Desmoplastic Ameloblastoma Arising in a Dentigerous Cyst – A Case Report and Discussion
Muruganandhan Jayanandan1, Shaik Mohamed Shamsudeen2, Satish Kumar Srinivasan3, S. Nalin Kumar4, Tyson Babu Pulikkottil5
1 Reader, Department of Oral and Maxillofacial Pathology, Sri Venkateswara Dental College and Hospital, Chennai, Tamilnadu, India.
2 Senior Lecturer, Department of Oral and Maxillofacial Pathology, Sri Venkateswara Dental College and Hospital, Chennai, Tamilnadu, India.
3 Senior Lecturer, Department of Oral and Maxillofacial Pathology, Sri Venkateswara Dental College and Hospital, Chennai, Tamilnadu, India.
4 Professor and Head, Department of Oral and Maxillofacial Pathology, Sri Venkateswara Dental College and Hospital, Chennai, Tamilnadu, India.
5 Dental Surgeon, Mar Timotheus Charitable Hospital, Thrissur, Kerala, India.
NAME, ADDRESS, E-MAIL ID OF THE CORRESPONDING AUTHOR: Dr. Muruganandhan Jayanandan, Reader, Department of Oral and Maxillofacial Pathology, Sri Venkateswara Dental College and Hospital, Thalambur-600130, Chennai, Tamilnadu, India.
E-mail: drmurugan@outlook.com
Dentigerous cyst is a fairly common odontogenic cyst with an uncomplicated prognosis. However, reports of odontogenic and non-odontogenic tumors arising from the cysts have been published for many years. Therefore, the importance of thorough histopathological examination and regular follow-up cannot be overemphasized. Here is a case report of a possible development of desmoplastic ameloblastoma from a dentigerous cyst. This case is unique for two main reasons. Firstly, to our knowledge such a case has not been reported previously. Secondly, the tumor seemed to arise from the capsule and not from lining of the dentigerous cyst. Transformation of odontogenic rests into cysts is known to occur in the syndromic cases of odontogenic keratocysts (resulting in recurrent and daughter cysts) but not in dentigerous cysts. This case report may prompt greater interest in the role of the supposedly inactive odontogenic nests in the capsule of these cysts.
Cyst-tumor transformation, Desmoplasia, Odontogenic cyst, Odontogenic tumor
Case Report
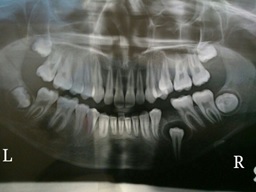
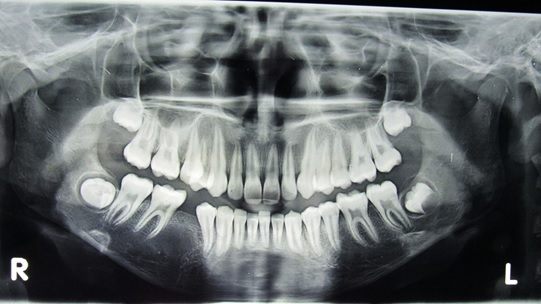
A 13-year-old female patient reported to the dental clinic with complaint of dental protrusion and persisting milk teeth in her lower right dental arch. Her medical history was unremarkable. Clinical examination revealed a retained deciduous mandibular right 2nd molar (FDI-85) and absence of the second premolar (FDI-45). There was no significant bony expansion or other remarkable intraoral features. Radiographs were recommended to determine the development of the permanent mandibular right 2nd premolar. The retained deciduous tooth was extracted. The orthopantomograph revealed an impacted FDI-45. A well–defined radiolucency surrounded its crown extending to the cervical third of the root. The lesion was completely radiolucent with a sclerotic rim and seemed to have displaced the roots of the 1st premolar and 1st molar [Table/Fig-1].
Orthopantomograph of the patient showing radiolucency around the crown of impacted 45.
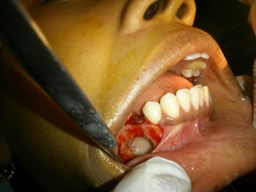
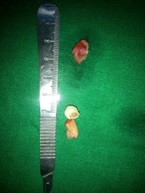
A provisional diagnosis of dentigerous cyst was given with differential diagnoses of follicular keratocyst, and other odontogenic cysts/tumors etc., being considered. The patient was advised complete surgical excision of the lesion. Surgical exploration revealed a cystic lesion which was enucleated from the bony wall along with removal of the impacted tooth [Table/Fig-2]. The cystic fluid was straw colored and clear. The specimen was sent for histopathological evaluation [Table/Fig-3].
Post-surgical photograph.
Gross specimen before processing.
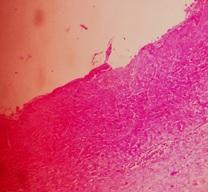
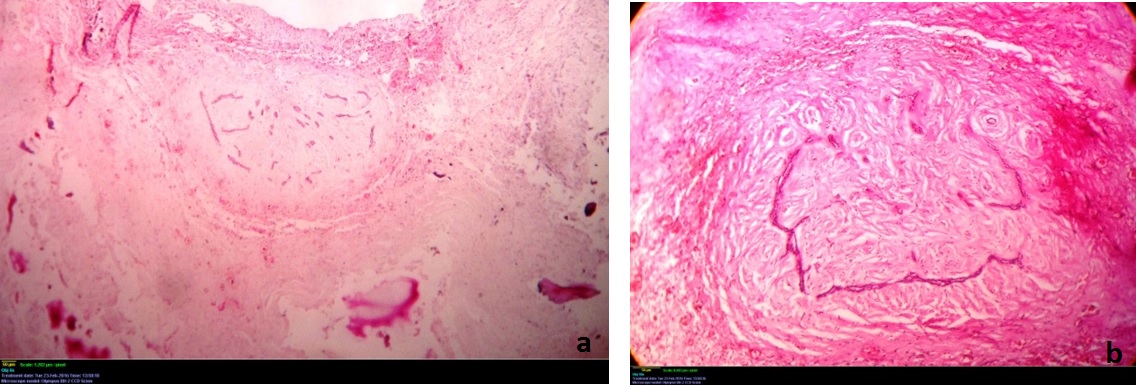
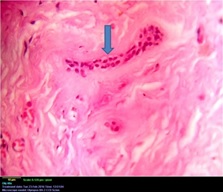
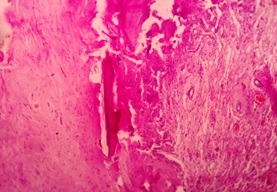
Histopathological examination revealed a cystic lesion which was lined by 3-5 layers of stratified non-keratinized epithelium consisting of cuboidal basal cells and flattened suprabasal cells. The cells had large hyperchromatic nuclei and eosinophilic cytoplasm. However, the lining was attenuated in most regions. The capsular tissue was cellular with delicate collagen fibers and resembled dental follicular tissue [Table/Fig-4]. Numerous nests of odontogenic epithelium were evident in the capsule, which are apparently inactive, as evident from the lack of inductive change (hyalinization) at the juxtaepithelial region [Table/Fig-5a,5b]. At a particular region, cords and strands of odontogenic epithelium were observed surrounded and compressed by a thick hyalinized collagenous stroma, distinct from the surrounding capsular tissue [Table/Fig-6a,6b]. High power view (40X) of the odontogenic strands clearly show columnar ameloblast-like cells exhibiting reversal of nuclear polarity and subnuclear vacuolation [Table/Fig-7]. The juxtaepithelial induction was evident [Table/Fig-8a,8b]. There was osseous metaplasia at places [Table/Fig-9]. Histopathological diagnosis of dentigerous cyst exhibiting focus of desmoplastic ameloblastoma was given.
Photomicrograph showing the cyst lining and capsule – hematoxylin and eosin, 10X magnification.
Photomicrograph showing the odontogenic nests – hematoxylin and eosin, 4X and 10X magnification respectively.
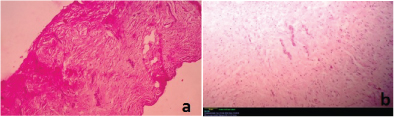
Photomicrograph showing the desmoplastic focus – hematoxylin and eosin, 4X and 10X magnification respectively.
Photomicrograph showing the ameloblastic cells – hematoxylin and eosin, 40X magnification.
Photomicrograph showing the stromal induction of the epithelial cells – Hematoxylin and eosin, 40X magnification.
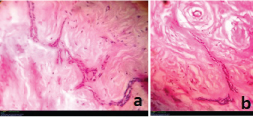
Photomicrograph showing osseous metaplasia – hematoxylin and eosin, 10X magnification.
Six month follow-up of the patient revealed no signs of recurrence, and showed radiographic evidence of healing at the surgical site [Table/Fig-10].
Discussion
The incidence of tumors arising from odontogenic cysts has been described by Gorlin et al., in 1965, who reported ameloblastomatous changes of cysts in nevoid basal cell carcinoma syndrome [1]. Stoelinga [2] and Eversole et al., [3] reported that on rare occasions, odontogenic tumors, epidermoid and muco-epidermoid carcinomas can develop from the lining of odontogenic cysts.
Dentigerous cyst is a fairly common odontogenic cyst arising from the reduced enamel epithelium of impacted teeth. The cyst is mostly innocuous in behavior but reports of transformation into odontogenic tumors have surfaced in the literature [4–7].
Desmoplastic ameloblastoma was first described by Eversole in 1984 [8]. Since then, numerous reports of the lesion have been reported [9–11]. The lesion was given a separate classification mainly due to its unusual appearance and different biologic behavior. The 2005 WHO classification categorizes ameloblastoma into solid/multicystic, unicystic, desmoplastic and peripheral variants. It is observed in a 1:1 jaw ratio, mostly occurring in anterior mandible, and radiographically resembles a fibro-osseous lesion due to mixed radiolucent-radiopaque appearance. Microscopically the lesion is predominantly composed of desmoplastic stroma compressing the odontogenic strands, which are seen in irregular and bizarre shapes. Osseous metaplasia may be seen. Immunohistochemistry shows positivity for TGF-β [12].
Incidence of desmoplastic ameloblastoma arising from dentigerous cysts seems to be extremely rare. A PubMed search with keywords “dentigerous cyst” and “desmoplastic ameloblastoma” displayed no reports of such a lesion. Our case report was clinically diagnosed as a dentigerous cyst due to its typical history, clinical and radiographic presentation. Histopathological examination revealed dentigerous cyst with numerous odontogenic epithelial strands and nests in the capsular tissue. Such nests were expected in dentigerous cyst walls; however, ameloblast-like cells inducing stromal change, was unusual. The microscopic pictures clearly revealed compressed epithelial strands surrounded by dense hyalinized stroma. The strands appeared active with columnar cells, reversed nuclear polarity and sub-nuclear vacuolation. Moreover, the aforementioned focus was circumscribed and appeared to exhibit local expansion. In other regions, smaller odontogenic nests seemed to be mostly inactive without stromal induction.
Recent studies have found that tumor-associated markers like p63, CD10 and osteopontin seemed to be up-regulated in dentigerous cysts in the process of transformation to ameloblastoma [13,14]. Levanat S et al., [15] and Pavelic B et al., [16] conducted studies on Loss of Heterozygosity (LOH) in odontogenic keratocysts and dentigerous cysts and found that in many dentigerous cysts, there were mutations in the Patched (PTCH) gene of chromosome 9q22.1–q31. They opined that the lining of the dentigerous cyst originated from a single cell (monoclonal proliferation) that implied a neoplastic genotype. Shear however, believed that most of the reports of ameloblastomatous transformation of dentigerous cyst linings are exaggerated and were mostly histopathological variations that usually exist in odontogenic tumors, especially cystic components [5]. Even if one were to agree with Shear, the unique histopathological presentation should warrant a discussion and further research into such lesions [5].
The literature on cyst-tumor transformation till now has established that tumors mostly arise from the cyst lining [5,6]. Our case appears to be unique in that the focus of desmoplastic ameloblastoma appears to arise from the odontogenic nests in the capsule. This to our knowledge has not been reported in dentigerous cysts. Medical literature does not emphasize the role of odontogenic nests in dentigerous cysts. They are of greater relevance in syndromic cases of keratocystic odontogenic tumor, where they are suspected to be one of the major causes of recurrence [4,5]. The present finding may imply that these nests may not always be inactive in dentigerous cysts either. Under conditions not completely understood, these nests may proliferate and induce changes in the stroma, which is central to the pathogenesis of odontogenic tumors. Therefore, even in dentigerous cysts, long term follow-up appears to be indicated.
Conclusion
The present case report describes a rare development of a desmoplastic ameloblastoma within a dentigerous cyst wall. Such a case has not been reported previously. This finding may heighten scientific interest on the relevance of odontogenic nests in the capsule of dentigerous cysts, which have hitherto been assumed to be innocuous. Further studies should throw more light into the pathogenesis and behavior of such lesions, given the fact that the prognosis of the cyst and the tumor are vastly different.
Ethical approval – Not applicable
[1]. Gorlin RJ, Vickers RA, Kelln E, Williamson JJ, The multiple basal-cell nevi syndrome-an analysis of a syndrome consisting of multiple nevoid basal-cell carcinoma, jaw cysts, skeletal anomalies, medulloblastoma, and hypo responsiveness to parathormoneCancer 1965 18:89-104. [Google Scholar]
[2]. Stoelinga PJ, Peters JH, van de Staak WJ, Cohen MM Jr, Some new findings in the basal-cell nevus syndromeOral Surg Oral Med Oral Pathol 1973 36:686-92. [Google Scholar]
[3]. Eversole LR, Sabes WR, Rovin S, Aggressive growth and neoplastic potential of odontogenic cysts: with special reference to central epidermoid and mucoepidermoid carcinomasCancer 1975 35:270-82. [Google Scholar]
[4]. Neville BW, Damm DD, Allen CM, Chi A, Oral and Maxillofacial Pathology 2015 First South Asia editionIndiaReed Elsevier [Google Scholar]
[5]. Shear M, Speight P, Cysts of the oral and maxillofacial regions 2008 New YorkJohn Wiley & Sons [Google Scholar]
[6]. Moosvi Z, Tayaar SA, Kumar GS, Neoplastic potential of odontogenic cystsContemp Clin Dent 2011 2:106-09. [Google Scholar]
[7]. Bhushan NS, Rao NM, Navatha M, Kumar BK, ameloblastoma arising from a dentigerous cyst-a case reportJ Clin Diagn Res 2014 8:ZD23-25. [Google Scholar]
[8]. Eversole LR, Leider AS, Hansen LS, Ameloblastomas with pronounced desmoplasiaJ Oral Maxillofac Surg 1984 42:735-40. [Google Scholar]
[9]. Philipsen HP, Ormiston IW, Reichart PA, The desmo- and osteoplastic ameloblastoma. Histologic variant or clinicopathologic entity? Case reportsInt J Oral Maxillofac Surg 1992 21:352-57. [Google Scholar]
[10]. Thompson IO, van Rensburg LJ, Phillips VM, Desmoplastic ameloblastoma: correlative histopathology, radiology and CTMR imagingJ Oral Pathol Med 1996 25:405-10. [Google Scholar]
[11]. Keszler A, Paparella ML, Dominguez FV, Desmoplastic and non-desmoplastic ameloblastoma: a comparative clinicopathological analysisOral Dis 1996 2:228-31. [Google Scholar]
[12]. Barnes L, Eveson JW, Reichart P, Sidransky D, WHO Classification of TumoursPathology and Genetics of Head and Neck Tumours 2005 LyonIARC Press:298-99. [Google Scholar]
[13]. Jaafari-Ashkavandi Z, Geramizadeh B, Ranjbar MA, P63 and Ki-67 Expression in dentigerous cyst and ameloblastomasDent Shiraz Univ Med Sci 2015 16:323-28. [Google Scholar]
[14]. Masloub SM, Abdel-Azim AM, Abd Elhamid ES, CD10 and osteopontin expression in dentigerous cyst and ameloblastomaDiag Pathol 2011 6:44 [Google Scholar]
[15]. Levanat S, Pavelic B, Crnic I, Oreskovic S, Manojlovic S, Involvement of PTCH gene in various noninflammatory cystsJ Mol Med (Berl) 2000 78:140-46. [Google Scholar]
[16]. Pavelic B, Levanat S, Crnic I, Kobler P, Anic I, Manojlovic S, PTCH gene altered in dentigerous cystsJ Oral Pathol Med 2001 30:569-76. [Google Scholar]