. This disproportionate increase in the LVEF improvement in one group when compared to other could be the reason for the differential vascular effect. The better perfused myocardium is now contracting well with more stroke volume, which may be the reason for increase in central and brachial systolic pressure. This response can be used clinically in treating patients with severe LV dysfunction associated with hypotension and advanced symptoms (NYHA III-IV), where due to hypotension standard heart failure medical regime comprising of diuretics, ACE inhibitors and beta blockers may be difficult to initiate. In this group of patients a course of EECP not only improves the blood pressure but also helps in improving myocardial contractility and thereby LVEF. This effect of EECP im-proving LVEF is already documented in CAD patients [19,20]. Our study shows how the dual mechanism of action of EECP treatment, both peripheral vascular and central angiogenic effect can be utilized in treating patients with symptomatic CAD patients with preserved LV function and normal brachial systolic pressure and in patients with LV dysfunction and low brachial systolic pressure.
Limitation
The limitations of this study are: This is a single centre open label non-randomized trial with no control or placebo group. The second group of patients with <100mmHg is a smaller group so direct comparison of both the groups was not done. Drug changes during the trial were not monitored.
Conclusion
EECP treatment has favourable effect on vascular and LV function in patients with symptomatic CAD. The treatment improves patient’s heart failure and angina symptoms with significant im-provement in LVEF and has a differential effect on the brachial and aortic systolic pressure depending on the baseline systolic blood pressure. In patient with severe LV dysfunction with hypotension the treatment is safe and can significantly increase both brachial and central aortic systolic pressure.
CABG- Coronary artery bypass graft