AUB can be defined as alteration in the volume, pattern, and/or duration of menstrual blood flow. It is most common cause of gynaecologic referral [1,2]. In June 2011 International Federation of Gynecology and Obstetrics (FIGO) gave PALM -COEIN classification for non-gravid women in reproductive age groups. The classification system is divided into nine categories and is arranged according to the acronym PALM-COEIN meaning Polyp, Adenomyosis, Leiomyoma, Malignancy and hyperplasia, Coagulopathy, Ovulatory Disorders, Endometrium, Iatrogenic, and Not Classified respectively [3]. Causes of the PALM group are structural and can be diagnosed by imaging techniques, and/or by histopathology and COEIN group contains entities which cannot be defined by imaging or histopathology alone [4].
Other investigations and proper clinical history will help in further classifying the COEIN group. AUB-E is diagnosis of exclusion and at present no specific test is available to diagnose this group [4]. AUB-E (DUB) is mostly due to any one of these mechanisms-estrogen breakthrough bleeding, estrogen withdrawal bleeding and progesterone breakthrough bleeding [5]. Estrogen and progesterone exert their effect by acting on specific nuclear receptor proteins, Estrogen Receptor (ER) and Progesterone Receptor (PR). These receptors are present in endometrial stromal and glandular cells [6]. Estrogen (ER) and Progesterone (PR) receptors are placed in nuclear steroid receptor superfamily [7]. Estrogen and progesterone mediate their effect through intra-and extranuclear receptors. ER exists in 2 main forms, ER-α and ER-β [8]. PR receptor occurs in PR A and PR B [6].
IHC is beneficial because of tissue localization and aids in assessing tissue distribution and intensity in glandular and stromal cells [9]. IHC may be a useful investigation which can be used along with pelvic ultrasound and histopathology of endometrial biopsies in management of AUB-E in reproductive age group. Treatment of AUB includes both medical therapies and surgical procedures [10]. Surgical treatment should be used for patients who are clinically unstable, not suitable for medical management, or have inappropriate response to medical therapy [11]. Present study was undertaken with an aim to classify patients presenting with AUB according to FIGO/PALM-COEIN classification into structural (PALM) and AUB-E group and to evaluate AUB-E group using ERα and PR expression to find if evidence based hormonal treatment can be used to prevent patient from undergoing unnecessary surgical interventions.
Materials and Methods
An ethical approval was obtained from the institute and the cross sectional study was conducted in patients presenting with AUB in reproductive age group (15-45 years), for the period between July 2014- August 2015. Six hundred endometrial samples were evaluated. Out of these, 447 were from hysterectomy specimen and 153 were endometrial biopsies. Out of these 50 cases of endometrial biopsies were of pregnancy and pregnancy related complications. These cases were excluded from the study as FIGO/PALM-COEIN classification includes only non-pregnant females and 550 cases were classified as per FIGO classification of AUB in non-gravid women in reproductive age group [3].
Females of age group 15-45 presenting with abnormal uterine bleeding were included. Any female presenting with systemic diseases like diabetes mellitus, hypertension, chronic liver and kidney disease and organic genital tract lesion were excluded from present study. Patients presenting with mental illness were also excluded.
For IHC 20 control samples were taken from hysterectomy specimen of similar age group presenting with vaginal prolapse.
Hysterectomy specimen or endometrial tissue were stained with H&E and examined thoroughly to note the histopathological details and classified as per FIGO/PALM-COEIN classification into Structural (PALM) and AUB-E group. IHC for quantification of ERα and PR expression was carried out in AUB-E (100 cases) and control group endometrium (20 cases) due to technical constraints. On histopathological examination in control group either proliferative phase (10 cases) or secretory phase (10 cases) endometrium was seen, whereas in AUB-E group, features of Proliferative endometrium (26 cases), secretory endometrium (32 cases), disordered proliferative endometrium (28 cases) and hyperplasia without atypia (14 cases) were seen. Primary antibody used for ERα was Monoclonal mouse Anti- Estrogen Receptor clone ID5 (BioGenex, Fremont CA) and for PR was Monoclonal mouse Anti-Progesterone Receptor clone PR88 (BioGenex, Fremont CA). A case was considered positive when brown colouration of the nucleus was observed. Expression of ERα and PR receptors was positive in endometrial lining as well as in stroma The intensity and distribution of ERα, PR was evaluated using a semi-quantitative method (IRS-score) [12]. The IRS score was calculated as follows: IRS=SI x PP, where SI is the optical staining intensity (graded as 0 = no, 1 = weak, 2 = moderate and 3= strong staining) and PP the percentage of positive-stained cells. The PP was estimated by counting approximately 200 cells and it was defined as 0 = no staining, 1 = <10%, 2 = 11-50%, 3 = 51-80% and 4 = >81.9 [Table/Fig-1,2]. Unpaired student t’ test was performed. P-value ≤ 0.05 was taken as critical level of significance.
AUB-E endometrium secretory phase, ER α-glands 3x4 and stroma 3x3 (IHC- 40x).
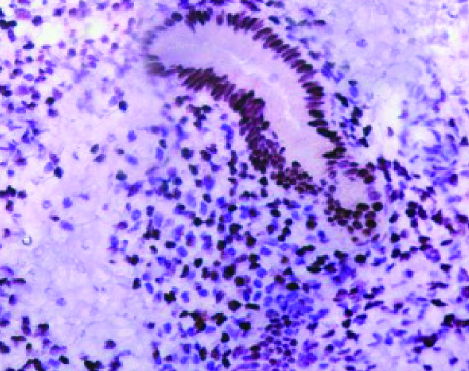
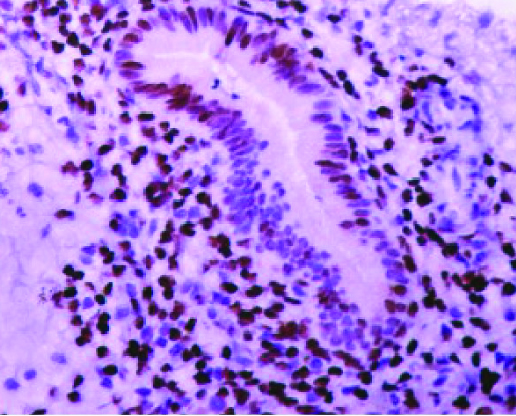
AUB-E endometrium secretory phase, PR- glands 3x3 and stroma 3 x 4 (IHC- 40x).
Results
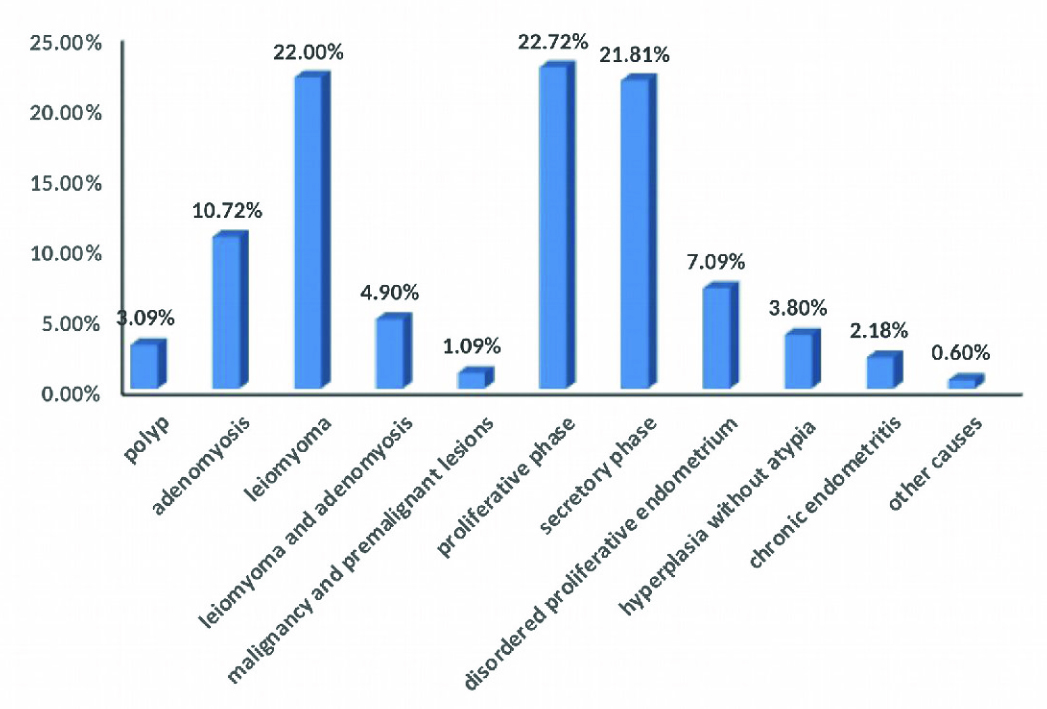
Most of the cases presenting with AUB were of 36-45 years of age which accounted for 74.73%. Mean age of presentation was 37.89 years. Most of the patients presented with heavy menstrual bleeding (84%), irregular menstrual bleeding (12%) followed by prolonged menstrual bleeding (4%). 41.81% cases (230) comprised of structural causes (PALM) and remaining 58.19% (320) were endometrial (AUB-E) causes cases. Most common cause for AUB taking into account both the structural as well as non-structural causes was AUB –E with Proliferative endometrium (22.72%) followed by AUB-L (Leiomyoma) accounting for 22%. Third most common cause was AUB-E with Secretory endometrium accounting for 21.81% [Table/Fig-3].
Histological pattern of cases presenting with Abnormal uterine bleeding
AUB-E group when compared with control group showed statistically significant difference for ERα and PR receptor [Table/Fig-4]. When cases of control group showing proliferative phase (10 cases) were compared with AUB –E Proliferative endometrium/non secretory endometrium –Proliferative endometrium (26 cases), hyperplasia without atypia (14 cases) and AUB –E disordered proliferative endometrium (28 cases) increased receptor expression was noted. In AUB-E proliferative endometrium and AUB-E hyperplasia without atypia group difference was found to be statistically significant for both ERα and PR expression [Table/Fig-5,6]. However, AUB–E disordered proliferative endometrium (28 cases) showed statistically significant expression in stroma for PR and total score just touched level of statistical significance (p=.05). ERα expression was not found to be statistically significant [Table/Fig-7].
Comparison of ERα and PR expression scores in control and AUB-E group.
| ER a | Control (n = 20) | AUB-E (n = 100) | p-value |
|---|
| Gland | 3.6(2.85) | 6.86(3.89) | 0.0005 |
| Stroma | 1.95(1.85) | 5.27(3.92) | 0.0003 |
| Total score | 5.55(3.65) | 12.12(7.0) | 0.0001 |
| PR | Control (n =20) | AUB- E (n =100) | p-value |
| Gland | 4.5(3.12) | 6.61(4.15) | 0.0334 |
| Stroma | 2.6(2.69) | 7.40(3.63) | 0.0001 |
| Total score | 7.1(4.53) | 14.01(6.94) | 0.0001 |
Comparison of ERα and PR expression in control and AUB-E with proliferative /non secretory morphology.
| ER α | Control groupproliferative phasen = 10 cases | AUB-E Proliferativephase n = 26 cases | p-value |
|---|
| Gland | 4.0 (3.22) | 10.34(2.43) | 0.0001 |
| Stroma | 2.1(2.25) | 7.46 (3.49) | 0.0001 |
| Total score | 6.1 (4.32) | 17.86 (5.24) | 0.0001 |
| PR | Control groupproliferative phasen = 10 cases | AUB-E proliferativephase n = 26 cases | p-value |
| Gland | 4.0 (3.31) | 7.80 (4.41) | 0.0190 |
| Stroma | 2.4 (1.35) | 7.69 (3.06) | 0.0001 |
| Total score | 6.40 (4.05) | 15.50 (6.50) | 0.0002 |
Comparison of ERα and PR expression in control and AUB-E with proliferative /non secretory morphology.
| ERα | Control Proliferativephase n=10 | AUB–E hyperplasiawithout atypia n=14 | p-value |
|---|
| Gland | 4.0 (3.22) | 7.5(3.26) | 0.0161 |
| Stroma | 2.1(2.25) | 6.92(4.41) | 0.0045 |
| Total score | 6.1(4.32) | 14.42(6.90) | 0.0028 |
| PR | Control Proliferativephase n=10 | Hyperplasia withoutatypia (n=14) | p-value |
| Glands | 4.0 (3.31) | 9.0(2.47) | 0.0003 |
| Stroma | 2.4(1.35) | 7.42(3.28) | 0.0002 |
| Total Score | 6.40(4.05) | 16.42(4.22) | 0.0001 |
Comparison of ERα and PR expression in control and AUB-E with proliferative /non secretory morphology.
| ERα | Control proliferativephase (n=10) | AUB-E disorderedproliferative (n=28) | p-value |
|---|
| Gland | 4.0 (3.22) | 6.03(3.60) | NS |
| Stroma | 2.1(2.25) | 3.89(3.01) | NS |
| Total score | 6.1(4.32) | 9.92(5.84) | NS |
| PR | Control Proliferativephase (n=10) | AUB-E Disorderedproliferative (n=28) | p-value |
| Gland | 4.0 (3.31) | 5.60(4.26) | NS |
| Stroma | 2.4(1.35) | 5.89(3.93) | 0.0097 |
| Total score | 6.40 (4.05) | 11.50(7.63) | 0.05 |
When cases of control group showing secretory phase endometrium (10 cases) were compared with AUB –E secretory endometrium (32 cases), expression of PR in total score as well as stromal expression was found to be statistically significant [Table/Fig-8].
Comparison of ERα and PR expression in control and AUB-E group with secretory morphology.
| ERα | Control groupsecretory phase(n= 10 cases) | AUB-E secretoryPhase (n = 32 cases) | p-value |
|---|
| Gland | 3.2 (2.35) | 4.46 (3.21) | N.S |
| Stroma | 1.8 (1.32) | 3.90(3.59) | N.S |
| Total score | 5.0 (2.72) | 8.37 (6.06) | N.S |
| PR | Control groupsecretory phase(n= 10 cases) | AUB-E secretoryPhase (n = 32 cases) | p-value |
| Glands | 5.0(2.82) | 5.75(4.98) | NS |
| Stroma | 2.8(3.54) | 7.90(3.36) | 0.0002 |
| Total score | 7.8(4.87) | 13.65(6.56) | 0.0131 |
Discussion
Investigating and treating a non-gravid female presenting with AUB in reproductive age group is quite challenging due non standardized method of investigation and lack of proper categorization of various causes [13,14]. FIGO/PALM-COEIN classification categorizes causes into structural (PALM) and non structural (COEIN) causes [4]. In AUB-E (DUB) group study of steroid receptors in endometrium is valuable as it supports role of hormone receptors in its aetiopathogenesis [15,16]. It was suggested that action of estrogen and progesterone may be potentiated in AUB-E (DUB) through raised concentration of ER and PR in endometrial glands and stroma which can be one of the mechanism in development of AUB [17].
In our study, we found that AUB-E (non-structural) cause is more common cause of AUB. It is in accordance with study [18] where structural component (PALM) was 40% and the non-structural (COEIN) was 60% cases of AUB which included endometrial specimens along with clinical and radiological evidence. However, another study noted higher incidence of structural component (59.5%) than nonstructural causes (40.5%) which included hysterectomy specimen only [19].
Of the PALM component AUB-L was the most common cause. This is in accordance with two other studies [19,20]. AUB-A (10.72%) was second most common cause leading to AUB. This is in accordance with previous researches where the incidences were 9.1% and 12.15% [19,20]. Leiomyoma and adenomyosis coexisted in 4.90% in present study similar to the study by Neena Y et al., [20].
Many researchers are in agreement that polyps are not a common cause of AUB. It accounted for 1.90%, 1.72% and 2% in few studies [19,21,22]. There is discrepancy in the prevalence of polyp as cause of AUB in most of the earlier studies, due to inclusion of endometrial biopsies also that may sometimes fail to detect polypoidal changes without clinical and radiological data.
AUB-M is the least common cause for AUB in reproductive age group. Only one case of endometrioid carcinoma (0.18%) was seen. Authors also agree that malignancies are not important cause of AUB in reproductive age group and should be considered in case of post-menopausal bleeding [21,23–25].
Patients with other non-structural causes of bleeding were not included in the present study and only patients presenting with AUB-E were studied in detail. When classified according to the histological patterns. Proliferative morphology was the commonest (22.72%). This was in accordance with the earlier studies [23,26–29].
Secretory phase endometrium (21.81%) was the next most common endometrial morphology observed in patients with AUB-E. Some of the earlier studies have noted secretory phase as the most common finding (24.9%, 28.9%) [21,30]. Normal cyclical phases of endometrium or the functional causes of endometrium have been reported to be the most common cause of AUB in earlier reports. One study reported proliferative and secretory phases of endometrium, as the most common histological findings present in 71.75% of cases [31]. Another researcher observed normal cyclical endometrium in 40.94% cases [24].
Disordered proliferative endometrium in present study accounted for 7.09%; it is in accordance with other studies [21,29] which observed disordered proliferative endometrium in 5.7% and 6.56% respectively.
Hyperplasia without atypia accounted for 3.80%. None of the earlier studies have classified hyperplasia in atypical hyperplasia and hyperplasia without atypia according to recent WHO 2014. However most of the studies have noted simple hyperplasia as most common cause of hyperplasia. That may correspond with hyperplasia without atypia which was found to be more common than atypical hyperplasia.
Chronic endometritis accounted for 2.18% in present study similar to 2% and 3.23% as in previous studies [23,24].
Raised receptor expression was noted in AUB-E cases. Thus it is in accordance with the study by Fraser IS, which suggested that the action of estrogen and progesterone may be potentiated in DUB (AUB-E) through raised concentration of ER and PR in endometrial glands and stroma which can be one of the mechanism in development of AUB-E (DUB) [17].
In Proliferative/non secretory AUB-E group - AUB-E proliferative phase endometrium and hyperplasia without atypia differs from normal proliferative endometrium by increased receptor expression. Previous study [27] shows similar result for hyperplasia without atypia however was not in accordance in proliferative phase. Excess estrogen stimulates endometrium to proliferate in an undifferentiated manner. Also, there is insufficient progesterone to provide structural support, causing sloughing of endometrial lining. Also, progesterone-guided vasoconstriction and platelet plugging fails to take place leading to profuse bleeding [32]. Estrogen and progesterone exert their effect by ER and PR receptors present in endometrial stromal and glandular cells [6]. Disordered proliferative endometrium group showed persistence of progesterone in stroma. This is in accordance to a previous study, which found higher levels of both ERα and PR level [27].
Secretory phase AUB-E endometrium also showed increased PR receptor expression in stroma. One author also found raised levels of ER and PR receptors in late secretory phase in patient presenting with DUB [32]. This is in contrast to a study by Chakravarty BK et al., [27]. Bleeding in this group may be due to high progesterone- to-estrogen ratio [33].
Main limitation of this study was the use of Immunohistochemistry in only 100 out of 320 cases of AUB-E which was either due to inadequate tissue which was mostly due to small amount of endometrial biopsy tissue or lack of patient’s consent for doing IHC on tissue.
Conclusion
FIGO/PALM-COEIN system of classification will be helpful in deciding treatment in AUB cases. In AUB-E group patients IHC can play important role along with ultrasound and histopathology of endometrial biopsies. It will give insights to pathogenesis in this group. It will be helpful in providing evidence based treatment and prevent from surgical procedures like hysterectomy and endometrial ablation.