Palato-Radicular Groove: A Rare Entity in Maxillary Central Incisor Leading To Endo-Perio Lesion
Priyadarshini Prasad Nadig1, Isha Suresh Kumar Agrawal2, Vineet Suresh Kumar Agrawal3, Sarath Chandran Srinivasan4
1 Reader, Department of Periodontics and Implantology, Manubhai Patel Dental College and Hospital, Vadodara, Gujarat, India.
2 Postgraduate Student, Department of Periodontics and Implantology, Manubhai Patel Dental College and Hospital, Vadodara, Gujarat, India.
3 Reader, Department of Conservative and Endodontics, Manubhai Patel Dental College and Hospital, Vadodara, Gujarat, India.
4 Head of Department, Department of Periodontics and Implantology, Manubhai Patel Dental College and Hospital, Vadodara, Gujarat, India.
NAME, ADDRESS, E-MAIL ID OF THE CORRESPONDING AUTHOR: Dr. Priyadarshini Prasad Nadig, Reader, Department of Periodontics and Implantology, Manubhai Patel Dental College and Hospital, Vishwa Jyothi Ashram, Nr. Vishwamitri Bridge, Munjmahuda, Vadodara-390011, Gujarat, India.
E-mail: priya_darshinikud@rediffmail.com
Allograft, Biodentine, Discoloration, Localized periodontitis, PRF, Sausarization
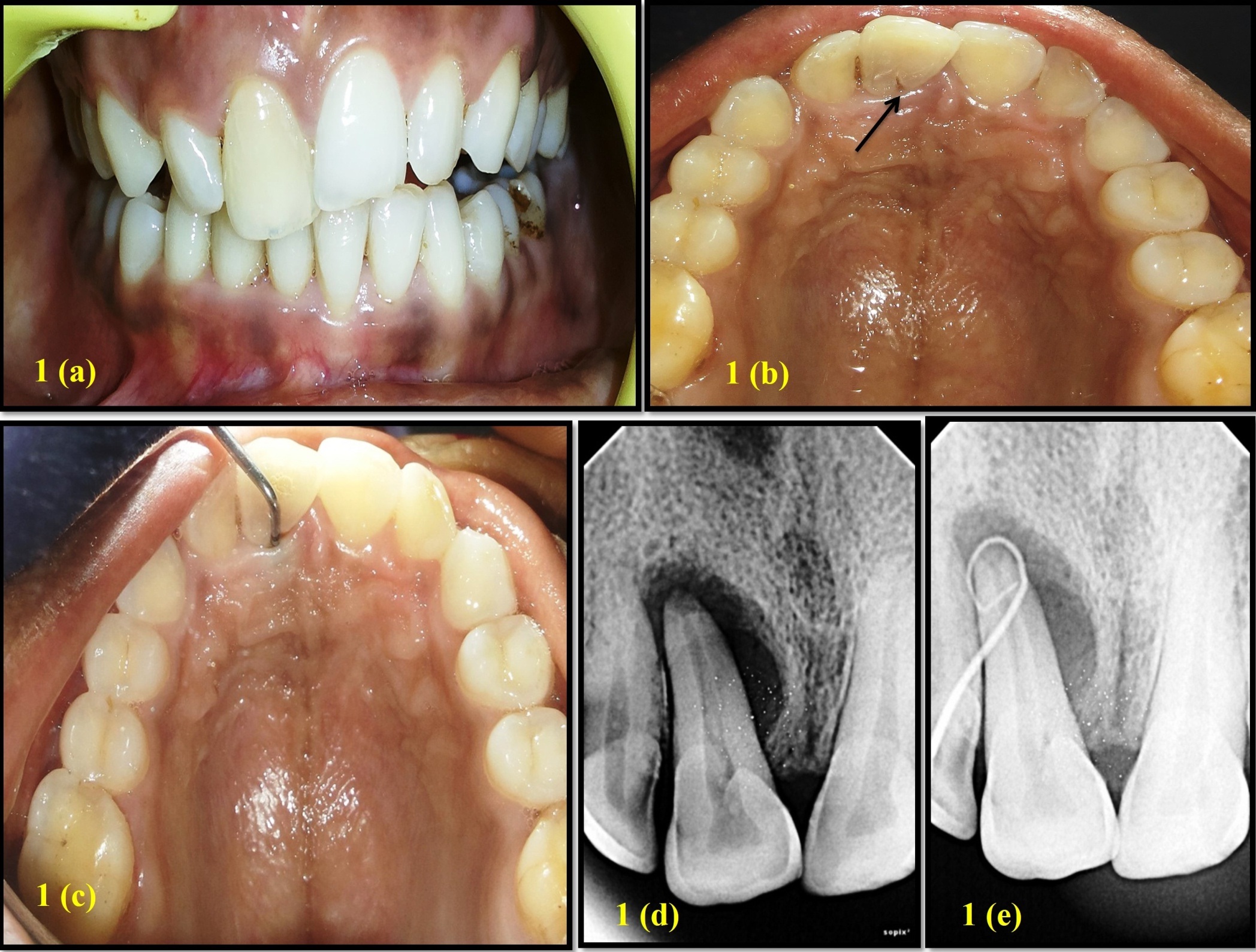
A 35-year-old female patient reported with chief complaint of discoloration and pus drainage from the upper right central incisor. On examination, brownish discoloration of teeth 11 was seen and a draining sinus tract was present on the labial alveolar mucosa [Table/Fig-1a]. A deep periodontal pocket of 13mm was found on the palatal aspect of 11 and a Palato Radicular Groove (PRG) extending into the gingival sulcus was identified [Table/Fig-1b,1c]. Periapical radiographs revealed an extensive peri-radicular radiolucency involving the apical and middle one-third of the mesial aspect of root [Table/Fig-1d]. The tooth tested negative to pulp vitality. The facial sinus tract was traceable with a gutta-percha cone to the periapical radiolucency [Table/Fig-1e]. Based on clinical and radiographic findings, it was concluded that tooth 11 having a PRG predisposed to the development of localized periodontitis with secondary endodontic involvement.
(a): Preoperative clinical photograph (labial view). (b): Preoperative clinical photograph (palatal view). (c): Periodontal pocket depth of 13mm on palatal aspect. (d): Preoperative radiograph. (e): Radiograph showing gutta-percha tracing.
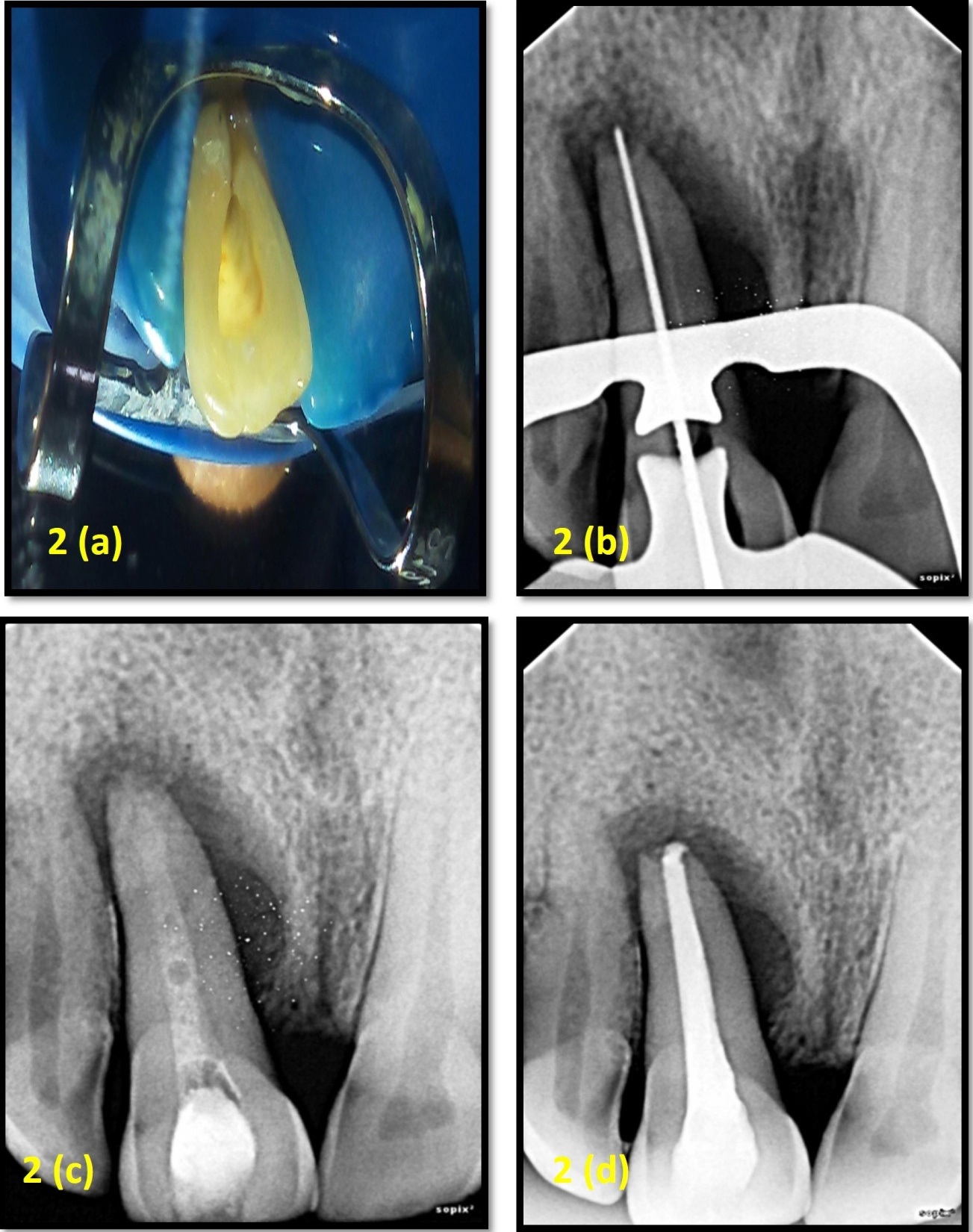
First, periodontal Phase I therapy was performed. In the endodontic phase of therapy, the tooth was accessed under rubber dam isolation [Table/Fig-2a,2b]. The canal was cleaned, shaped and was filled with calcium hydroxide paste (UltraCal XS, Ultradent Products Inc., South Jordan, UT) [Table/Fig-2c]. After three weeks, root canal was obturated with gutta-percha and sealer (AH Plus, Dentsply Maillefer, Tulsa, OK) [Table/Fig-2d].
(a): Access opening under rubber dam. (b): Working length determined using electronic apex locator and radiograph. (c): Radiograph after calcium hydroxide dressing. (d): After obturation radiograph.
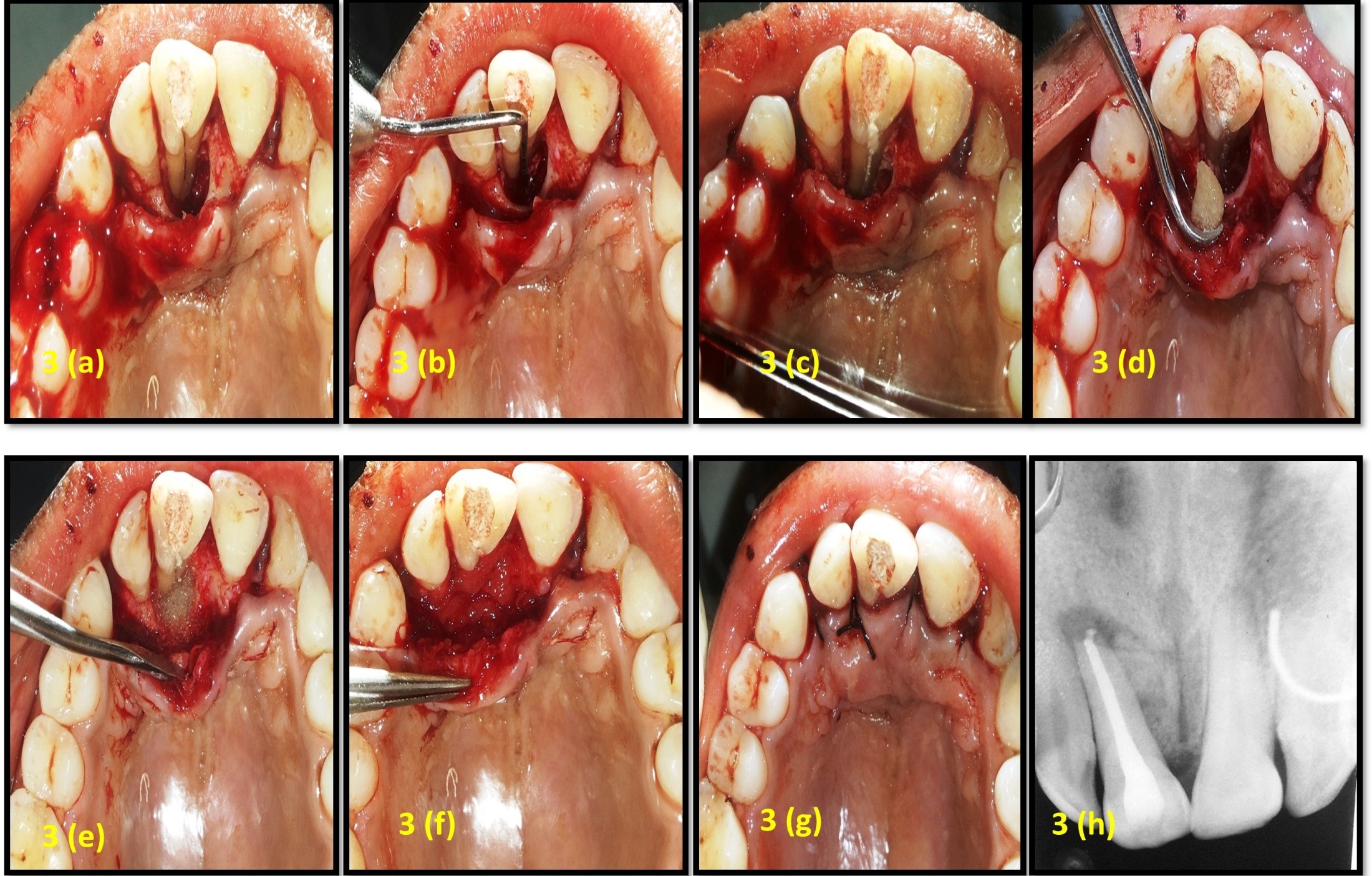
After eight weeks of endodontic therapy, periodontal flap surgery was carried out. Under local anesthesia a full thickness periosteal flap was raised and after complete debridement of granulation tissue, a deep combined bony defect was seen [Table/Fig-3a], PRG was seen to extend over the entire length of root [Table/Fig-3b]. Preparation of groove was done with high-speed round diamond and was blended smoothly with the adjoining surface. The entire length of the groove was then sealed completely with Biodentine™ (Septodont, St. Maurdes Fosses, France) [Table/Fig-3c]. Following which the bony defect was filled with bioactive synthetic bone allograft (Novabone Putty, NovaBone Products, LLC) in incremental manner [Table/Fig-3d,3e] and autologous PRF was placed over the graft like a membrane [Table/Fig-3f]. The flaps were secured with 4-0 mersilk sutures (Ethicon, Johnson and Johnson Ltd., Baddi, H.P., India) [Table/Fig-3g] and post-surgical radiograph was taken [Table/Fig-3h]. Post-operative instructions and medication were prescribed to the patient. The patient was asymptomatic post-operatively and sutures were removed after seven days. The patient was recalled at three months, six months and one year. Phase I therapy was carried at each appointment with reinforcement of oral hygiene. At one year follow-up, clinically, there was a reduction in the pocket depth from 13mm to 4mm [Table/Fig-4a]. Radiographically, there is significant reduction in radiolucency around the root surface [Table/Fig-4b].
(a): Bony defect on palatal aspect after flap elevation; (b): Extension of PRG on entire length of root; (c): Sealing of groove with biodentine; (d): Placement of bioactive synthetic bone allograft material; (e): Bioactive synthetic bone allograft material placed in defect; (f): Placement of PRF; (g): After suture placement; (h): Post-surgical radiograph.
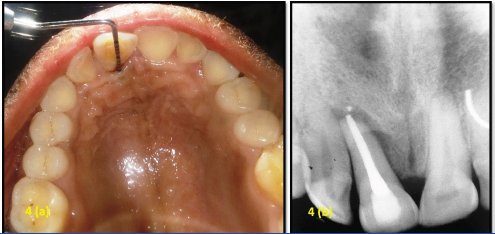
(a): Reduced pocket depth from 13mm to 4mm after one year; (b): One year follow-up radiograph.
Discussion
A complicated/Type III PRG existence on maxillary central incisor in our case was apparent on clinical probing, confirmed later on reflection of the flap. PRG acts as nidus for accumulation of bacterial bio-films leading to periodontal destruction [1]. This may also bring about pulpal changes through the accessory canals enabling the bacteria to move into pulp space. Hence, PRG leads to development of localized combined Endodontic-Periodontal lesion requiring both endodontic and periodontal therapies for tooth retention. In our case also, area of periodontal destruction followed the groove leading to tear drop shaped radiolucency which was evident on radiograph. Many materials can be used for sealing of PRG such as glass ionomer cement, composites, MTA and Emdogain. In the present case we have used biodentine due to its excellent mechanical properties, superior handling characteristic and good bicompatability [2]. Also, biodentine has a reduced setting time i.e., few minutes compared to several hours of MTA and has better marginal sealing properties, plus requires no surface conditioning and bonding making it easy to handle. For periodontal regeneration PRF and bioactive synthetic bone allograft was used as PRF is a complex matrix of autologous fibrin which releases growth factors over time (7-10 days) [3]. PRF has also been shown to stimulate the growth of osteoblasts and periodontal ligament cells, both of which are significant for the periodontal regeneration [4]. Also, PRF is completely an autologous product; there are no chances of disease transmission and graft rejection. Novabone Putty was used to fill the osseous defect because it is bioactive glass graft having good handling, hemostatic and osteoconductive properties and act as a barrier retarding epithelial down growth [5]. In addition Biodentine also aids in bone deposition by releasing alkaline phosphatase [6].
The combined endo-perio interdisciplinary treatment approach employed was successful as it can be observed that one year post-surgery, the sinus tract had resolved, decrease in pocket depth from 13mm to 4mm and significant radiographic evidence of bone fill is achieved.
[1]. Naik M, de Ataide Id, Fernandes M, Lambor R, Treatment of combined endodontic: periodontic lesion by sealing of palato-radicular groove using biodentineJ Conserv Dent 2014 17:594-97. [Google Scholar]
[2]. Johns DA, Shivashankar VY, Shobha K, Johns M, An innovative approach in the management of palatogingival groove using Biodentine TM and platelet-rich fibrin membraneJ Conserv Dent 2014 17:75-79. [Google Scholar]
[3]. DohanEhrenfest DM, de Peppo GM, Doglioli P, Sammartino G, Slow release of growth factors and thrombospondin-1 in Choukroun’s platelet-rich fibrin (PRF): A gold standard to achieve for all surgical platelet concentrates technologiesGrowth Factors 2009 27:63-69. [Google Scholar]
[4]. Blair P, Flaumenhaft R, Platelet alpha-granules: basic biology and clinical correlatesBlood Rev 2009 23:177-89. [Google Scholar]
[5]. Grover V, Kapoor A, Malhotra R, Uppal RS, Evaluation of the efficacy of a bioactive synthetic graft material in the treatment of intrabony periodontal defectsJ Indian Soc Periodontol 2013 17(1):104-10. [Google Scholar]
[6]. Weiwei P, Weining L, Long J, Lifen Li, Jiang C, Yqin Z, Effect of tricalcium silicate on proliferation and odontogenic differentiation of human dental pulp cellsJ Endod 2011 37:1240-46. [Google Scholar]