Cystic Dystrophy and Heterotopic Pancreas (CDHP) is a rare cause of chronic pancreatitis, duodenal obstruction and obstructive jaundice. The diagnosis is often delayed and requires multimodal imaging with Computed Tomography (CT), Magnetic Resonance Cholangio-Pancreatography (MRCP) and Endoscopic Ultrasound (EUS). The treatment is not standardized but Pancreatoduodenectomy (PD) provides long-term relief in some cases. The present case is about a 47-year-old male with history of alcohol abuse for 10 years who presented with a history of recurrent acute pancreatitis. CT scan revealed bulky head of pancreas with a hypodense area between pancreatic head and second part of duodenum, prominent pancreatic duct, Dilated Common Bile Duct (CBD) (15mm) and stenosis of second part of duodenum. EUS confirmed CT findings. A Fine Needle Aspiration Cytology (FNAC) showed only inflammatory infiltrate. A diagnosis of groove pancreatitis was made. The patient was subjected to a pylorus preserving PD. Histopathological examination showed cystic dystrophy of duodenum due to heterotopic pancreas. The patient is doing well at 5 years of follow-up.
Case Report
A 47-year-old male with history of alcohol abuse for 8 to 10 years visited the Department of Gastrointestinal Surgery for recurrent attacks of mild acute pancreatitis since 3 years. He also had recurrent epigastric pain, significant weight loss, early satiety and postprandial fullness. Multiple ultrasonography examinations and Computed Tomography (CT) scans of abdomen done in the past suggested chronic pancreatitis for which patient was advised pancreatic enzyme supplementation, alcohol abstinence, analgesics and dietary changes by different physicians, only to recur again.
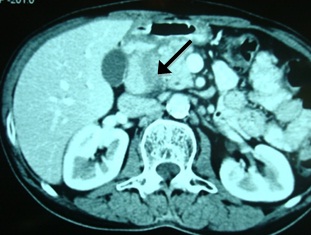
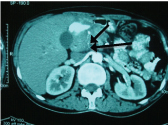
CT scan revealed bulky head and uncinate process of pancreas with a hypodense area between pancreatic head and second part of duodenum [Table/Fig-1], prominent pancreatic duct, dilated Common Bile Duct (CBD) (15mm), hugely distended gall bladder, irregular wall thickening of first and second part of duodenum with stenosis of second part of duodenum [Table/Fig-2]. Upper gastrointestinal endoscopy did not reveal significant duodenal obstruction. Duodenal biopsy showed Brunner’s gland hyperplasia. Endoscopic Ultrasound (EUS) confirmed the CT findings. An Fine Needle Aspiration Cytology (FNAC) showed only inflammatory infiltrate and no malignancy. Blood level of CA19-9 was normal. Liver function tests were normal.
Intravenous contrast enhanced CT scan showing hypodense area (black arrow) between pancreatic head & second part of duodenum.
Intravenous contrast enhanced CT scan showing duodenal stenosis (black arrow).
A diagnosis of groove pancreatitis was made. In view of inability to exclude a neoplasm with certainty, the patient was subjected to a pylorus preserving Pancreatoduodenectomy (PD). During surgery a hard mass in the head and the uncinate process of pancreas was felt. There was no pancreatic calcification. A dilated CBD and a fibrotic partially constricting ring in the midportion of second part of the duodenum were noted.
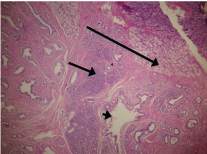
Histopathological examination [Table/Fig-3] revealed islands of heterotopic pancreatic tissue in the muscularis propria of the duodenum. The lobules comprised of unremarkable pancreatic acini, ducts and few clusters of islet cells. The acini were atrophic in places and few aggregates of ductules were seen. Cystic dilatation of ductules was seen in the duodenal wall with mild to moderate inflammatory infiltrate. Extensive brunner gland hyperplasia was seen in mucosa and submucosa of the duodenum leading to widening of submucosa. The duodenal muscle was markedly hypertrophied, especially in its medial wall, which contained the heterotopic pancreatic tissue. Both these features probably led to narrowing of the duodenal lumen. The head of pancreas showed focal evidence of chronic pancreatitis. The postoperative course was uneventful. The patient is asymptomatic five years after the surgery.
Low power microphotograph showing Brunner’s gland hyperplasia (long arrow) with heterotopic pancreas (short arrow) and dilated ductule (arrowhead) in duodenal wall. (Haematoxyline and Eosin stain, original magnification X40).
Discussion
Cystic Dystrophy and Heterotopic Pancreas (CDHP) is characte-rized by formation of cysts in the submucosal and muscular layer of the duodenal wall, with inflammation and fibrosis and intermingled heterotopic pancreatic tissue, first described by Potet and Duclert [1,2].
Ectopically located pancreatic tissue with a complete anatomic and vascular disconnection from main gland has been reported in different series [1]. The most commonly reported site is the duodenum, followed by the stomach, jejunum and rest of the gastrointestinal tract [3]. However, CDHP has been reported in approximately 200 cases only worldwide in English literature till date. Most reported cases describe Caucasian patients from western hemisphere. Reports from south Asia are rare [4].
Cystic dystrophy is a complication of heterotopic pancreas, which is situated in the medial border of the second part of duodenumin close proximity of the papilla [5]. Location within the duodenal wall, complete disconnection from pancreas proper and presence of an epithelial lining distinguishes it from pseudocyst [6].
Groove pancreatitis, myoadenomatosis, paraduodenal wall cyst, pancreatic hamartoma of duodenum and CDHP have similar clinical picture. Image findings and histopathology are grouped together by Adsay and Zamboni under paraduodenal pancreatitis [1,2].
The pathogenesis of CDHP is unclear. It is predominant in Caucasian males especially in their fifth decade of life and amongst alcoholics even though cases with the disease in female non-alcoholics and incidental detection too are reported [5]. CDHP could be an extension of existing chronic pancreatitis secondary to a heterotopic pancreas [1,7] or chronic pancreatitis could evolve in the ectopic gland since the excretion ducts of the ectopic gland are too small and develop recurrent obstructive acute pancreatitis, retention cysts and gradually chronic pancreatitis [6,7]. This could explain the disease in non-alcoholic patients. The rest of the pancreas may be healthy or have chronic pancreatitis.
Patients usually are asymptomatic with incidental detection. They can present with epigastric pain, postprandial fullness, vomiting, weight loss, obstructive jaundice, recurrent acute pancreatitis, palpable abdominal mass or chronic pancreatitis in the early part of disease. A case report has also been published on a patient presenting with upper gastrointestinal bleeding [7]. It is important to consider CDHP early in the differential diagnosis of patients with recurrent pancreatic pain with or without evidence of chronic pancreatitis and duodenal obstruction. The differential diagnosis includes groove pancreatic adenocarcinoma, duodenal adenocarcinoma at the medial wall of duodenum, ampullary adenocarcinoma, duodenal gastrointestinal stromal tumour, carcinoid, duodenal duplication and recurrent acute pancreatitis involving the pancreaticoduodenal groove. Diagnosis is often delayed. Patients can have mildly elevated CA 19.9 [1,2,7].
Diagnosis relies on Multidetector Computed Tomography (MDCT), Magnetic Resonance Cholangio-Pancreatography (MRCP) and Endoscopic Ultrasound (EUS). MDCT shows multiple cysts in the thickened wall of the duodenum with post contrast enhancement. The thickness of duodenal wall often correlates to the presence of heterotopic pancreas [2,8]. Duodenal dystrophy may appear solid if cysts are small. MRCP additionally gives information about biliary and pancreatic ducts [2,9].
EUS is the most useful diagnostic examination [10]. Three specific components in the duodenal wall dystrophy: wall thickening, presence of cysts and a network of channels around the cysts are picked up by EUS [6]. Intraluminal 20 Mhz mini probe sonography is more informative in cases of luminal stenosis [11]. Absence of gross cysts that can be seen on imaging often delays the diagnosis emphasizing the need for high degree of clinical suspicion. The indexed case was one such, where gross cysts were absent on preoperative imaging studies.
Conservative treatment is the mainstay in acute setting if confirmatory diagnosis is established. Treatment of CDHP is debated because pathology is benign, medical management and endoscopic therapy is helpful anecdotally, pancreatic resection carries high morbidity and mortality and pathology is diagnosed often postoperatively [12].
When diagnosed preoperatively, somatostatin analogues along with parenteral nutrition, alcohol abstinence and analgesics have been used successfully with a variable effect on pain, reduction in cyst size and weight gain [5]. Endoscopic cyst fenestration and endoscopic minor papilla drainage has shown good results in selected cases like voluminous cysts, few cysts and superficial cysts [13]. However, recurrence is higher because cysts are numerous, small and often deep [5,10,13]. Indications for surgery include severe symptoms at primary presentation, failure of medical and/or endoscopic treatment and suspicion of malignancy [14].
Biliary and gastrointestinal bypass, cystogastrostomy and pancreas preserving duodenectomy have been used in case reports with a prospective diagnosis of CDHP and are useful for patients without pain and malignancy [6]. However, flare-ups of pancreatic pain or adenocarcinoma can occur. Adenocarcinoma with CDHP may not get diagnosed with imaging or EUS raising doubts on the safety of conservative treatments. All these problems could be tackled with long-term relief by PD [14,15].
Conclusion
CDHP is a rare cause of chronic pancreatitis and needs high degree of clinical suspicion combined with CT, MRCP and EUS for diagnosis. Exact indication and long term efficacy of medical and endoscopic therapy is unclear. However, PD gives long-term relief from a potentially malignant problem.
Conflict of Interest
All the authors listed declare that there are no conflicts of interest and that no financial sponsorship was accepted in producing and presenting this article. Each of the authors listed is in agreement with the content of the paper.
[1]. Aly A, Saied T, Cystic dystrophy of the duodenal wall in heterotopic pancreas: A case reportThe Egyptian Journal of Radiology and Nuclear Medicine 2014 45(2):295-98. [Google Scholar]
[2]. Arora A, Dev A, Mukund A, Paraduodenal pancreatitisClin Radiol 2014 69(3):299-306. [Google Scholar]
[3]. Doland RV, ReMine WH, Dockerty MB, The fate of heterotopic pancreatic tissue. A study on 212 casesArch Surg 1974 109:762-65. [Google Scholar]
[4]. Rebours V, Lévy P, Vullierme MP, Couvelard A, O’Toole D, Ruszniewski P, Clinical and morphological features of duodenal cystic dystrophy in heterotopic pancreasAm J Gastroenterol 2007 102(4):871-79. [Google Scholar]
[5]. Di Serafino M, Severino R, Brigida R, Scarano E, Cystic dystrophy of the duodenal wall in heterotopic pancreas: an atypical case in non-alcoholic femaleBJR Case Reports 2016 2016:0058 [Google Scholar]
[6]. Flejou JF, Potet F, Molas G, Bernades P, Amouyal P, Fekete F, Cystic dystrophy of the gastric and duodenal wall developing in heterotopic pancreas: an unrecognized entityGut 1993 34:343-47. [Google Scholar]
[7]. Brotons A, Gastrointestinal Hemorrhage secondary to duodenal cystic dystrophy in heterotopic pancreasGastroenterology Research 2011 4(1):30-33. [Google Scholar]
[8]. Raman SP, Salaria SN, Hruban RH, Fishman EK, Groove pancreatitis: spectrum of imaging findings and radiology-pathology correlationAJR Am J Roentgenol 2013 201:W29-W39. [Google Scholar]
[9]. Kalb B, Martin DR, Sarmiento JM, Paraduodenal pancreatitis: clinical performance of MR imaging in distinguishing from carcinomaRadiology 2013 269(2):475-81. [Google Scholar]
[10]. Jouannaud V, Coutarel P, Tossou H, Butel J, Vitte RL, Skinazi F, Cystic dystrophy of the duodenal wall associated with chronic alcoholic pancreatitis. Clinical features, diagnostic procedures and therapeutic management in a retrospective multicenter series of 23 patientsGastroenterol Clin Biol 2006 30:580-86. [Google Scholar]
[11]. Silina T, EUS-Diagnostics of Cystic duodenal dystrophy, case reportEndoscopy 2011 43(S 03) [Google Scholar]
[12]. Basili E, Allemand I, Ville E, Laugier R, Lanreotide acetate may cure cystic dystrophy in heterotopic pancreas of the duodenal wallGastroenterol Clin Biol 2001 25(12):1108-11. [Google Scholar]
[13]. Beaulieu S, Vitte RL, Le Corguille M, Petit Jean B, Eugene C, Endoscopic drainage of cystic dystrophy of the duodenal wall: report of three casesGastroenterol Clin Biol 2004 28:1159-64. [Google Scholar]
[14]. Casetti L, Bassi C, Salvia R, “Paraduodenal” pancreatitis:results of surgery on 58 consecutives patients from a single institutionWorld J Surg 2009 33:2664-69. [Google Scholar]
[15]. Egorov VI, Butkevich AC, Sazhin AV, Pancreas-preserving duodenal resections with bile and pancreatic duct replantation for duodenal dystrophy. Two case reportsJOP 2010 11:446-52. [Google Scholar]