Benign Pulmonary Metastasizing Leiomyoma of the Uterus
Nurettin Aka1, Reyyan Iscan2, Gültekin Köse3, Isik Kaban4
1 Associate Professor, Department of Obstetrics and Gynecology, Haydarpasa Numune Teaching Hospital, Istanbul, Turkey.
2 Faculty, Department of Obstetrics and Gynecology, Haydarpasa Numune Teaching Hospital, Istanbul, Turkey.
3 Faculty, Department of Obstetrics and Gynecology, Haydarpasa Numune Teaching Hospital, Istanbul, Turkey.
4 Faculty, Department of Obstetrics and Gynecology, Haydarpasa Numune Teaching Hospital, Istanbul, Turkey.
NAME, ADDRESS, E-MAIL ID OF THE CORRESPONDING AUTHOR: Dr. Isık Kaban, Department of Obstetrics and Gynecology, Haydarpasa Numune Teaching Hospital, Istanbul, Turkey.
E-mail: drisik@mynet.com
Benign Metastasizing Leiomyoma (BML) is a rare disease which represents with multiple leiomyomatous lesions in many tissues and organs especially in lungs. These patients have been operated for leiomyoma of the uterus. Here we report a case of a 41-year-old woman who was evaluated in a thoracic surgery hospital for dyspnea and bilateral nodules in chest roentgenogram. She had no history of neoplasm, only myomectomy history of uterine leiomyoma 10 years ago. Biopsy and histopathological examination were consistent with pulmonary leiomyoma. The patient was reffered to our clinic and we performed a total abdominal hysterectomy for her multiple uterine leiomyomas. The final diagnosis was ‘benign pulmonary metastasizing leiomyoma’. After this diagnosis, surgical castration was performed but two years later, repeat imaging showed progression in pulmonary lesions and progesterone therapy was administered to the patient. Patient has continued on this hormonal therapy to date and during the 5-years follow-up, the persisting lesions in both lungs regressed.
Lung, Metastatis, Multiple pulmonary nodules
Case Report
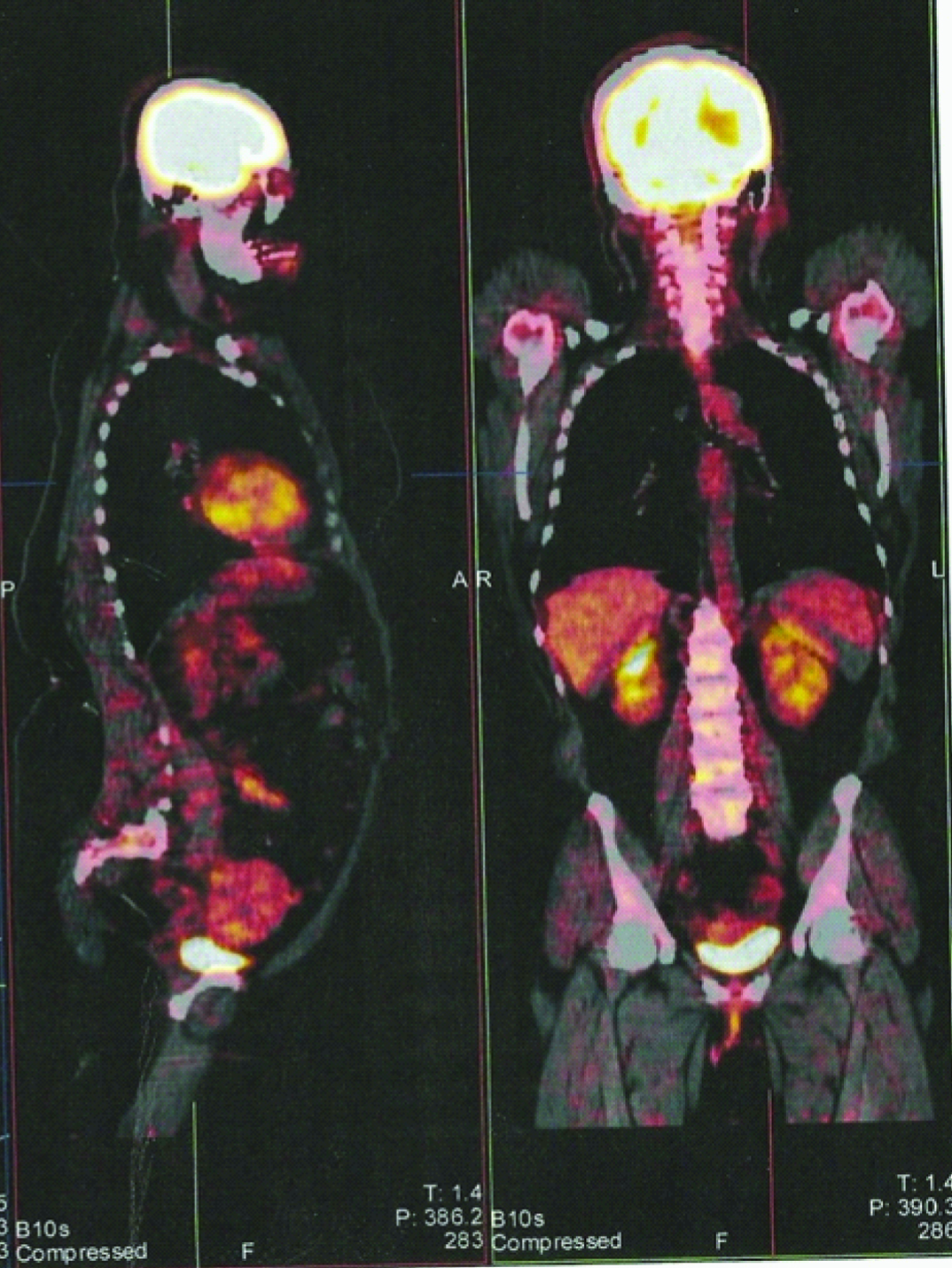
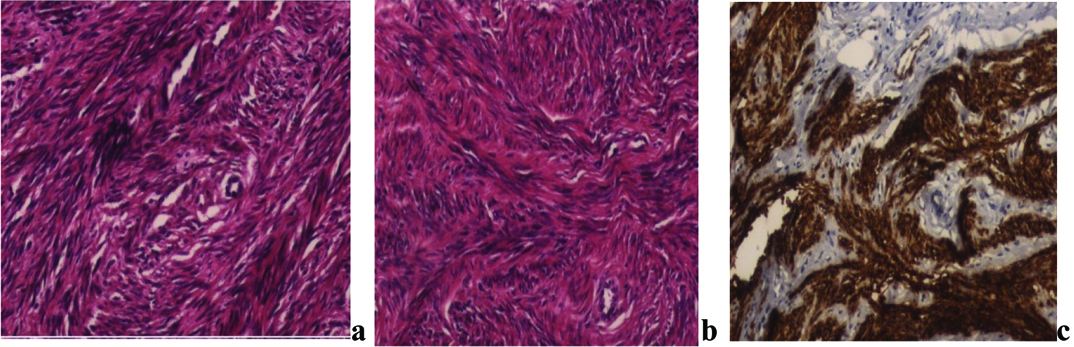
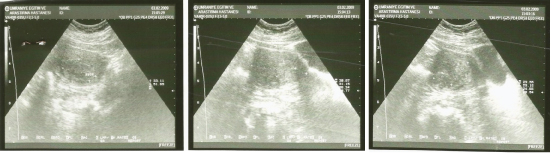
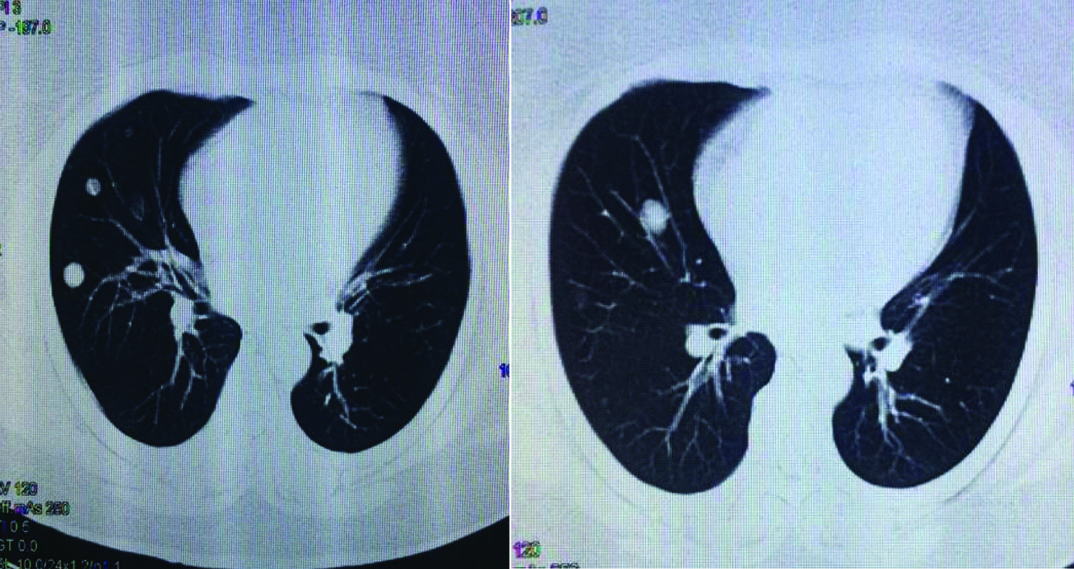
A 41-year-old non-smoker woman was admitted to a thoracic surgery hospital for dyspnea and bilateral nodules in chest roentgenogram. Her past medical history include myomectomy for uterine leiomyoma 10 years ago. Chest Computerized Tomography (CT) demonstrated multiple nodules in both lungs with a maximum diameter of 15 mm, located peripheral and central. A flexible bronchoscopy was performed, there were no abnormalities in the airway and bronchoalveolar lavage resulted as normal bronchial epithelial cells. Positron Emission Tomography (PET/CT) was performed to evaluate malignancy. There was no abnormal Fluorodeoxyglucose (FDG) uptake with the suspicious multiple nodules [Table/Fig-1] but the maximum standard uptake value (SUVmax) was about 5,92 in uterus that was interpreted as benign finding (uterine leiomyomas or endometrial cycle) [Table/Fig-2]. Video-Assisted Thoracoscopic Surgery (VATS) and wedge resection of the lesion in the right middle lobe was performed for pathological diagnosis. Histopathologic evaluation of the specimen resulted as benign leiomyoma without mitosis and necrosis [Table/Fig-3a&b]. Immunohistochemical staining was positive for Smooth Muscle Actin (SMA) [Table/Fig-3c] and Desmin, negative for Carcinoembryonic Antigen (CEA). With these results, the patient was referred to our clinic for evaluation of metastasizing leiomyoma. Transvaginal Ultrasonography (TV-USG) showed multiple intramural and submucosal myomas with diameters about 4-5cm [Table/Fig-4]. Magnetic Resonance Imaging (MRI) showed similarly and there was no pathological feature in adnexial structures. A total abdominal hysterectomy and unilateral salpingo oopherectomy was performed. Salpingo oopherectomy was performed for surgical reasons due to adhesions from the previous surgery. The postoperative period was uncomplicated and the patient was discharged 3 days after the surgery. Histopathological evaluation of the resection specimen confirmed the presence of uterine myomas, 5 cm in maximum diameter. Microscopically, mitosis, necrosis or atypical features were not observed in the tumoural tissue. Immunohistochemical assay for Ki-67, in pulmonary and uterine specimens, showed low proliferative index (0-1%) and after this result we excluded leiomyosarcoma or leiomyosarcoma metastasis. The tumoural lesions were positive for Desmin, Estrogen Receptors (ERs) and Progesterone Receptors (PRs) and negative for S100 and 34 beta 12. The final diagnosis was ‘benign pulmonary metastasizing leiomyoma’. After this diagnosis surgical castration was planned and two months later unilateral oopherectomy was performed. Approximately two years later, repeat imaging showed progression in pulmonary lesions [Table/Fig-5]. Due to this progression progesterone therapy (Megestroleacetat- 160mg/day) was started. The patient has continued on this hormonal therapy to date and currently she is asymptomatic. During the 5-year follow-up the remaining lesions in both lungs regressed.
PET/CT shows that there is no abnormal Fluorodeoxy Glucose (FDG) uptake with the suspicious multiple nodules (arrows).
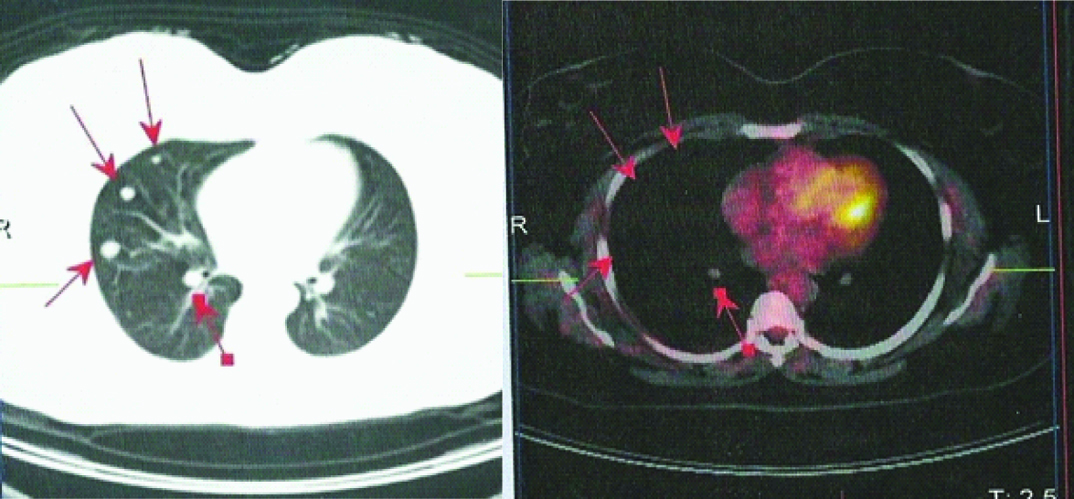
Minimal uptake in uterus due to uterine leiomyomas or endometrial cycle.
Histology and immunohistochemical staining of pulmonary and uterine leiomyomas. a) spindle cells are arranged in elongate fascicles. The cytoplasm is eosinophilic and nuclei are bluntended (20xHE); b) Elongated spindle cells with eosinophilic cytoplasm are arranged in fascicles. (20xHE); c) smooth muscle actin highlights the smooth muscle cells (SMAx20)
Transvaginal ultrasound images.
Computerized tomographic imagings.
Discussion
BML is a rare condition which is a similar histological appearance with benign smooth muscle tumours but present at distant sites, and it usually occurs years later after uterine leiomyoma surgery [1]. This condition is characterized by multiple leiomyomatous lesions in many tissues, organs and lungs are the most common site of the metastases. Benign metastasizing leiomyoma is rarely diagnosed and there have been over 150 cases reported in the literatüre [2]. The first case was presented in 1939 by Steinerin whom leiomyomas of the uterus had metastasized to the lungs and caused chronic pulmonary obstruction with cor pulmonale and right sided heart failure and her death at the age of 36. The microscopic appearence of the metastases and uterine tumours at the postmortem examination was benign and fully differentiated. He had used the term ‘metastasizing fibroleiomyoma’ for the description of this rare condition [3]. The term changed after several case reports and widely accepted as ‘benign metastasizing leiomyoma’in the literature. Because of any morphological or genetic signs of malignancy some of the writers also prefer to call this rare disease as ‘systemic leiomyomatosis’ [4].
The aetiology and risk factors of BML is still unclear but several hypothesis have been suggested on the histopatogenesis such as metastases of the highly differentiated sarcomas of the uterus, lung emboli of cells from benign leiomyoma of the uterus and generalized leiomyomatosis mainly involving the uterus and the lungs [5]. Due to the majority of women with BML, a prior surgery for uterine leiomyoma or dilatation and curettage, ‘surgically induced vascular spread’ becomes the most acceptable theory [1]. Clinical history has to be thoroughly evaluated to exclude other disorders which are mophologically similar to BML like primary leiomyoma of the lungs and metastasis of the low grade leiomyosarcoma of the uterus [5]. However, in some cases uterine leiomyoma is discovered after the metastasis [6] so the theories about pathogenesis of BML is still controversial.
Histological findings of the presented cases in the literature are similar and just like benign tumours. As was shown in our case, the lesions of BML macroscopically were solid, well delimited tumour masses which were composed of spindle-shaped cells with low mitotic rate and absence of necrosis and cellular atypia. Immunhistochemical examinations were done in all cases which include antibodies against desmin, SMA, vimentin, S-100, estrogen and progesterone receptors, antibodies against Ki-67 for measurement of proliferation. In our case, tumour cells of the uterine and pulmonary lesions were both positive for desmin and SMA that confirms the smooth muscle origin, the tumours were negative for S-100 that excludes the neural origin and are positive for ERs and PRs. Most BML’s were positive for ERs, But extra uterine leiomyomas are uniformly ER negative and only 13% of the leiomyosarcomas stained weakly for ERs [7,8]. This data about estrogen receptors defend the theory of multifocal origin that has been suggested in some cases with unusual distribution of metastatic leiomyomatous lesions. Ki-67 proliferation index was 0-1% in our case which excluded leiomyo sarcoma and metastases of leiomyomasarcoma. With in situ hybridizationtechniques using mir-221 micro-RNA analysis showed the use of these features in the differential diagnosis between leiomyoma and leiomyosarcoma [5]. According to this finding they suggested that BML originates from a genetically distinctive subset of uterine leiomyomas.
The diagnosis was based on the morphological, immunohistochemical and radiological findings of the metastatic and uterine masses in the patients with the surgical history for the uterine leiomyoma. Most pulmonary metastases were discovered during the routine chest roentgenogram as bilateral, multiple, nodular opacities. For further examination a thorax CT can be performed to determine the exact size and location of the lesions before the biopsy [5]. Recently, PET-CT is used in the differential diagnosis of BML. The nodules show any significant FDG uptake as in our case, despite the higher uptake of FDG in malignant tumours or in uterine leiomyosarcoma. In interpreting the PET-CT we should keep in mind that in some malignant tumours with mucinous component and carcinoid tumours, the metastatic lesions may also show any significant FDG uptake [9]. For the histopathologic diagnosis, biopsy from the lesions has to be performed. In order to take enough tissue sample from the metastatic pulmonary lesions VATS biopsy is recommended instead of Fine-Needle Aspiration (FNA) [10].
Currently there is no standardization for the treatment. But because of the presence of estrogen and progesterone receptors in BML, surgical resection of the lesions and bilateral oophorectomy seems to be the primary treatment. Otherwise hormonal therapy is the best alternative for the patients who refuse surgery or have unresectable masses or have progression of the disease despite surgery as in our case. Recently, novel hormonal treatment experiences have been reported in literature which are; progesterone, GnRH agonists, GnRH antagonists, aromatase inhibitors (anastrazole, letrozole) and selective estrogen receptor modulators (tamoxifen, raloxifene) [3,10]. The investigators who are using these novel medications for the hormonal management suggested that hormonal therapy could be more effective if we individualize doses and use combinations in selected patients [10].
Conclusion
Although rare, BML should be considered in patients with multiple metastatic masses in the lungs or other tissues occuring several years after the surgical intervention for uterine leiomyomas. Clinical presentation, radiological findings, immunohistochemical staining and histopathology of the metastatic lesions are characteristic in almost all patients in the literatüre. The cytogenetic studies seem to give us the most helpful findings in the differential diagnosis. There are no guidelines about the management but we commend the surgery primarily that includes the resection of the pulmonary nodules if possible, hysterectomy and bilateral oopherectomy. Addition of the hormonal therapy can be necessary in some cases and individually adjusted doses or combination therapies might be efficacious in selected cases.
[1]. Nakajo M, Nakayama H, Sato M, Fukukura Y, Nakajo M, Kajiy Y, FDG-PET/CT finding of benign metastasizing leiomyoma of thelungActa Radiologica Short Reports 2012 1(3):1-4. [Google Scholar]
[2]. Lewis EI, Chason RJ, DeCherney AH, Armstrong A, Elkas J, Venkatesan AM, Novel hormone treatment of benign metastasizing leiomyoma: an analysis of five cases and literature reviewFertil Steril 2013 99(7):2017-24. [Google Scholar]
[3]. Ki EY, Hwang SJ, Lee KH, Park JS, Hur SY, Benign metastasizing leiomyoma of the lungWorld J Surg Oncol 2013 11:279 [Google Scholar]
[4]. IuIu A, Danilova NV, Shikeeva AA, Kekeeva TV, Zavalishina LE, Frank GA, Benign metastatic leiomyoma of the corpusuteriArkh Patol 2012 74(6):39-43. [Google Scholar]
[5]. Nuovo GJ, Schmittgen TD, Benign metastasizing leiomyoma of the lung: clinicopathologic, immunohistochemical, and Micro-RNA analyses. diagnostic molecular pathologythe American Journal of Surgical Pathology, part B 2008 17(3):145-50. [Google Scholar]
[6]. Banner AS, Carrington CB, Emory WB, Kittle F, Leonard G, Ringus J, Efficacy of oophorectomy in lymphangio leiomyomatosis and benign metastasizing leiomyomaN Engl J Med 1981 305:204-09. [Google Scholar]
[7]. McGinley KM, Bryant S, Kattine AA, Fitzgibbon JF, Googe PB, Cutaneous leiomyomas lack estrogen and progesterone receptor immunoreactivityJ Cutan Pathol 1997 24:241-45. [Google Scholar]
[8]. Rivera JA, Christopoulos S, Small D, Trifiro M, Hormonal manipulation of benign metastasizing leiomyomas: report of two cases and review of the literatureJ Clin Endocrino Metab 2004 89(7):3183-88. [Google Scholar]
[9]. Radzikowska E, Szczepulska-Wojcik E, Langfort R, Oniszh K, Wiatr E, Benign pulmonary metastasizing leiomyoma uteri. Case report and review of literaturePneumonologia i Alergologia Polska 2012 80(6):560-64. [Google Scholar]
[10]. Chen S, Zhang Y, Zhang J, Hu H, Cheng Y, Zhou J, Pulmonary benign metastasizing leiomyoma from uterine leiomyomaWorld J Surg Oncol 2013 11:163 [Google Scholar]