Ulcer is a major disease of gastrointestinal system which affects 10% of the world population with different aetiologies. Chronic alcohol intake, smoking, excessive stress, chronic usage of non-steroidal anti-inflammatory drugs and H.pylori bacterial infection are the crucial causes of peptic ulcer characterized by inflammation, mucosal bleeding and abdominal pain in patients [1,2]. These ulcers can develop when the imbalance occurs between the gastroprotectives (mucus, bicarbonate and prostaglandins) and aggressives (acid, pepsin, bile salts and Helicobacter pylori bacteria) [3]. The recent approach to peptic ulcer is managed by inhibition of gastric acid secretion, promotion of gastro-protection, blocking apoptosis and stimulation of epithelial cell proliferation for effective healing. The conventional drugs used in the treatment of ulcer include histamine receptor antagonists, prostaglandins analogues, proton pump inhibitors, cytoprotective agents, antacids and anticholinergics, but most of these drugs produce undesirable side effects or drug interactions and may even alter biochemical mechanisms of the body upon chronic usage. Hence, herbal medicines are generally used in such chronic cases, wherein drugs are required to be used for long periods [4].
Materials and Methods
In-vivo antiulcer study was performed from October 2015 to November 2015 and the ethical approval was permitted from Sri Sivani College of Pharmacy, Srikakulam, Andhra Pradesh, India.
Drugs and Chemicals
Ethanol (Bengal Chemicals, Kolkatta, India), Cysteamine hydrochloride (Hi-media, Mumbai, India) and Ranitidine (Kopran Pharma Ltd., Mumbai, India) were obtained. All other chemicals and solvents used in this study were of analytical grade.
Plant material
The fresh leaves of S.indica were collected in the month of July 2015 from the local areas of Srikakulam, Andhra Pradesh, India. The plant was identified and authenticated by Dr. K. M. Chetty, Sri Venkateswara University, Tirupathi, where the voucher specimen was deposited for further reference.
Preparation of Plant Extract
The leaves of S.indica were washed thoroughly in water to remove foreign matter and allowed to shade dry with a relative humidity of 40–45%. Then, the leaves were powdered in roller grinder and passed through a sieve (No. 40). Then, the fine powder (Approx. 150 gm) was defatted with petroleum ether and extracted with 1 litre of 95% ethanol at room temperature by using Soxhlet apparatus for 72 hour. The resultant extract was filtered and concentrated in a rotary evaporator under reduced pressure to obtain a thick semi solid brown paste, which was stored at −20°C until required. The yield of the extract was found to be 11.87 %w/w.
Phytochemical Screening
The preliminary phytochemical screening was performed with the ethanolic extract of S. indica leaves (ESIL) for the detection of various phytochemicals [10].
Experimental Animals
Adult albino rats (40 numbers) of 130-160g of body weight of either sex were procured from the animal house of Sri Sivani College of Pharmacy, Srikakulam, Andhra Pradesh, India. The study protocol was approved from the Institutional Animal Ethics Committee (IAEC) under the reference no. 1427/PO/a/11/CPCSEA and CPCSEA guidelines were adhered during the maintenance and experiment. All animals were maintained under standard husbandry conditions with food and water ad libitum.
Acute Oral Toxicity Study
Healthy adult male albino rats were fasted overnight prior to the experiment. Different doses (50-2000 mg/kg, P.O) of the ethanolic extract of S. indica were administered to each group of rats (Each group carries 8 rats) and they were observed continuously for 1 hour and then at half-hourly intervals for 4 hour, for any gross behavioural changes and further up to 72 hour, followed 14 days for any mortality as per the OECD (Organization for Economic Co-operation and Development) Guideline 425 [11]. The leaf extract of S. indica was found to be non-toxic up to the maximum dose of 2000 mg/kg body weight. Dose selected for antiulcer evaluation was 150, 300 and 600 mg/kg respectively [11].
Animal Grouping and Treatment Schedule for Antiulcer Study
In all the experimental models, male albino rats were selected and divided into five groups of six animals each. Animals were fasted for 24 hour before the study, but had free access to water. Group I treated as vehicle control, received only distilled water; group II, III and IV treated as treatment groups, received the graded dose of ethanol extract of S. indica (ESIL) at 150, 300 and 600 mg/kg, (P.O.) for 7 days (once in a day) respectively and group V as standard group, received ranitidine 50 mg/kg (P.O.) [Table/Fig-1].
Groups of experimental animals for antiulcer study.
| S. N. | Groups | Sample Size |
|---|
| 1 | Vehicle control (with water) | 6 |
| 2 | Ethanolic extract of S.indica (ESIL) at 150 mg/kg, | 6 |
| 3 | Ethanolic extract of S.indica (ESIL) at 300 mg/kg | 6 |
| 4 | Ethanolic extract of S.indica (ESIL) at 600 mg/kg | 6 |
| 5 | Standard group ranitidine 50 mg/kg (p.o.). | 6 |
Pyloric Ligation Induced Gastric Ulceration
In this method, albino rats were fasted in individual cages for 24 hour ESIL, reference drug and control vehicle was administered 1 hour prior to pyloric ligation. Then the pre-treated animals were anaesthetised by anaesthetic ether; the abdomen was opened by a small midline incision below the xiphoid process. The pyloric portion of the stomach was ligated without causing any damage to its blood vessels. The stomach was isolated carefully and the abdominal wall was sealed by interrupted sutures. The animals were deprived of water during the postoperative period. Four hours after ligation, the stomach was dissected out and contents were collected into clean tubes. The volume, pH and total acid content of gastric juice were determined. The contents were centrifuged, filtered and subjected to titration for estimation of total acidity. From the supernatant, aliquots (1 ml each) were taken for the determination of pH, total or free acidity and pepsin activity [12]. Each stomach was examined for lesions in the fore stomach portion and indexed according to severity [13].
The numbers of ulcers were counted and scoring of ulcer was made as follows: Normal colored stomach (0), Red coloration (0.5), Spot ulcer (1), Haemorrhagic streak (1.5), Deep ulcers (2) and Perforation (3). Mean ulcer score for each animal was expressed as ulcer index [14]. Ulcer index (UI) was measured by using following formula: UI = UN + US + UP × 10−1 Where, UI (Ulcer Index); UN (Average number of ulcers per animal); US (Average number of severity score); UP (Percentage of animals with ulcers). The percentage inhibition of ulceration was calculated and compared with control.
Ethanol Induced Mucosal Damage in Rats
The rats were fasted for 24 hours before the experiment. After 1 hour of administration of ESIL, ranitidine and vehicle control treatment, 1ml of absolute ethanol (0.5 ml/100g) was orally administered to each rat of every group. After 1 hour, the animals were sacrificed with excess of anaesthetic ether and stomach was opened along the greater curvature, cleared of residual matter with saline and the inner surface was examined for severity of ulceration [15]. Ulcer index and % ulcer protection were calculated.
Cysteamine Induced Duodenal Ulceration
Cysteamine HCl (400 mg/kg, P.O. in 10% aqueous solution) was administered twice in a day at an interval of 4 hour to produce duodenal ulcers in rats. The S.indica or reference drug or control vehicle was administered 30 min before each dose of cysteamine HCl. After 20 hours of last dose of cysteamine, the animals were sacrificed and the duodenum was isolated carefully and opened the portion of the antimesenteric side. The ulcer score was obtained by measuring the dimensions of the duodenal ulcer(s) in square millimeters and ulcer index was determined using the method described earlier [12].
Statistical Analysis
The results were expressed as the Mean±SD for each group. Statistical differences were evaluated using a one-way analysis of variance (ANOVA) followed by Dunnet’s t-test. Results were considered to be statistically significant at p<0.05.
Results
Preliminary Phytochemical Screening
The ESIL showed the presence of flavonoids, alkaloids, phenolics, terpenoids, tannins, saponins, carbohydrates and amino acids.
Effect of the ESIL on Pylorus Ligation Induced Gastric Ulceration
In pylorus ligated rats, the volume of gastric content, pH, pepsin activity, total and free acidity are shown in [Table/Fig-2]. In ESIL treated groups, the volume of acid secretion, total acidity and pepsin activity was decreased and pH of the gastric juice was increased compared to ulcer control group. The effects of ethanolic extract of S. indica on acid parameters showed significant (p<0.01and p<0.05) effect at 150, 300 and 600 mg/kg doses compared to ulcer control animals. In addition, S. indica showed a dose dependent curative ratio compared to ulcer control groups. The extracts exhibited an inhibition percentage of 37.65, 45.88 and 67.48 at doses of 150, 300 and 600 mg/kg doses respectively; and, the standard drug, ranitidine, exhibited an inhibition percentage of 75.10. ESIL at 600 mg/kg was shown equipotent antiulcer activity with ranitidine.
Effect of ESIL on gastric parameters by pyloric ligation induced ulceration in rats.
| Treatment | GastricContent (ml) | pH | Acidity (mEq/lt) | Pepsin activity(Per ml/h) |
|---|
| Total | Free |
|---|
| Vehicle Control | 10.6 ±0.34 | 3.41±0.12 | 121.3±1.30 | 97.3±0.52 | 3.75±0.15 |
| ESIL 150 mg/kg | 4.26±0.62* | 4.80±0.11* | 71.16±1.42* | 64.39±1.27 | 3.42±0.90* |
| ESIL 300 mg/kg | 4.86±0.20* | 5.08±0.15* | 63.13±1.60* | 57.7±0.16* | 3.17±0.22* |
| ESIL 600 mg/kg | 5.53±0.15** | 5.62±0.08** | 54.17±0.54** | 47.5±0.05* | 2.93±0.25* |
| Ranit. 150 mg/kg | 5.68±0.18** | 5.86±0.18** | 45.2±0.54** | 39.2±0.04** | 2.84±0.77** |
Values are expressed as Mean ± SEM, n= 6, *p < 0.05 and ** p < 0.01 when compared with vehicle control group. (Statistically analysed by one-way ANOVA followed by Dunnet’s t-test).
Effect of the ESIL on Ethanol Induced Gastric Ulceration
The animals pre-treated with ESIL groups showed significant (p<0.01) as well as dose dependent inhibition of ulcer index as compared to control group [Table/Fig-3]. ESIL showed 41.76, 50.06 and 61.30 % ulceration inhibition at 150, 300 and 600 mg/kg respectively whereas ranitidine showed 69.47% ulceration inhibition [Table/Fig-4].
Effect of ESIL on ulcer index by pyloric ligation, ethanol and cysteamine induced ulcers in rats.
| Treatment | Ulcer index |
|---|
| Pyloric ligation | Ethanol | Cysteamine |
|---|
| Vehicle Control | 4.86±0.21 | 7.83 ±0.46 | 4.32 ±0.14 |
| ESIL 150 mg/kg | 3.03±0.54* | 4.56±1.08* | 2.88±0.98* |
| ESIL 300 mg/kg | 2.63±0.74* | 3.91±0.15* | 2.53±0.42* |
| ESIL 600 mg/kg | 1.58±0.63** | 3.03±0.36** | 1.49±0.87** |
| Ranitidine 150mg/kg | 1.21±0.43** | 2.39±0.08** | 1.38±0.21** |
Values are expressed as Mean ± SEM, n= 6, *p < 0.05 and ** p < 0.01 when compared with vehicle control group. (Statistically analysed by one-way ANOVA followed by Dunnet’s t-test.).
Effect of ESIL on percentage of ulcer inhibition by pyloric ligation, ethanol and cysteamine induced ulcers in rats.
Values are expressed as Mean ± SEM, n= 6, *p < 0.05 and ** p < 0.01 when compared with control group. (Statistically analysed by one-way ANOVA followed by Dunnet’s t-test).
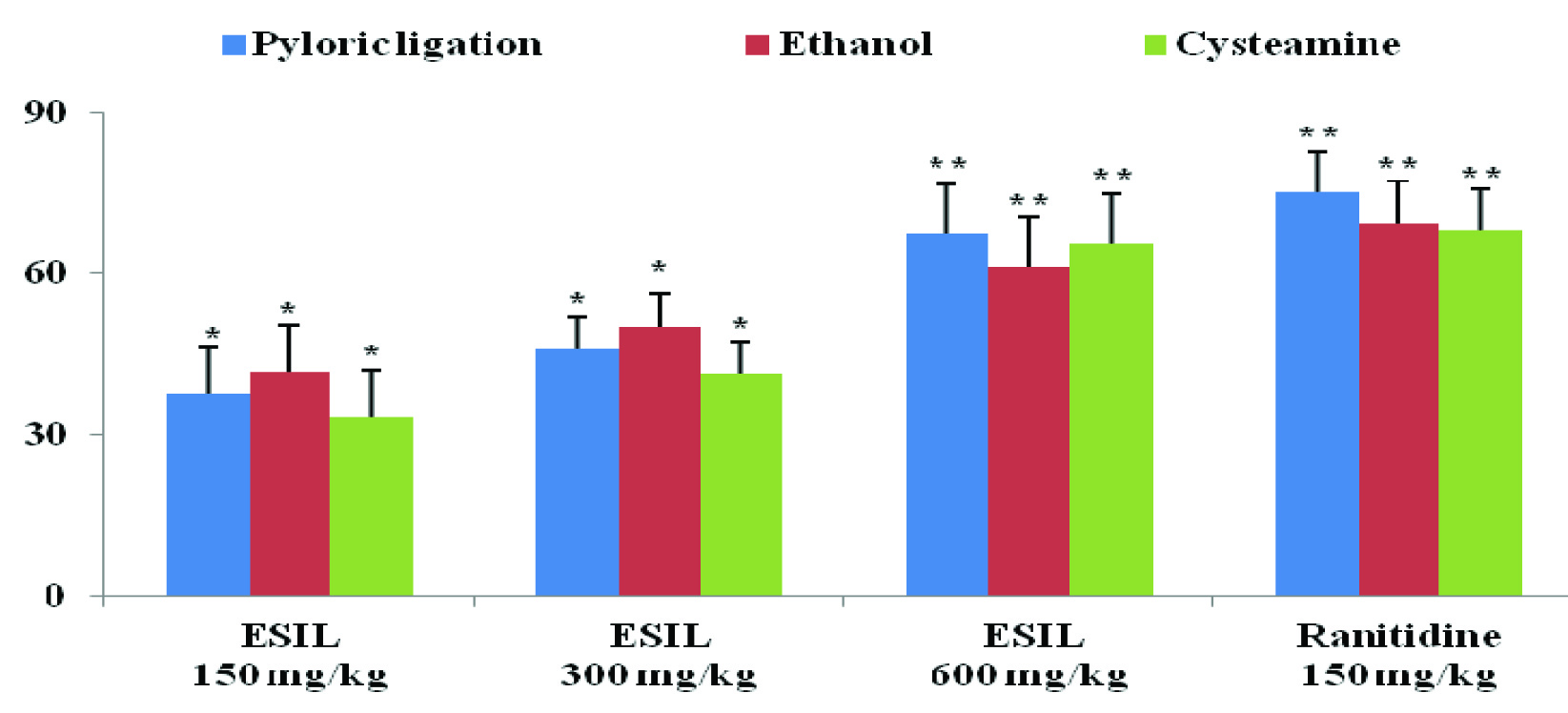
Effect of ESIL on Cysteamine Induced Duodenal Ulcers
In the cysteamine induced duodenal ulcers, oral administration of ESIL showed a reduction in ulcer index in a dose dependent manner. The percentage ulcer inhibition was 33.33, 41.43 and 65.5 for 150, 300 and 600 mg/kg respectively and that of the standard was found to be 68.05 [Table/Fig-3], [Table/Fig-4].
Discussion
The cause of gastric ulcer is due to stress induced increase in gastric acid (HCl) secretion and these acid secretions promote ulceration due to exposure of the unprotected lumen of the stomach to the accumulating acid [16–18] Pylorus ligation induced ulcers are shown by auto digestion of the gastric mucosa and breakdown of the gastric mucosal barrier which resulted as upper gastrointestinal damage including lesions, ulcers and life threatening perforation and haemorrhage. The pyloric ligation of the stomach causes accumulation of gastric acid which leads to development of ulceration in stomach. The agents who decrease gastric acid secretion and increase mucus secretion are effective in preventing the ulcers induced by this method. Like ranitidine, omeprazole acts as anti-ulcer agent by antisecretory mechanism via inhibition of gastric secretion and pepsin activity [12]. In the present study, ESIL prevents the ulcer may be by antisecretory and cytoprotective property.
Ethanol is responsible for disturbances in gastric secretion, damage to the mucosa, alterations in the permeability, gastric mucus depletion and free radical production. The generation of free radicals was produced by continuous release of superoxide anion and hydroperoxy free radicals during metabolism of ethanol [19]. Ethanol induced gastric ulceration may be occurred due to stasis in gastric blood flow which contributes to the development of the haemorrhage and necrotic tissue injuries. Alcohol has ability to penetrate the gastric mucosa and causing the cellular damage which increases the permeability to sodium and water. In other hand, the accumulation of intracellular calcium causes the pathogenesis of gastric injury that leads to cell death and exfoliation of surface epithelium [16]. The present study observed that the ESIL significantly reduced ethanol induced ulcer by cytoprotective action via antioxidant effect. The ESIL extract showed cytoprotection against the ethanol and cysteamine induced ulceration by reducing the gastric acid secretion. Similarly, the cytoprotective effect of ESIL against cysteamine induced duodenal ulcers may be due to strengthening of duodenal mucosa or by increase of duodenal alkaline secretion or luminal prostaglandin level [12].
The results of this study found that ESIL established a cytoprotective action against ethanol induced cellular damage in the gastric mucosa of rats. Cytoprotection of anti-ulcer drugs has been recognised due to the generation of prostaglandins [20]. It has also been observed that ESIL significantly and dose dependently reduced the extent of gastric ulceration in pylorus ligated rats without affecting the gastric secretion or pepsin activity. The defence potential of mucus perimeter of gastric mucosa depends upon a delicate balance between the processes affecting the synthesis and secretion of mucin constituents. ESIL prevented the mucosal lesions induced by alcohol [21]. The modern approach towards a potent antiulcer agent involves a delicate balance of controlling the synthesis, secretion and metabolism of proteins, glycoproteins and lipids, so as to strengthen the mucosal integrity [22].
Several scientific studies revealed that the phytoconstituents like flavonoids, tannins, terpenoids and saponin were responsible for gastro protective agents [23]. Tannins possess as an antiulcer agent by its astringency property and vasoconstriction effects. Due to precipitation of micro proteins on the ulcer site, a protective layer was formed which hinders gut secretions and protects the mucosa from toxins and other irritants. Previous studies have recommended that these above active compounds had ability to stimulate mucus, bicarbonate and prostaglandin secretion and neutralize with the deteriorating effects of reactive oxidants in gastrointestinal lumen [24]. Therefore, ESIL possess antiulcer activity, may be due to presence of tanins, flavonoids and terpenoids.
Conclusion
The present study concluded that the antiulcer activity of ESIL may be attributed to antisecretory, cytoprotective and antioxidant properties. The bioactivity-guided phytochemical screening of ESIL revealed the presence of flavonoids, tannins and triterpenoids, which may be responsible for the anti-ulcer effect and can be further fractionated and investigated for their role and utility in any of the anti-ulcer mechanisms.