Follicular Dendritic Cells (FDC) is non-lymphoid, non-phagocytic cells in the stroma of lymph nodes. FDCs are critical mediators of adaptive humoral immune response through antigen presentation and regulate the germinal centre reaction in primary and secondary follicles [1,2]. Neoplasms of histiocytic and dendritic cells are rare disorders of the lymph node and soft tissues. Follicular Dendritic Cell Sarcomas (FDCS) are classified under histiocytic and dendritic cell neoplasms by the WHO classification of tumours [3]. FDCS are uncommon tumours; the majority occur in lymph nodes whereas the extra nodal FDCS, presenting in a wide variety of sites such as the Gastrointestinal Tract (GIT), pharynx, mediastinum, skin, liver, spleen etc, are rarer [4–7]. The tonsil is the most common site of FDCS in the head and neck region and the first case of FDC tumour of tonsil was reported in 1986 [8]. Accurate characterization of this neoplasm is important in planning optimal treatment given its potential for recurrence and metastasis. Due to its rarity, optimal management of this entity is still under evaluation. In this study, we have analysed the clinicopathological profile and outcomes of 10 cases of follicular dendritic cell sarcoma arising in the head and neck region diagnosed at our regional cancer centre.
Materials and Methods
A retrospective analysis of all patients with follicular dendritic cell sarcoma diagnosed and treated between 2007 and 2013 in our Institute was done. A total of 10 cases diagnosed with FDCS of the head and neck were retrieved and reviewed from our hospital cancer registry.
Clinical details such as age, gender, symptoms and location of the tumour were noted. Paraffin embedded tissue blocks, Haematoxylin-Eosin (H&E) stained and immunohistochemistry (IHC) stained slides of all the 10 patients were re-reviewed and pathological features such as tumour site, grade, IHC staining for immune-markers consisting of CK, EMA, Desmin, HMB45, CD21, CD23, LCA and S-100 were noted. Treatment details such as surgery, radiotherapy and chemotherapy received were noted. Fine Needle Aspiration Cytology (FNAC) of the cervical lymph nodes was done in three cases and the diagnoses of paraganglioma was given in one case and poorly differentiated carcinoma in the other two cases. In our series, all the patients with enlarged and hard cervical lymph nodes underwent surgical procedures with cervical lymph node dissection. All patients with cervical lymph node involvement were recommended adjuvant radiotherapy after cervical lymph node dissection whereas, patients without cervical lymph node enlargement were recommended adjuvant chemotherapy. Patients with high grade tumours and cervical lymphadenopathy were recommended ‘tri-modality treatment’ (surgery followed by radiotherapy and then chemotherapy). All patients were followed up for a period of 60 months during which all the recurrences were recorded. The Recurrence Free Survival (RFS) of all the patients was recorded.
Results
The important clinicopathological characteristics of the ten patients are outlined in [Table/Fig-1].
Clinical data of 10 cases of FDCS
| S.N | Sex | Age | Presentation | Site | Grade | + ve IHC Markers | Treatment | RFS (m) |
|---|
| 1 | M | 63 Y | Dysphagia | L Tonsil | Low | CD21, CD23 | Sx RT | 52 |
| 2 | M | 26 Y | Nasal Stuffiness B/L Neck LNE | Naso-pharynx | High | CD21, CD23, S100, EMA | Sx® RT →CTh | 44 |
| 3 | M | 64 Y | Dysphagia and R Neck LNE | Hypo-pharynx | Low | CD21, CD23 S-100 | Sx→ RT | 39 |
| 4 | M | 28 Y | Dysphagia and R Cervical LNE | R Tonsil | High | CD21, CD23 EMA | Sx→ RT → CTh | NED |
| 5 | M | 66 Y | Dysphagia and L Cervical LNE | R Tonsil | Low | CD21, CD23 EMA, | Sx→ RT | 31 |
| 6 | F | 68 Y | Throat pain | L Tonsil | Low | CD21, CD23 EMA | Sx | 19 |
| 7 | F | 65 Y | Dysphagia | L Tonsil | Low | CD21, CD23 | Sx→CTh | 47 |
| 8 | M | 40 Y | Dysphagia | L Tonsil | Low | CD21, CD23 EMA | Sx→CTh | NED |
| 9 | F | 51 Y | Dysphagia | L Tonsil | Low | CD21, CD23 | Sx→CTh | NED |
| 10 | M | 38 Y | Dysphagia and R Neck LNE | R Tonsil | High | CD21, CD23 S-100 | Sx→ RT →CTh | 45 |
Y – Years, B/L – Bilateral, R – Right, L – Left, LNE – lymph node enlargement
Sx – Surgery, RT – Radiotherapy, CTh – Chemotherapy, RFS – Recurrence Free Survival, m – months, NED – No evidence of disease.
Clinical features: Of all the ten head and neck FDCS cases, seven were males and three were females. The median age of the series was 57 years (Range – 26 to 68 years). In eight of the ten patients, the tumours were located in the tonsils. Nasopharynx and hypopharynx were the other two sites. All patients except one presented with dysphagia and feeling of lump in the throat. The duration of presenting symptoms ranged from 2 months to 16 months. Cervical lymph node enlargement was present in three cases. In all the ten patients, the sizes of primary tumours as well as the neck nodes at presentation were less than 5cm.
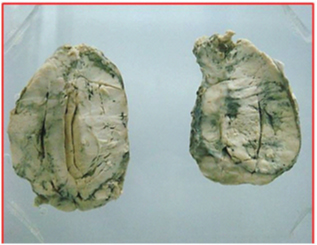
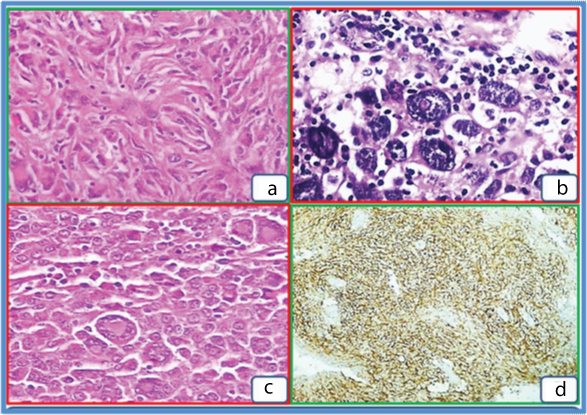
Pathological features: All the ten cases underwent surgical excision of the tumour and also the cervical lymph nodes if they were enlarged. The specimens on gross examination showed fairly circumscribed nodular, grey white masses with firm consistency [Table/Fig-2]. The 10 cases had similar histo-morphological features. The tumours were composed of uniform, oval to spindle cells arranged in a vaguely storiform pattern, at places exhibiting a diffuse pattern and interlacing fascicles [Table/Fig-3a]. The cell type varied from bland ovoid spindle cells to markedly pleomorphic cells in one case. Tiny foci of necrosis were also noted in all cases; three cases showed prominent tumour giant cells [Table/Fig-3b]. A notable feature in one case was the presence of a spectrum of cells from bland to pleomorphic forms. The presence of Touton type of giant cells in another case was also a notable feature [Table/Fig-3c]. Three patients had high grade tumours classified by the presence of more than five mitotic figures per 10 high power fields (hpf). A constant feature noted was the presence of scattered small lymphocytes and plasma cells throughout the tumour. All the tumour cells showed diffuse cytoplasmic CD21 and CD23 positivity by IHC [Table/Fig-3d]. S-100 was positive in two cases. All other markers were negative.
Well circumscribed, firm, grey mass.
a – H&E (X 100) storiform architectural pattern.
b – Nuclear pleoorphism, pseudo-inclusions, scattered lymphocytes.
c – Touton giant cells.
d – CD23 positive cells (rabbit polyclonal Anti – CD 23,1:40 dilution, Biogenex, CA, USA.
Treatment Outcomes: All patients underwent complete resection of the tumours [Table/Fig-1]; patients with cervical lymphadenopathy underwent selective neck node dissection additionally. Surgery was followed by radiation therapy in six patients (30 Gray delivered in 15 fractions) to include the primary and the enlarged neck node sites. One patient declined adjuvant therapy after surgery but remained on follow-up for five years. Six patients received adjuvant Cyclophosphamide, Hydroxydaunorubicin, Oncovin (vincristine), Prednisone (CHOP) chemotherapy after surgery; three patients received chemotherapy after adjuvant radiotherapy whereas three patients received chemotherapy after surgery. All the three patients with high grade tumours were uniformly managed with Tri-modality treatment.
Three patients remained recurrence free at the end of the 60 months, of which two patients had low grade tumours. Hence the recurrence rate was 70%. The mean and median recurrence free survival was 39.6 and 44 months, respectively (range 19 – 52 months). The mean Radio Frequency Systems (RFS) of the two patients with recurrent high grade tumours was 44.5 months. In the six patients with recurrent low grade tumours, the mean and median RFS were 37.6 and 39 months respectively (range 19 – 52 months).
Discussion
Follicular dendritic cell sarcomas are rare tumours of dendritic-histiocytic cell origin. The first case of tonsillar FDCS was reported by Monda et al., in 1986 and later by Nayler et al., in 1996 [8,9]. Subsequently, Chan et al., reported the first extranodal tumour in 1994 [10]. Nodal FDCSs are more common and often involve cervical lymph nodes. In a pooled analysis by Saygin et al., 58% of cases occurred in extranodal sites, including the tonsil, oral cavity, GIT, intra-abdominal soft tissue and breast. Ten percent of all patients presented with both nodal and extranodal disease [11]. The diagnosis is challenging as the neoplasm mimics several tumours of the head and neck region; however, the possibility of FDCS should be considered in all “difficult-to-fit” neoplasms. Recognition of extranodal FDCS requires a high index of suspicion as most cases as the presentation of most cases is similar to that of the commoner histologies of carcinoma and lymphoma. Correct characterization of these neoplasms is imperative given their potential for recurrence and metastasis.
In the review by Wang et al., a total of 142 cases of extranodal FDCS from different publications were reviewed. The cumulative male: female ratio was 1.2:1, with the age ranging from 9 to 82 years [12]. The predominant extranodal site of FDCS was the head and neck region followed by the liver, spleen, gastrointestinal tract and thoracic viscera.
Recently, Li et al., combined and reviewed all the head and neck FDCSs published in the literature [13]. No gender predominance was noted in head and neck FDCS and the median age was 46 years. In our study of ten cases, the median age was 57 years (range – 26 to 68 years), and male: female ratio was 7:3 [Table/Fig-1]. Five patients presented with cervical lymph node involvement.
FDCS are tumours of mesenchymal origin. Histologically, morphological resemblance to commoner tumours such as tumours of the minor salivary gland, inflammatory myofibroblastic tumour, smooth muscle and fibrohistiocytic tumours, metastatic sarcoma, undifferentiated carcinoma and also melanoma is possible. Hence, expertise complemented by IHC is essential for diagnosis [14,15]. All the tumours in this series had similar morphological features.
Follicular dendritic cell markers such as CD21, CD23, CD35 positivity complement the morphological diagnosis [14,15]. Variable positivity for desmoplakin, vimentin, HLA-DR, EMA, S-100 and CD68 (positive in histiocytic tumours) can be occasionally encountered [5]. In our series, all patients were CD21, CD23 positive whereas EMA and S-100 positivity was seen in 50% and 30% of cases respectively. Newer markers like clusterin and podoplanin help to differentiate FDCS from other histiocytic tumour but are not commonly available for testing. FDCS have an indolent course with a substantial risk of local recurrence in 50 % and distant metastasis in 25% cases [14]. The rate of recurrence in our series was 70%. Surgery is the mainstay of treatment for early and locally advanced FDCS cases; non – metastatic patients treated with surgery had significantly better survival rates when compared to those treated with non – surgical modalities. Adjuvant local radiotherapy of 30 Gray and chemotherapy regimens such as CHOP such as that used for non – Hodgkin lymphoma treatment are commonly used for adjuvant treatment [16]. There is no conclusive data for the effective management of FDCS due to the rarity of these tumours. In the study by Chan et al., high mitosis, nuclear pleomorphism and lack of adjuvant therapy were predictors of higher recurrence rate [14]. In the study by Li et al., comprising of head and neck FDCS, it was noted that low grade FDCS tumours had a lower recurrence rate compared to the high grade tumours [17]. In our study the RFS was better for the high grade tumours compared to that of low grade tumours. This contrast may be due to the fact that all high grade tumours were treated uniformly with tri-modality treatment and that there was heterogeneity in the management of low grade tumours in our series. However, due to small numbers and due to treatment heterogeneity, it is difficult to make conclusion on these results. Due to the small numbers, the advantage with tri-modality treatment for high grade tumours is not conclusive, but suggestive. The RFS of patient treated with surgery followed by RT management was comparable to outcomes seen in other similar studies [14–22].
Limitation
This study is limited by the sample size to conclude on the effectiveness of tri-modality treatment in high grade FDCS. Tri-modality management may have a role in such scenarios, the benefit of which needs to be studied in larger studies.
Conclusion
Early recognition of follicular dendritic cell sarcomas requires a high index of suspicion and bi-modality or tri-modality treatment may cure a subset of low and high grade tumours respectively and prolong recurrence in a large subset of patients. Surgery is the mainstay and the definitive modality of treatment; the advantages and benefit of adjuvant radiotherapy and chemotherapy are yet to be established. Tri-modality management may have a role in high grade patients which needs to be studied further.
Y – Years, B/L – Bilateral, R – Right, L – Left, LNE – lymph node enlargement
Sx – Surgery, RT – Radiotherapy, CTh – Chemotherapy, RFS – Recurrence Free Survival, m – months, NED – No evidence of disease.