Leprosy is an age old disease affecting mankind with myriad clinicopathological forms. It is a chronic infectious disease caused by Mycobacterium leprae (M. leprae). The immune response of the patient and the density of the bacteria in the lesion (Bacterial Index -BI) determine the clinical manifestation and the infectivity of the disease. Accordingly the disease manifests as a spectrum beginning from lesions having low immunity and high infectivity to those having high immunity and low infectivity [1]. This clinicopathological spectrum determines the treatment regimen [2,3].
Although the incidence of leprosy is declining, it continues to prevail in practically every corner of the globe [4]. As far as tropical countries like India are considered, it is still one of the problems of public health importance [4,5]. This problem can be tackled by correct diagnosis and timely treatment. Diagnosis of leprosy is by demonstration of the lepra bacilli in slit skin smears and skin biopsies. Histopathological examination of skin remains the cornerstone in this regard [5,6]. The clinicopathological type is derived by correlation of clinical, histopathological features and the presence of lepra bacilli with assessment of the bacillary index in the lesional biopy. Bacillary index is an important determining factor in the choice of treatment regimen which is based on the clinicopathological type [5]. Ziehl-Neelsen (ZN) staining is the old and conventional method of detection of the organism in clinical specimens [7]. But the sensitivity of this stain is lower than that of Modified Fite-Faraco (FF) technique as far as its utility in tissue sections is considered. Fite-Faraco stain is the routinely used method to demonstrate Mycobacterium leprae in tissue sections [3].
We hereby undertook the study with the aim of comparing the sensitivity of fluorescent microscopy with that of ZN staining and FF technique in detecting Mycobacterium leprae in tissue sections as well as the effect on grading of the disease, if any, which may have treatment implications.
Materials and Methods
The current study is retrospective one, spanning four years, from July 2006 to June 2010 at Shri BM Patil Medical College, Bijapur including a total of sixty skin biopsies from patients clinically diagnosed as leprosy. Ethical approval was obtained from the Institutional ethical clearance committee. The disease was classified according to clinical, histopathological and modified Fite-Faraco staining results into Indeterminate (IL), Tuberculoid (TT), Borderline- Tuberculoid (BT), Mid-Borderline (BB), Borderline Lepromatous (BL) and Lepromatous Leprosy (LL) [3]. Skin biopsies received were routinely processed and after embedding in paraffin blocks, 5μ thick sections were cut. One section each were stained with Haematoxylin and Eosin (H&E) stain, FF, ZN and fluorescent stain (FL) using the standard methods.
For fluorescent staining ribbons containing four to five serial sections were taken on clean scratch free slides. No adhesives like egg albumin were used. After deparaffinisation in xylene-peanut oil, the auramine–rhodamine (HiMedia, Mumbai, India) staining was done according to the procedure of Kuper and May [15]. For each batch of sections that were stained, sections from a skin biopsy of a typical lepromatous leprosy patient and a skin biopsy from a normal individual were used as controls.
Determination of Bacteriological Index
The stained tissue sections were observed immediately under the fluorescent microscope. All sections were screened under 10X and 40X objectives. Sections showing organisms with typical morphology of Mycobacterium leprae bacilli by the 40X objective were confirmed using 100X objective. Only strongly fluorescing organisms were considered for a definitive diagnosis. Bacillary fragments were not taken into consideration. Mycobacterium leprae that appeared as solid bright yellow green fluorescing rod shaped organisms and only when interspersed with the light staining artifacts was considered diagnostic for Mycobacterium leprae [Table/Fig-1a,b]. Bacteriological index (BI) [3] was calculated under oil immersion field. According to Ridleys logarithmic scale, it is graded from zero to six +, which is based on the number of bacilli seen on an average microscopic field under 100X objective.
(a): A case of indeterminate leprosy showing brightly flourescing lepra bacilli. BI-1+ (arrows). (b): A case of tuberculoid leprosy showing a single fluorescing bacilli not detected by Fite-Faraco. BI- 1+ (arrows) (Auramine- Rhodamine, 40X).
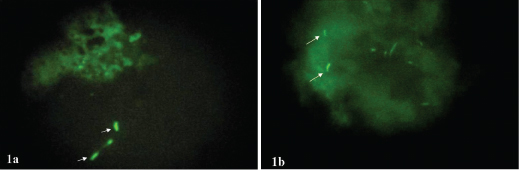
Since, BI is a continuous variable we divided the cases into two groups i.e., those with BI<3 and those with BI>3, for comparison between groups. Considering Fite-Faraco (FF) method to be the standard test, we compared its performance to that of ZN and Fluorescent (FL) methods.
Statistical Analysis
Data was analysed using SPSS v 12 (SPSS Inc. Chicago). Sensitivity, specificity, Positive Predictive Value (PPV) and Negative Predictive Values (NPV) for each tests were calculated. Chi-Square test was used to calculate significance between differences, p-value of < 0.05 was considered as statistically significant. Pearson correlation (r-value determined) was also used for comparison between groups.
Results
Fluorescent stain in Indeterminate Leprosy (IL) cases was significantly more positive than that with ZN or FF stain [Table/Fig-2].
Comparison of positivity rates of ZN (Ziehl Neelsen), Modified Fite-Faraco and fluorescent stains. IL- Indeterminate Leprosy, TT- Tuberculoid Leprosy, BT- Borderline Tuberculoid Leprosy, BB- Mid Borderline Leprosy, BL- Borderline Lperomatous Leprosy, LL- Lepromatous Leprosy.
| Histopathological Diagnosis | Total No. of Patients | ZN Stain | Modified Fite-Faraco Stain | Fluorescent Stain |
|---|
| Positivity Rate n (%) | Positivity Rate n (%) | Positivity Rate n (%) |
|---|
| IL | 30 | 1 (3.3) | 1 (3.3) | 8 (26.7) |
| TT | 2 | 0 | 0 | 0 |
| BT | 14 | 2 (14.3) | 4 (28.6) | 4 (28.6) |
| BB | 0 | 0 | 0 | 0 |
| BL | 2 | 1 (50) | 2 (100) | 2 (100) |
| LL | 12 | 12 (100) | 12 (100) | 12 (100) |
| Total | 60 | 16 (26.7) | 19 (31.7) | 26 (43.3) |
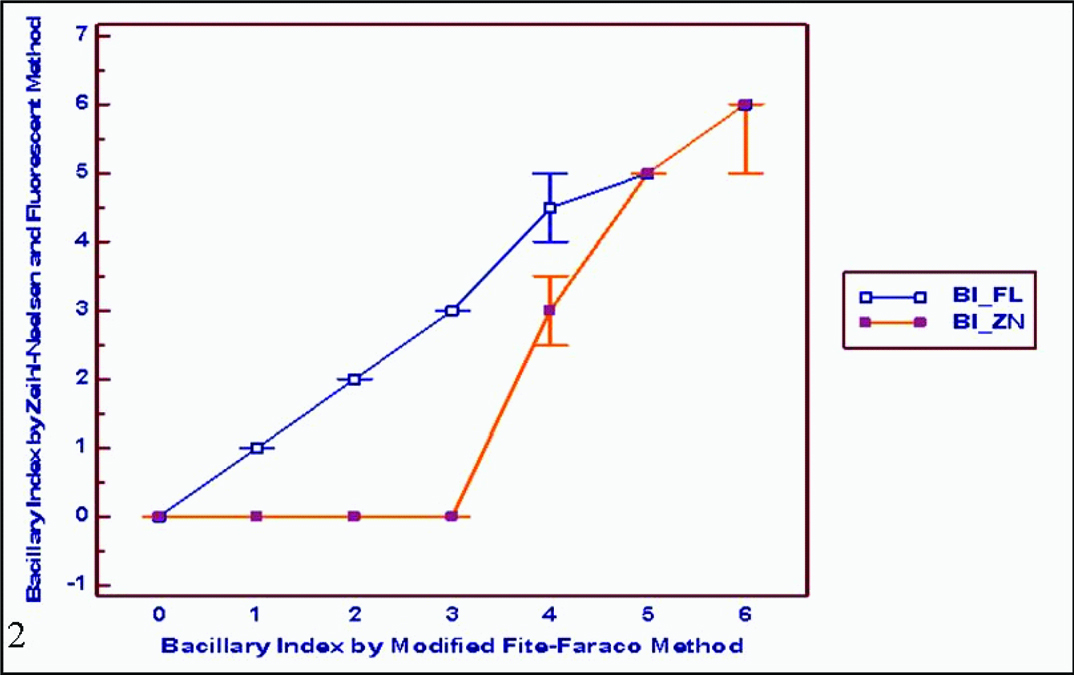
Ziehl-Neelsen method correlated well (r=0.89) with Fite-Faraco method at higher BI (>3) but poorly and insignificantly (p=0.81) with lower BI (<3). However, fluorescent method retains good (r=0.73) and statistically significant correlation (p<0.0001) even at low bacillary loads. Thus, fluorescent method is more sensitive in detecting lepra bacilli in cases with low bacillary load (BI<3) [Table/Fig-3]. FL stain showed 100% sensitivity as against ZN which showed only 75% sensitivity compared to FF method [Table/Fig-4].
Bacillary Index (BI) correlation among patients with lower BI (BI<3) and higher BI (BI>3) between the three staining types.
| Staining Method | Fite- Faraco |
|---|
| BI<3, Pearson’s ‘r’ | BI>3, Pearson’s ‘r’ |
|---|
| Ziehl-Neelsen | -0.04 | 0.89 |
| p=0.81 | p<0.0001 |
| Fluorescent Method | 0.73 | 0.84 |
| (p<0.0001) | (p=0.0004) |
Histological findings and correlation of modified Fite-faraco stain (FF) with Ziehl-Neelsen (ZN) and fluorescent stain (FL). PPV (Positive Predictive Value), NPV (Negative Predictive Value).
| Histopathological diagnosis | Sensitivity | Specificity | PPV | NPV |
|---|
| ZN Stain | FL Stain | ZN Stain | FL Stain | ZN Stain | FL Stain | ZN Stain | FL Stain |
|---|
| IL | 100 | 100 | 100 | 75.86 | 100 | 12.5 | 100 | 100 |
| TL | - | - | 100 | 100 | - | - | 100 | 100 |
| BT | 50 | 100 | 100 | 100 | 100 | 100 | 83.33 | 100 |
| BB | - | - | - | - | - | - | - | - |
| BL | 50 | 100 | - | - | 100 | 100 | 0 | - |
| LL | 100 | 100 | - | - | 100 | 100 | - | - |
| Mean | 75 | 100 | 100 | 91.95 | 100 | 78.125 | 70.8325 | 100 |
Among paucibacillary cases, FL showed a higher BI compared to FF in 9 cases, while among multibacillary cases, only 1 additional case had a higher BI compared to FF. No net additional case could be detected by ZN stain compared to FF. ZN stain showed a lesser BI compared to FF among 7 multibacillary cases [Table/Fig-5].
Cases showing upgrading of BI by fluorescent stain (FL) compared to Ziehl- Neelsen (ZN) and Modified Fite Faraco (FF) among paucibacillary and multibacillary cases.
| BI | PB Cases | MB Cases | Total |
|---|
| FL>FF | 9 | 2 | 11 |
| FL<FF | 0 | 1 | 1 |
| Net additional cases detected by FL | 9 | 1 | 10 |
| BI | PB Cases | MB Cases | Total |
| ZN>FF | 1 | 0 | 1 |
| ZN<FF | 1 | 7 | 8 |
| Net additional cases detected by FL | 0 | -7 | -7 |
Use of FL stain resulted in diagnosis of an additional case of multibacillary category while ZN stain failed to correctly classify one case of MB as diagnosed by FF stain [Table/Fig-6].
Comparison of shift in Ridley’s BI scale by Fluorescent (FL) stain and Ziehl Neelsen (ZN) stain with that of Fite Faraco (FF) method among paucibacillary and multibacillary cases.
| BI | FF PaucitoFL Multi | FF MultitoFL Pauci | NetUpgradationof Ridley’sScale | FF PaucitoZN Multi | FF MultitoZN Pauci | NetUpgradationof Ridley’sScale |
|---|
| No. of Cases | 1 | 0 | 1 | 1 | 2 | -1 |
Overall, both ZN and fluorescent method correlated significantly (p<0.0001 in both and r= 0.96 and 0.98 respectively). However, when we looked at groups with lower BI (<3) and higher BI (>3), fluorescent method retained good (r=0.73) and statistically significant correlation (p<0.0001) with FF method even at low bacillary loads; however ZN method showed poor (r=-0.03) and insignificant correlation with FF method (p=0.81) with lower BI (<3). This is similar to the observation seen with different histopathological types, where fluorescent method retained useful sensitivity even in histopathological types with lower bacillary load [Table/Fig-7].
Comparison of positivity rates of ZN staining, modified Fite-Faraco and fluorescent stain with that of other studies.
| Various Studies | ZN Stain | Fite-Faraco Procedure | Fluorescence Method |
|---|
| No. of Positive Cases | No. of Positive Cases | No. of Positive Cases |
|---|
| Present study (2016) | 26.7% (16/60) | 31.7% (19/60) | 43.3% (26/60 ) |
| Jariwala et al., [16] (1979) | - | 40.0% (20/50) | 44.0% (22/50) |
| Bhatia et al., [11] (1988) | 67.8%(57/84) | - | 89.2% (75/84) |
| Lacordaire Lopes de Faria [17] (1974) | - | 86.6% (26/30) | 33.3% (10/30) |
| Nayak SV et al., [9] (2003) | - | 44.64% (25/56) | 69.64% (39/56) |
| Manandhar U et al., [21] (2013) | NA | 25% (18/72) | NA |
| Shivamurthy V et al., [20] (2013) | NA | 23% (46/200) | NA |
| Reja AHH et al., [6] (2013) | 50.9% (84/165) | 60% (99/165) | - |
Discussion
Although being on a path of declining burden globally [4], leprosy continues to be a major public health problem in India with an annual new case detection rate of 0.84 per 10, 000 population [8]. Leprosy affects skin, peripheral nerves and other organs directly or indirectly, leading to progressive and permanent deformities in the patients. Clinical presentations are varied and much diversity exists between the clinical and histopathological features [1].
Timely and accurate diagnosis has a pivotal role to play as far as management of the disease and prevention of complications is concerned. Demonstration of the Mycobacterium leprae in the lesional skin biopsies with special stains is the method of diagnosis. For the purpose of treatment those cases with no bacilli were classified as Paucibacillary (PB) while those which showed bacilli as Multibacillary (MB) types. This classification has undergone several modifications [4].
The present study demonstrates that fluorescent staining showed a higher positivity rate in detecting the bacilli in contrast to FF which correlated with the studies done by Nayak AS et al., Bhatia et al., Jariwala et al., and Lacordaire Lopes de Faria [9,12,16,17] [Table/Fig-7].
Also, in the present study, ZN staining showed a lower positivity compared to FL. Bhatia et al., also showed more positives by fluorescent method compared to ZN stain, though they did not use FF in their study [11].
In the study done by Nayak AS et al., difference in rate of positivity between FF and FL stains were higher in tuberculoid leprosy [9], whereas, in our study the difference was higher in indeterminate leprosy. This could be because of fewer TT cases in our study, leading to insufficient sample size for statistical evaluation.
Lacordaire Lopes de Faria and Hardas et al., demonstrated that modified FF produced higher positivity rates than that with fluorescent microscopy [17,18]. In their study, they used egg albumin as adhesive and phenol which is known to produce considerable artifactual staining. However, confounding artifacts were absent in our study as well as in that done by Nayak AS et al., because adhesives were not used [9]. The solid stained bacilli were highlighted due to bright yellow-green fluorescence in contrast to pale yellow color of the artifacts.
Considering Fite-Faraco (FF) method to be the standard test, we compared the performance of ZN and FL stain methods. FL stain showed 100% sensitivity as against ZN which showed only 75% sensitivity compared to FF method [Table/Fig-4]. The apparent lower specificity of FL may in fact reflect the lower sensitivity of Fite-Faraco method which we considered as gold standard for comparison since we excluded any artifacts and non-solid bacilli as positive in our results.
However, from the present as well as other studies [9–17], it is evident that the positivity rate with FL stain was more compared to modified FF. Furthermore, the higher positivity rates with FL stain were seen in cases with lower bacillary load while the difference leveled out with lepromatous and borderline lepromatous cases (no difference) [Table/Fig-8]. Among paucibacillary cases in particular, FL stain showed a higher BI compared to FF stain. This highlights the superiority of FL, especially in cases with lower bacillary load. Added to this, with FL stain, there was a shift of paucibacillary to additional multibacillary cases in the present study. This can shift a paucibacillary case to multibacillary category, which in turn has implications in therapy, prognosis, possibility of relapse and complications [4]. Hence, FL stain has higher case pickup rate particularly among those with bacillary index less than three as proved by this study. According to Reja et al., positivity with ZN stain was better than that with FF, which was comparable with the present study [6]. The positivity rates with FF stain by Manandhar U et al., and Shivamurthy V et al., were 25% and 23% which was close to that of the present study [19,20].
Graph showing the performance of FL and ZN as compared to the standard FF. The graph highlights the higher case pickup rates of cases with lower bacillary load.
Newer diagnostic modalities like PCR for leprosy are being investigated. Difficult cases of leprosy like pure neuritic and indeterminate types can be detected and disease burden estimated by quantitative PCR [6]. However, for the more common cases, Polymerase Chain Reaction (PCR) runs the risk for being falsely positive and thus overestimating the disease burden. Additionally, PCR is a much costlier higher technology [21]. In the world, as per WHO recent reports [4], leprosy is a public health problem mainly in developing and underdeveloped countries. In these regions, where there are financial and infrastructural constraints, fluorescent based staining methods are difficult to implement, let alone PCR.
Limitation
The availability and cost of the fluorescent microscope is probably a limiting factor especially in resource poor setting. However, in areas with high disease burden, this method of diagnosis outweighs the concerns of cost. Very few studies including the current study are available on the effect of staining methods in bacteriological indexing of leprosy. More such studies using larger sample size, with possible effects on inter-observer variability are looked forward to.
Conclusion
Fluorescent microscopy can be used as a supplementary tool when tissue sections stained by modified Fite-Faraco method fail to detect the bacilli or categorize as paucibacillary cases. The current study proved the superior sensitivity of the Fluorescent method compared to Fite-Faraco or ZN staining. Additionally, paucibacillary case diagnosed on ZN/Fite-Faraco may be shifted by the fluorescent method to the multibacillary category, thus influencing the treatment and outcome. According to us, fluorescent microscopic technique is worthy diagnostic practice in every leprosy burden areas.