Ischemic Heart Disease (IHD) is the leading cause of death worldwide, accounting for 12.7% of global mortality [1]. Acute Coronary Syndrome (ACS) covers a wide spectrum of clinical conditions ranging from unstable angina to Non-ST Elevation Myocardial Infarction (N-STEMI) and ST Elevation Myocardial Infarction (STEMI). Insufficient coronary perfusion due to variable degrees of acute plaque change, thrombosis and vasospasm over chronic, progressive atherosclerotic narrowing of the coronary arteries is the underlying pathology. Although they share this pathophysiology, outcomes and effective management strategies differ by the patient’s place in the spectrum [2]. As new treatments and management guidelines emerge for care of patients with ACS, risk stratification is the cornerstone in the initial evaluation of these patients [2]. Simple prognostic markers that reflect pathophysiologic mechanism may aid in tailoring the therapy to match the intensity of patient’s disease [3].
Oxidative stress and inflammation play vital roles in pathogenesis and progression of Coronary Artery Disease (CAD) leading to Acute Coronary Syndromes (ACS) [4]. Inflammation is implicated in all stages of atherosclerosis, from early lipid deposition to plaque rupture. Inflammation mediates the weakening of fibrous cap and makes the plaque more susceptible to rupture, thus precipitating ACS [5]. A study shows that the inflammatory process persists even after the resolution of clinical symptoms and there is a link between recurrent ischemic episodes and persistent inflammation [6]. Thus, inflammatory biomarkers may provide independent information regarding pathophysiology, prognosis and help in formulating optimal therapeutic strategies.
Myeloperoxidase (MPO) is present in neutrophils and monocytes and is released when they undergo degranulation [7]. It is a marker of inflammation and oxidative stress that has been consistently demonstrated to be elevated in patients with ACS [7]. Several experimental and epidemiologic studies indicate MPO as a candidate marker for clinical application [7]. In addition to being a causative factor for CVD and acute cardio vascular events, MPO can also extend the prognostic information gained from traditional biochemical markers. So, further investigation is needed to expand our knowledge on the prognostic role of MPO in ACS.
Sialic acids are 10 carbon derivatives of neuraminic acid. They comprise the terminal sugar of the oligosaccharide chain in glycolipids and glycoproteins. Studies have shown that serum total sialic acid is elevated in cardiovascular disease; which is attributed to the increase acute phase reactants in these patients [8]. Following acute myocardial infarction, shedding or secretion from damaged cell can lead to elevated sialic acid [9]. Though many studies have evaluated its role in pathogenesis of atherosclerosis its role as a prognostic marker has not been studied.
The purpose of the present study was to analyse the prognostic value of plasma myeloperoxidase and total sialic acid levels in patients with suspected acute coronary syndrome. In samples of patients with suspected ACS, myeloperoxidase and total sialic acid levels were measured and their concentrations were compared with certain prognostic markers of ACS.
Materials and Methods
This prospective study was conducted at the Department of Biochemistry, PSG Institute of Medical Sciences and Research, Coimbatore, India. It was approved by the Institutional Human Ethics Committee (ethical clearance letter no:11/206).
Study subjects
Consecutively admitted patients with chest pain in PSG Hospitals from July 2011 to September 2011 were studied. Patients with cardiac chest pain as shown by ECG changes or elevated cardiac Troponin T levels (> 0.03ng/ml) were included in the study. Patients diagnosed with non cardiac chest pain were excluded from this study. The left over plasma samples (collected in EDTA vial) sent for Troponin T estimation obtained within 24 hours of onset of chest pain were used for analysis.
Clinical variables
The prognostic indicators used in our study were: i) extent of disease (single vessel disease/ multi vessel disease) as indicated by coronary angiogram; ii) incidence of complications like reinfarction, congestive cardiac failure, arrhythmias, renal failure during the hospital stay; iii) Left Ventricular Ejection Fraction (LVEF); and iv) duration of stay in hospital. These were collected by reviewing the case records.
Estimation of biochemical parameters
MPO activity was estimated using o-dianisidine and hydrogen peroxide spectrophotometrically [10]. A 25 μl plasma sample was added to 3 ml of 50 mmol phosphate buffer solution containing 0.167mg/ml of O-dianisidine Hydrochloride (Sigma-aldrich product) and 0.0005% hydrogen peroxide. Reading was taken at 470 nm for 2 min at 250C. One unit (U) of MPO activity was defined as that degrading 1 μmol of hydrogen peroxide per minute at 250C. Sialic acid was estimated using thiobarbituric acid as described by D Aminof spectrophotometrically [11]. A 10μl of plasma sample was treated with strong acid to release sialic acid. To this periodate reagent (25mM in 0.125N H2SO4) was added. Excess periodate reagent was reduced by adding sodium arsenite (2% in 0.5N HCl). To this 2 ml of Thiobarturic acid (0.1M in 1N NaOH solution) was added and incubated for 8 min in boiling water bath. This is cooled in ice and the colored complex formed is extracted by acid butanol (butanol in 5% HCl) and optical density measured at 549 nm.
Statistical Analysis
Continuous variables were expressed as mean and standard deviation. Student t-test and ANOVA were used to compare the means of parametric variables. For variables which did not follow normal distribution, logarithmic transformation was done and parametric tests applied. Discrete variables were expressed in percentage. Chi-square test was used to compare frequency distribution. Two tailed p-value <0.05 was considered to be statistically significant.
Results
The study group included 93 patients, of which 70 were male and 23 were female. Their age ranged from 30-81 years and the mean age of the group was 59 years. Based on the diagnosis, the patients were categorized into three groups namely - patients with stable angina (n=23), patients with unstable angina (n=33) and patients with myocardial infarction (n=37). Patients with myocardial infarction included those with STEMI and N-STEMI. Patient characteristics in these 3 groups are compared in [Table/Fig-1].
Patient characteristic in the study groups.
| Patientcharacteristics | Stableangina(n=23) | Unstableangina(n=33) | Myocardialinfarction(n=37) | p-value |
|---|
| Age in yrs(Mean ± SD) | 58.3 ± 12.8 | 60.8 ± 11.0 | 58.3 ±14.8 | 0.668 |
| Females (n) | 1 (4.3%) | 14 (42.2%) | 8 (21.6%) | 0.004 |
| Diabetesmellitus (n) | 9 (39.1%) | 20 (60.6%) | 20 (51.4%) | 0.286 |
| Hypertension (n) | 14 (60.9%) | 20 (62.5%) | 21 (56.8%) | 0.882 |
| Smokers (n) | 3 (13%) | 2 (6.1%) | 13 (35.1%) | 0.005 |
| Dyslipidemia (n) | 8/11 | 11/21 | 18/32 | 0.525 |
| Ejectionfraction (%) | 59±9.7 | 64±9.8 | 53.9±12.98 | 0.004 |
| Complications (n) | 2 (8.7%) | 4(12.1%) | 8(21.6%) | 0.333 |
| Plasmamyeloperoxidase(U/L) | 53.98±29.53 | 54.45±36.99 | 112.91±57.66 | <0.001‡ |
| Plasma totalsialic acid levels(mmol/L) | 1.35±0.53 | 1.63±0.52 | 1.86±0.46 | 0.007‡ |
‡ after correction for sex, smoker and ejection fraction
Association between plasma myeloperoxidase and sialic acid levels with diagnosis: Plasma MPO levels did not show normal distribution, log transformation was done. Log MPO was distributed normally and parametric tests were applied to log MPO levels. Mean plasma MPO and sialic acid levels were significantly different between patients with stable angina, unstable angina and myocardial infarction [Table/Fig-1]. This difference remained significant even after adjusting for sex, smoking and ejection fraction. Results of Tamhane post-hoc comparisons are shown in [Table/Fig-2]. The plasma MPO levels were significantly elevated in myocardial infarction when compared with angina (stable or unstable). There was no difference in the plasma MPO levels between patients with stable and unstable angina. Plasma sialic acid was elevated in patients with MI when compared to patients with stable angina. But there was no significant difference in its levels between MI and unstable angina patients.
Post-hoc Multiple comparison between plasma myeloperoxidase and sialic acid levels with diagnosis.
| Analyte | Groups compared | p-value |
|---|
| Plasmamyeloperoxidase | Stable angina vs. unstable angina | 0.901 |
| Stable angina vs. Myocardial infarction | <0.001 |
| Unstable angina vs. Myocardial infarction | <0.001 |
| Plasma totalsialic acidlevels | Stable angina vs. unstable angina | 0.177 |
| Stable angina vs. Myocardial infarction | 0.002 |
| Unstable angina vs. Myocardial infarction | 0.165 |
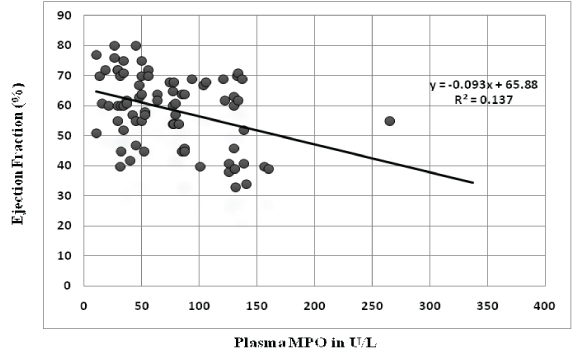
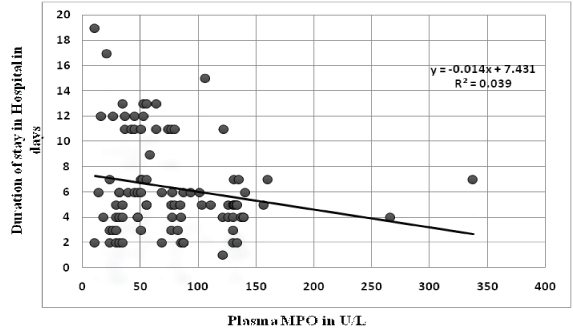
Association of plasma myeloperoxidase with prognostic indicators: There was no difference in the mean plasma MPO of patients with the extent of disease (single vessel disease vs. multivessel disease). But plasma MPO levels were significantly higher in patients who developed complications during their stay in hospital [Table/Fig-3]. These complications included reinfarction, congestive cardiac failure, arrhythmias and acute renal failure. Also plasma MPO levels had significant correlation with ejection fraction (r= -0.365, p-value - 0.001) and duration of stay in hospital(r= -0.224, p-value - 0.031) [Table/Fig-4,5]. When patients were divided into tertile groups based on the plasma myeloperoxidase levels, the mean ejection fraction of the highest tertile group was found to be significantly lower than the lowest tertile group [Table/Fig-6]. But there was no significant difference in the mean duration of stay among the tertile groups.
Association of plasma myeloperoxidase and sialic acid with extent of disease and complications.
| Prognostic indicator | Plasma myeloperoxidase in U/L (Mean ± SD) | p-value | Plasma sialic acid in mmol/L (Mean ± SD) | p-value |
|---|
| Extent of disease |
| Single Vessel Disease (n=20) | 71.38 ± 34.13 | 0.169 | 1.61 ± 0.59 | 0.407 |
| Multi Vessel Disease (n=44) | 62.19 ± 39.79 | 1.50 ± 0.47 |
| Incidence of Complications during the hospital stay |
| Patients without complications (n=79) | 71.15 ± 46.05 | 0.011 | 1.59 ± 0.54 | 0.006 |
| Patients with complication (n=14) | 113.97 ± 74.45 | 2.01 ± 0.38 |
Correlation of plasma MPO levels with ejection fraction.
Correlation of plasma MPO levels with duration of stay in hospital.
Association of plasma myeloperoxidase levels with ejection fraction and duration of stay in hospital.
| Plasma MPOLevels (U/L) | EjectionfractionN=77 | p-value | Mean Durationof Patient inHospital in days | p-value |
|---|
| Lowest TertileGroup (< 47.8) | 62.51±11.22 (29) | - | 7.27 ± 4.79(30) | - |
| Intermediate TertileGroup (47.8 - 84.9) | 59.88±8.7 (25) | 0.345‡ | 6.28 ± 3.69(34) | 0.943‡ |
| Highest TertileGroup (>84.9) | 53.39±13.76 (23) | 0.011‡ | 5.35 ± 2.39 (29) | 0.109‡ |
‡ Lowest tertile group taken as reference
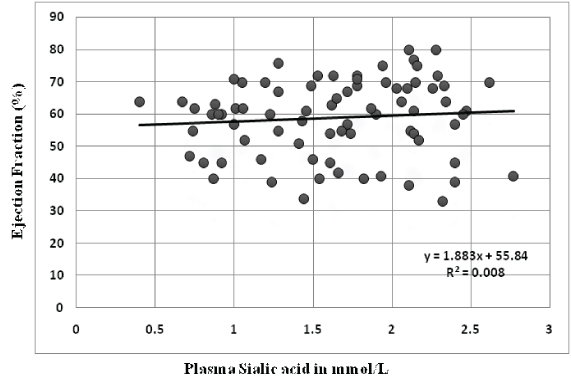
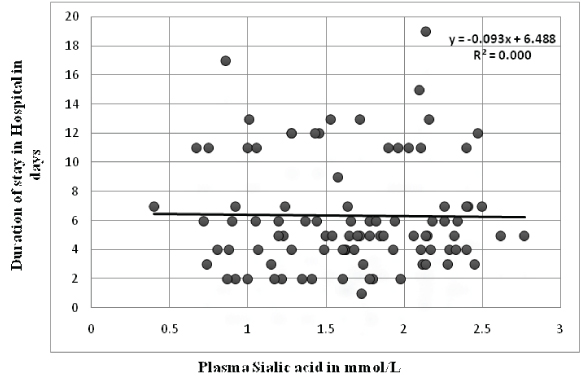
Association of plasma sialic acid levels with prognostic indicators: There was no difference in the mean plasma sialic acid levels of patients with the extent of disease (single vessel disease vs. multivessel disease). But plasma sialic acid levels were significantly higher in patients who developed complications during their stay in hospital [Table/Fig-3]. Unlike plasma MPO, no correlation was observed between plasma sialic acid levels and ejection fraction (r= 0.089, p-value - 0.440) [Table/Fig-7]. Duration of stay in hospital had no significant correlation with plasma sialic acid (r= -0.013, p-value - 0.899) [Table/Fig-8]. When patients were divided into tertile groups based on the plasma sialic acid levels, there was no significant difference in the mean ejection fraction and duration of stay in hospital among the tertile groups [Table/Fig-9].
Correlation of plasma sialic acid levels with ejection fraction.
Correlation of plasma sialic acid levels with duration of stay in hospital.
Association of plasma sialic acid levels with ejection fraction and duration of stay in hospital.
| Plasma sialicacid (mmol/L) | EjectionfractionN=77 | p-value | Mean Durationof Patient inHospital in days | p-value |
|---|
| Lowest TertileGroup (< 1.35) | 58.08±9.9 (25) | – | 6.44 ± 4.1(29) | – |
| Intermediate TertileGroup (1.36-1.93) | 56.8±11.7 (25) | 0.967‡ | 5.71 ± 3.27(32) | 0.833‡ |
| Highest TertileGroup (1.94-2.77) | 61.70±13.28(27) | 0.609‡ | 6.84 ± 3.9 (32) | 0.974‡ |
‡ Lowest tertile group taken as reference
Discussion
Risk stratification is the cornerstone for selection of optimal therapy in patients with ACS [2]. Extent of cardiac damage, extent of coronary artery disease and instability of the disease are the three major determinant of prognosis in ACS [3]. Currently, risk classification is mainly based on assessment of the extent of coronary artery disease and cardiac damage through clinical findings, ECG, angiographic data and biomarkers, including markers of inflammation may increase the prognostic value since these reflect the disease instability.
The current study evaluates the prognostic value of two inflam-matory biomarkers- MPO and total sialic acid in patients with ACS. Plasma myeloperoxidase and sialic acid levels were studied in ninety three patients admitted with complaints of cardiac chest pain. Elevation of these analytes in patients with coronary artery disease when compared to controls is already well established in previous studies [7,12]. The aim of this study was to evaluate the prognostic role of these analytes in patients with ACS, hence their levels in patients without complications were taken as baseline and compared with those with complications. Higher levels of plasma MPO and sialic acid at admission were associated with development of complications during their stay in hospital. Plasma MPO levels at admission correlated negatively with ejection fraction in patients with ACS.
Myeloperoxidase and ACS prognosis
MPO, a haem containing microcidal enzyme, is the principal mediator secreted on activation of neutrophils. Few prospective studies have shown an association between circulating MPO levels and prognosis after ACS. Baldus in his prospective study followed 1090 patients with ACS for 6 months and showed that patients with elevated serum MPO levels had increased risk for subsequent non fatal MI at 72 hours, 6 weeks and 6 months [13]. High plasma MPO levels were also associated with higher risk for major adverse cardiovascular events after acute STEMI at 2-year follow-up [14]. In yet another long term follow-up study of 512 patients with MI, Mocatta et al., concluded that above median MPO level was associated with higher risk for mortality [15]. Brennan et al., concluded that, in troponin T negative patients, plasma myeloperoxidase in patients with chest pain independently predicts the early risk of myocardial infarction, as well as the risk of major adverse cardiac events in the ensuing 30-day and 6-month periods [16]. In agreement with these studies, our study shows that patients who developed complications during their stay in hospital following ACS had significantly higher plasma myeloperoxidase levels when compared to the ACS patients who did not develop complications.
We found left ventricular ejection fraction correlated negatively with plasma MPO levels. Patients in the highest tertile plasma MPO level group had significantly lower ejection fraction when compared with lowest tertile group. Similar results were also noted by Kaya et al., in their prospective study including 73 patients [14]. They found significantly lower ejection fraction in patients with higher MPO levels. Chang et al., also noted a lower LVEF in patients with higher MPO levels [17].
In the present study, MPO levels did not correlate with troponin T which was also seen in the study conducted by Baldus [13]. In their study of 1090 patients, MPO levels were not significantly different between patients with Cardiac troponin T levels <0.01 and >0.01μg/L. They concluded that MPO release actually precedes myocardial injury and that MPO elevation identifies patients with unstable atherosclerotic plaque formation even before complete microvascular obstruction. Studies [14,17] have shown an inconsistent association between the extent of disease and MPO levels, we found no association between the two.
MPO is a pro-oxidant enzyme and it favors oxidation of LDL promoting atherogenesis [18]. It is also implicated in the development of endothelial dysfunction, by consuming nitric oxide and limiting its bioavailability [19]. Increased MPO expression and presence of hypochlorous acid (HOCl) modified proteins were seen in atherosclerotic lesions [20]. Low doses of HOCl led to endothelial cell activation and increases thrombogenicity. High doses of MPO triggered endothelial cell death by apoptosis and desquamation. Endothelial death leads to plaque erosion and exposure of prothrombogenic surface contributing to plaque vulnerability [21]. MPO also oxidatively inactivates protease inhibitors and activates matrix metalloproteinases. Matrix metalloproteinases are capable of degrading the extracellular matrix of fibrous layer and favour plaque rupture [22].
Sialic acid levels and ACS prognosis
Sialic acid is associated with atherosclerosis independent of other cardiovascular risk factors [23]. In a large prospective study, high serum sialic acid level was found to be a strong predictor of cardiovascular mortality [24]. In a more recent 17 year follow up study elevated serum sialic acid was found to be predictor of cardiovascular event in apparently healthy individuals [25]. Serum total sialic acid levels were high in patients with acute coronary syndrome when compared to the healthy controls [26]. Serdar et al., found that total sialic acid levels were progressively increasing in unstable angina, N-STEMI and STEMI groups and this difference was statistically significant [26]. Similar progressive increase in sialic acid levels was also seen in our study, between stable angina, unstable angina and MI patients but on post-hoc analysis the difference was statistically significant only between stable angina and MI group. Gokmen et al., showed that total sialic acid level was significantly different between patients with single vessel disease and multi vessel disease [27]. However, we were unable to duplicate this in our study.
Serum sialic acid levels are raised in inflammation as an innate immune system response. Increased production of sialated acute phase reactants increases the sialic acid levels [8]. It is hypothesized that following ACS its level increases either due to secretion or shedding from the cell or cell membrane [9]. It is a potential marker in assessing myocardial cell damage in patients undergoing cardiac surgery [28]. Its prognostic predictive value following ACS has not been studied so far. Our study shows that total sialic acid levels are high in patients with ACS who develop complications during their stay in hospital. Sialic acid levels did not correlate with troponin T levels and left ventricular ejection fraction in our study.
Limitation
Myeloperoxidase can be either estimated as mass or activity. Since estimation of activity is done in saturating amounts of substrates it can be taken as a proxy estimation of mass [7]. O-dianisidine- hydrogen peroxide method can have interference due to ceruloplasmin or haemoglobin levels. This was partly overcome by excluding haemolysed samples from analysis. Convenient sampling method was chosen due to time constraint and hence sample size was restricted to 93.
Conclusion
Myeloperoxidase and sialic acid are markers of inflammation and elevated following acute coronary syndrome. Patients who developed complications following ACS during their stay in hospital had higher levels of plasma MPO and sialic acid at admission. MPO levels correlated negatively with left ventricular ejection fraction. Their role as predictors of in-hospital risk in patients with ACS needs to be explored further.
‡ after correction for sex, smoker and ejection fraction
‡ Lowest tertile group taken as reference
‡ Lowest tertile group taken as reference