Idiopathic Guttate Hypomelanosis (IGH) is a common benign dermatosis which was first described in 1951 by Costa [1] as “symmetric progressive leukopathy” of the extremities which is characterized by discrete, round to oval porcelain-white macules of approximately 2-6 mm in diameter, which increase in number with aging. A familial tendency to develop IGH has been reported [2]. Histopathological section of lesion stained with the Fontana- Masson shows markedly reduced the melanin content in lesional skin and there is either a significant reduction or an absence of dopa-positive melanocytes [3].
Various treatment options like topical application of corticosteriods, retinoid [4], tacrolimus [5], pimecrolimus [6] and minor procedure like dermabrasion [7], cryotherapy [8] & minigrafting [9] have yielded good results in IGH. It was noted that therapeutic wounding stimulates melanocytes from the periphery and surrounding hair follicles to proliferate, migrate, and repigment the lesions [10]. Phenol or carbolic acid is an antiseptic and antipruritic agents that also act as local anaesthetic. Full strength phenol (88%) is used as a medium depth chemical peel for facial rejuvenation and also used as a spot peel for treating stable vitiligo and alopecia areata [11].
Similarly, Placental extract is a known treatment modality for vitiligo lesions, used topically or parentrally along with photo exposure [12]. It is claimed that the placental extracts have a mitogenic effect on the skin melanocytes and stimulates melanogenesis in surviving melanocytes [13].
The present study was designed with an aim to find out the efficacy of placental extracts when used with 88% phenol for the treatment of IGH macules.
Materials and Methods
The study was designed as a follow up case control study, conducted over a period of one year from December 2015 to December 2016 in Dermatology, Venereology & Leprosy department of Geetanjali Medical College and Hospital, Udaipur, Rajasthan, India, after obtaining the permission of Institutional Ethical Committee.
Sample
The sample size was calculated on the basis of a pilot study conducted in our outpatient department, where the prevelance of cases with IGH was 10% of all the dermatological diseases. Sample size (n) was calculated as = 4pq/ L 2; where p is prevalence = 10%, q = 110-10 = 90 and L = 10% (maximum allowable error). Hence, the sample size was calculated as 36. We have recruited 40 patients in this study, which were randomly divided into two groups (n=20) in the alternate patients of IGH and were recruited for the study in the two groups), viz group P, (the control group treated with only 88% phenol) and group PP (study group treated with Placental extracts along with 88% phenol). An informed written consent was obtained, for treatment and participation in the study was obtained from all the patients before starting the procedure.
Patients with any of the following conditions were excluded from the study:
- Active infection (systemic or cutaneous)
- Keloidal tendencies
- Bleeding disorders
- Progressive vitiligo
- Post traumatic atrophic lesions
- Non compliant patients
A detailed physical examination was done for all the patients and pre-treatment photographs were taken for record. No oral medicine was used during the treatment. It was also ensured that none of the subjects had any previous skin lesion. The patients from group P were treated with spot peel of 88% phenol on the hypo pigmented lesion whereas in group PP, they were advised to apply placental extracts (commercially available as Placentrex gel) at night for 3 months after the spot treatment with 88% phenol as in group P.
After cleaning the area with spirit, 0.5ml phenol was applied with the help of cotton tipped applicator on lesions of IGH. Feathering of the border was done to cover 0.5 to 1mm of peripheral normal skin and uniform white frost appeared within few seconds. All the patients were advised to apply topical 2% fusidic acid cream, twice daily for 5 to 7 days. Patients were asked to expose the treated area to sunlight for 3-5 min every day after the healing of the lesion. Almost all the patients complained of immediate mild burning sensation for 2-3 min after application of phenol which was followed by severe burning sensation in some of them. The uniform frost disappeared in 30 minutes, followed by erythema and dark brown to black discoloration of the lesions with crusting in 24 hours. The skin peeled off in next 15 to 20 days leaving behind erythema. The procedure was repeated after 1 month, if required.
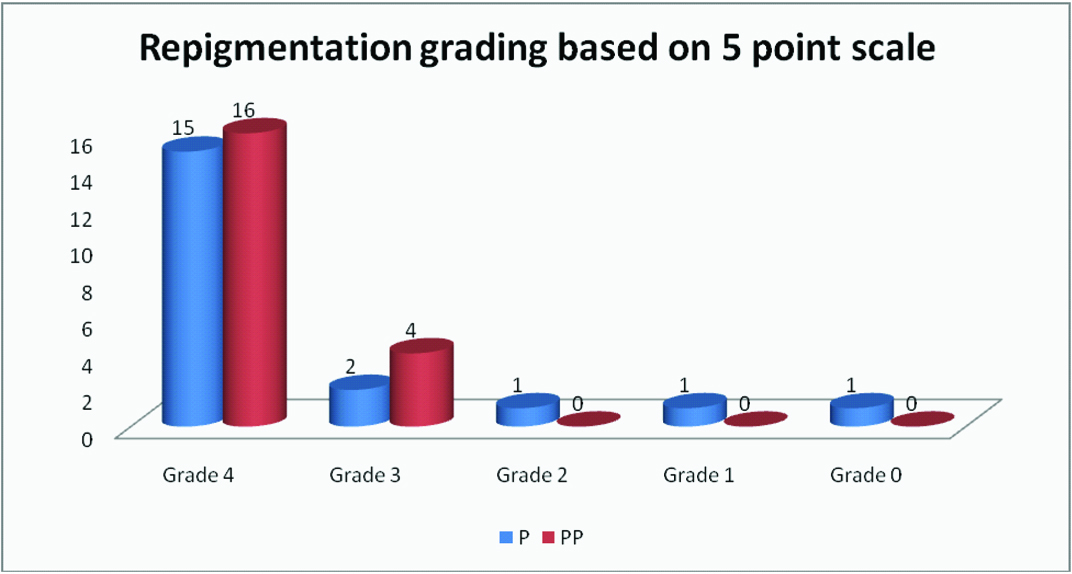
All the patients were reassessed at the end of 3 months. Re-pigmentation was noted in all the patients between 4 and 6 weeks. The response to treatment was assessed on the basis of re-pigmentation which was based on five point grading system ranging from G0 to G4
Grade 0-No re-pigmentation
Grade 1-Up to 25% re-pigmentation
Grade 2-25-50% re-pigmentation
Grade 3-50-75% re-pigmentation
Grade 4-Up to or more than 75% re-pigmentation.
The statistical analyses of the results were performed using percentage decrease in lesions and Chi-square test, z-test and p-value <0.05 was considered as significant.
Results
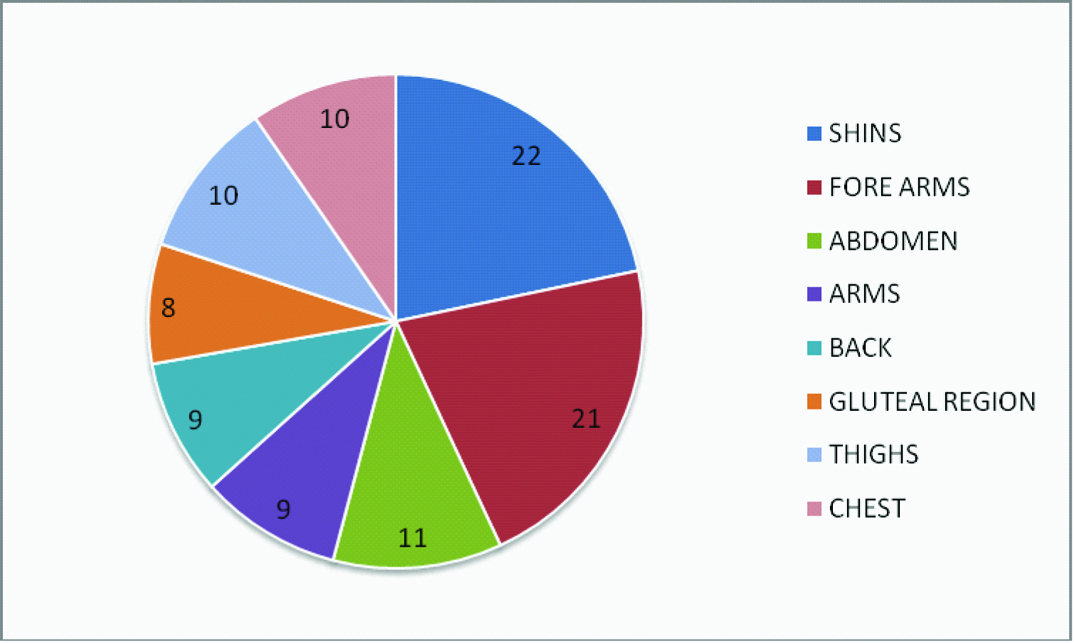
In this study, the sample population was randomly divided into group P and PP, where the mean age was 43.6 ± 12.5 years and 42.05 ± 11.4 years, respectively. It was observed that 80% of the participants were females while only 20% were males which were equally divided among both the groups. The macules of IGH were found to have some predilection for forearm (28.8%, 28.06%) followed by shin (14.79%, 14.39%) in group P and group PP, respectively as compared to other sites as depicted in [Table/Fig-1]. Most of the individuals had more than two sites of involvement.
Showing distribution of IGH lesions in percentage on different sites (both group P and PP).
At the end of three months, the improvement in the lesions was observed using 5 point grading system. The number of IGH lesions were recorded and found out to be significantly decreased in both the groups, after the treatment [Table/Fig-2,3].
Pretreatment & postreatment photograph of group P.

Pretreatment and postreatment photo of group PP.

It was observed that 76 .8% lesions responded in Group P where as 79.1% lesions responded in group PP [Table/Fig-4].
Comparison of different parameters of IGH lesions in group P and in group PP, before and after treatment.
| Statistics of the IGH lesions, before and after treatment | Group P(n= 20) | Group PP(n=20) |
|---|
| Average number of lesions before treatment | 142 | 139 |
| No. of lesions treated | 109 | 110 |
| Non responding lesions (lesions remaining after treatment) | 33 | 29 |
| Percentage decrease in lesions in response to treatment | 76.8 | 79.1 |
But there was a non-significant (p=0.8203) difference in response to the treatment in group PP as compared to group P. However, the percentage of lesions responding to treatment in group PP was apparently better than group P [Table/Figure-5].
Percentage of lesions responding to treatment in group PP was apparently better than group P.
Discussion
In the present study, the mean age of patients having IGH lesions was 43.6 ± 12.5 years and 42.05 ±11.4 years in group P and group PP respectively, which corroborates with the literature, where it is stated as the disease of the people beyond 40 years of age. Excessive exposure to the sun appears to be the main etiology behind the hypo-pigmented macules and the incidence of these lesions have been reported to be over 60% in middle aged people [2,15,16]. Although it is a disease of middle and old aged, it might affect the young adults in their 20’s [15,17].
The sample recruited in both the groups comprised of 32 females and 8 males. Though these lesions have an equal incidence in both the sexes [3], but the higher number of females in our study could be attributed due to the cosmetic awareness among them. We had also observed, that the IGH lesions were mainly distributed on extremities like forearms (22%) and shin (21%) which could be explained by the exposure to the sun or repeated trauma on exposed surfaces of the limbs [Table/Fig-1]. High incidence of lesions on the anterior surface of tibia was observed in patients using body scrubs clearly showing the repeated irritation of vulnerable body parts might essentially contribute to the formation of IGH lesion [2,18].
In the pretreatment stage, we observed that average numberof lesions per patient in group P and PP were 7.1 ± 4.32 and 6.95 ± 4.30, respectively. However, the average number of lesions responding to respective treatment modalities in both the groups; were observed as 5.45 ± 3.81 and 5.5 ± 2.95 indicating 76.76% and 79.14% decrease in lesions in group P and PP respectively. Though the percentage improvement in the lesions wassignificantly high in both the groups, as compared to pre treatment levels; but was non- significant (p=0.8203) between the groups [Table/Fig-4].
Further, it was observed that 80% patients (n=16) of group PP had shown grade 4 re-pigmentation as compared to 75% in group P (n=15). Another significant observation was that, 15% of patients of group P had non responding lesions with less than grade 2 re-pigmentation, whereas none of the patients from group PP had refractory lesions [Table/Fig-4]. This finding though statistically non-significant, but is quite encouraging to use placental extracts as a potent adjuvant.
Various studies, till date, have reported different therapeutic modalities, for the management of IGH, but the response has been variable from one individual to the other. Usually, therapeutic wounding using dermabrasion [7,18,19], laser ablation [20], cryosurgery [8] (liquid nitrogen spray), needling [20], and local application of phenol [12] or trichloroacetic acid are reserved for cosmetically sensitive sites with stable vitiligo, those are refractory to medical treatment. In these procedures, the treatment modality produces the iatrogenic wound followed by the tissue response as inflammation and healing. As a result, the re-epithelialization takes place from the remnants of dermal appendages and stimulate the follicular and perilesional melanocytes through liberation of cytokines like leukotrienes C4, D4, Transforming Growth Factor-Alpha (TGF-α), interleukin-1 and endothelin-1 to induce perifollicular and perilesional pigmentation [7,9,10].
Savant has also reported therapeutic wounding with 88% phenol in cases of stable vitiligo, followed by post inflammatory hyperpigmentation [11]. Similarly, Ravikiran SP et al., used 88% of phenol in IGH lesions and reported 75% improvement in 45%of their patients [14].
Topical Placental extracts along with exposure to sunlight had been used for induction of re-pigmentation in the lesions of vitiligo. It is thought to induce adhesion, spread and motility of melanocytes, secondary to the stimulatory effect on the dopa-negative, amelanotic melanocytes in the outer hair-root sheaths of hair follicles of amelanotic patches through glycosphingolipids and endothelins present in the extract [12].
It was also observed that the group P had shown persistent scabbing for more than 15 days as compared to group PP, where the scabbing period was less. Other side effects like ulceration, secondary infection and scarring and post inflammatory pigmentation were also less in group PP. This resulted in better patient satisfaction in group PP.
Limitation
Small sample size and lack of control group are the limitations of our study. Larger studies with control group are required in the future.
Conclusion
The clinical and patient acceptability of phenol along with the placental extracts as an adjuvant was better with similar results. Hence, the use of placental extract is recommended along with phenol in IGH lesions.