Polymyxins include colistin (polymyxin E) and polymyxin B (the rest being too toxic for human use) [6]. They were popular in the 1970s, after which their use declined due to the advent of better antibiotics [7]. In recent years, colistin is again attracting considerable interest as a potent antibiotic against many multi drug resistant pathogens [7].
This study examines the sensitivity patterns of colistin, polymyxin B and rifampicin against carbapenem resistant Acinetobacter baumannii. Also, it identifies the sensitivity pattern of colistin by two methods and compares the results thus obtained.
Materials and Methods
A cross-sectional pilot study was conducted that included 100 clinical isolates of carbapenem resistant A.baumannii, screened from an initial sample size of 20,282 samples received in the microbiology laboratory of PSG Hospitals, Coimbatore, Tamil Nadu, over a period of 9 months (from March 2014 to November 2014). The isolates were from specimens like tracheal aspirate, endotracheal tube tip, sputum, broncho alveolar lavage fluid, blood, ascitic fluid and wound swabs. The cases include inpatients of various wards – the Medical Intensive Care Units of General Medicine, Pulmonology, Nephrology, Gastroenterology, Neurology, wards of Neurosurgery, Trauma, Medicine, Surgery, Nephrology, Neurology, Gastroenterology and special wards. Only those A. baumannii strains that were carbapenem resistant were included in this study. Approval was obtained from the Institutional Human Ethics Committee to carry out this study.
Isolation and Identification
A.baumannii was first isolated from the specimens, after incubation at 37°C in blood agar and MacConkey agar, after microscopy (Gram negative cocco bacilli on Gram staining). They were then confirmed by biochemical tests [9] – catalase positive, oxidase negative, carbohydrate fermentation (glucose fermenter, lactose fermenter, sucrose non-fermenter, mannitol non-fermenter and non motile), citrate positive, urease positive and growth at 44°C positive.
The carbapenemase producing (carbapenem resistant) strains were then identified by Kirby-Bauer disk diffusion method [10], using meropenem disks of 10mcg, according to the norms of CLSI guidelines [11].
Antimicrobial Sensitivity Testing
These strains were then inoculated on the surface of a Mueller-Hinton agar plate from a fresh preparation of 0.5 McFarland suspension. Three antibiotic disks namely colistin (10mcg), polymyxin B (300 U) and rifampicin (5mcg), purchased from HiMedia Laboratories, Mumbai, were incorporated into the media. After incubation for 24 hours, by Kirby-Bauer disk diffusion method [10], the zone diameters were measured and categorized as sensitive, intermediate sensitive or resistant based on the breakpoints obtained from various sources [Table/Fig-1] [12–15].
Susceptibility of all 3 Antibiotics by disk diffusion – breakpoints / criteria [12–15].
| Zone Diameters of | Sensitive | Intermediate Sensitive | Resistant |
|---|
| Colistin* | ≥14 mm | 13 mm | ≤12 mm |
| Polymyxin B† | ≥12 mm | - | ≤11 mm |
| Rifampicin‡ | ≥20 mm | 17-19 mm | ≤16 mm |
* Galani et al [12] † Andrea L Kwa, Vincent H Tam, Matthew E Falagas [13] considering the breakpoints of Pseudomonas aeruginosa as Thomson JM, Bonomo RA [14] find resistance patterns between the two organisms similar. ‡ Thapa B, Tribuddharat C, Rugdeekha S, Techachaiwiwat W, Srifuengfung S, Dhiraputra C [15] considering the breakpoints of Staphlococcus aureus
Simultaneously, a suspension of 2-3 colonies from a pure culture was made (145μL) of 0.5 McFarland standard to which 3 mL of saline was added, followed by broth dilution. This was then subjected to VITEK 2C (bioMérieux) automated microbial identification system, making use of a Gram negative susceptibility card AST-N281. The MIC values for colistin were observed according to the breakpoints [Table/Fig-2] [11,12]. The above mentioned antibiotic susceptibility tests were all done according to the CLSI guidelines [11].
Colistin susceptibility criteria / breakpoints [11,12].
| Parameter | Sensitive | Intermediate Sensitive | Resistant |
|---|
| Zone Diameters by Kirby-Bauer’s Disk Diffusionmethod § | ≥14 mm | 13 mm | ≤12 mm |
| MIC values by VITEKautomated systems|| | ≤2 μg/mL | - | ≥4 μg/mL |
§ Galani et al., [12] ||according to CLSI guidelines 2015 [11]
Results
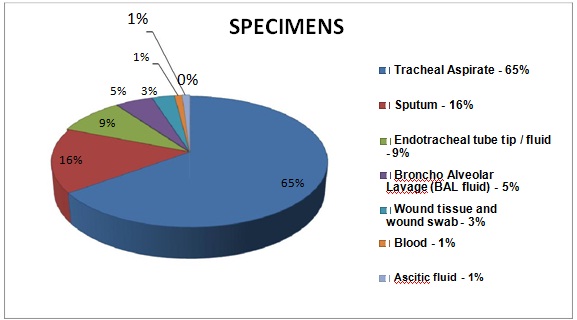
A 95% of the carbapenem resistant A.baumannii were from respiratory samples and the maximum number were isolated from tracheal aspirates (65%) [Table/Fig-3]. Majority of the samples were from Medical Intensive Care Units, Trauma and Neurosurgery wards following close behind [Table/Fig-4].
Ward wise distribution of samples.
 |
| Ward Wise Distribution of Samples | Number of Clinical Samples (n) |
| MICU – Medical Intensive Care Unit - Medicine | 26 |
| MICU – Pulmonology | 6 |
| MICU – Neurology | 2 |
| MICU – Gastroenterology | 4 |
| MICU - Nephrology | 2 |
| Neurosurgery/ Trauma ward | 16 |
| Medicine and Surgery wards | 4 |
| Pulmonology ward | 8 |
| Neurology ward | 4 |
| Gastroenterology ward | 4 |
| Miscellaneous (namely special wards) | 24 |
*MICU – Medical Intensive Care Unit
Numerical values
Following the breakpoints [Table/Fig-1], it was found that 78% of the organisms were sensitive to colistin and all of them (100%) were sensitive to polymyxin B. rifampicin resistance was seen in 80% of the organisms [Table/Fig-5].
Susceptibility of all 3 antibiotics by disk diffusion.
| No. of isolates | Sensitive | Intermediate Sensitive | Resistant |
|---|
| Colistin* | 78 | 12 | 10 |
| Polymyxin B† | 100 | - | - |
| Rifampicin‡ | 12 | 8 | 80 |
VITEK reported 99% to be sensitive, whereas only 78% turned out to be sensitive by disk diffusion method [Table/Fig-6].
Colistin susceptibilty observed by both methods.
| No. of Isolates | Sensitive | Intermediate Sensitive | Resistant |
|---|
| By Kirby-Bauer’s DiskDiffusion method§ | 78 | 12 | 10 |
| MIC Determination by VITEK automated systems|| | 99 | - | 1 |
The comparison of sensitivity and resistance to colistin by the two methods – Kirby-Bauer disk diffusion method and MIC determination by VITEK shows that 90 isolates were sensitive to colistin by both the methods [Table/Fig-7].
Comparison of results by disk diffusion and MIC determination by VITEK for colistin.
 No. of isolates sensitive to colistin by both methods
No. of isolates sensitive to colistin by both methods
 No. of isolates sensitive to colistin by disk diffusion but resistant by VITEK
No. of isolates sensitive to colistin by disk diffusion but resistant by VITEK
 No. of isolates sensitive to colistin by VITEK but resistant by disk diffusion
No. of isolates sensitive to colistin by VITEK but resistant by disk diffusion
 No. of isolates resistant to colistin by both methods
No. of isolates resistant to colistin by both methods

However, the 9 isolates that showed resistance by disk diffusion and sensitivity by VITEK had zone diameters in the range of 10-12 mm in the disk diffusion method. (≤12 mm – colistin resistant).
Discussion
A.baumannii, commonly known for causing ventilator associated pneumonia, blood stream infections, urinary tract infections and wound infections, is becoming difficult to treat with the limited options available. Also, it is one of the most easily transmitted pathogen by health care workers [16]. Carbapenem resistance is being increasingly reported, necessitating their detection at the earliest [17].
This study observed a remarkably high susceptibility rate of carbapenem resistant A.baumannii isolates to polymyxins. Hence, we infer that older antibiotics like colistin and polymyxin B could be used to treat multidrug resistant infections.
A study in New Delhi, India showed 96.4% sensitivity to polymyxin B in A.baumannii [18]. Another Indian study showed 100% sensitivity to both colistin and polymyxin B [19]. Only 1.6% of the A.baumannii isolates showed resistance to colistin and polymyxin B in a study based in Korea [20]. Studies from the west and Middle East also observed a similar trend, a 100% susceptibility to colistin and polymyxin B was reported [21].
However, invitro activities of colistin vary from its invivo actions [4,22]. Clinical efficacy of colistin has limited data because of low number of patients, study design, varying dosage regimens, differences in susceptibility testing methods and inaccurate pharmacokinetic knowledge of colistimethate sodium [23,24]. A study of the West shows 57-77% cure rate with colistin in patients with A.baumannii infections [24]. Another study from Greece reports 93% cure rate with colistin in MDR A. baumannii CNS infections [25].
This study showed a high rate of rifampicin resistance by carbapenem resistant A.baumannii. Similarly, a study from Spain showed development of high level of rifampicin resistance Invitro when used in monotherapy against carbapenem resistant A. baumannii [26].
There is an increased need for reliable susceptibility testing methods to predict the clinical response. Our study showed a slight discrepancy in the results obtained by disk diffusion and automated VITEK. It shows that there could be chances of missing out on colistin sensitive organisms in smaller set ups where only disk diffusion is carried out (since the automated VITEK is considered superior) [27]. The lack of consensus regarding breakpoints for resistance between Clinical and Laboratory Standard Institute (CLSI), British Society for Antimicrobial Chemotherapy (BSAC) and Société Française de Microbiologie (SFM) has resulted in varied interpretation of the sensitivity patterns [18]. This also brings about lacunae in studies done on this group of antimicrobials. A study based in Boston calls for the need to confirm disk diffusion methods of colistin by determining MIC values to eliminate falsely susceptible and resistant organisms [27]. A European based study emphasizes on the reliability of VITEK automated method results over the routine disk diffusion method in antibiotic susceptibility testing [28].
Then arises the question of efficacy of monotherapy. As the drug concentration of colistin decreases in the body, there is re-growth of the surviving bacteria with a higher MIC [29]. Combination therapy with rifampicin for both colistin and polymyxin B has given much better results than monotherapy [4,30].
Limitation
The invivo action of polymyxins cannot be expected to be the same as the invitro activities thus seen, because of their pharmacokinetics. More studies in the future will be required to establish the same.
Conclusion
This study shows a good number of carbapenem resistant A.baumannii to be sensitive to polymyxins – namely colistin and polymyxin B. Hence, they ought to be considered as the next drug of choice for carbapenem resistant strains. The study also shows rifampicin resistance to be high and hence is not advised for monotherapy. There are also substantial differences between the two methods of antibiotic susceptibility tests – disk diffusion and MIC determination by VITEK automated systems.
*MICU – Medical Intensive Care Unit
Numerical values