Lymphomas occurring in the oral cavity are rare. They account only for about 2% of extra-nodal sites. Most of the lymphomas occur in the lymph nodes and in the oral cavity, the most commonly affected region is the Waldeyer’s ring. Its occurrence in the mandibular gingiva is rare. Here, we describe a case of Diffuse Large B-cell Lymphoma manifested as a non-healing extraction socket in the mandibular right posterior region in a 62-year-old male patient.
Diabetic, Lymph nodes, Lymphadenitis, Sialadenitis
Case Report
A 62-year-old male patient who is a school teacher presented with a chief complaint of swelling on right lower one-third of face since 10 days. The patient was referred to our institution by a private dentist. History revealed that he had pain and mobility of right lower back teeth since one month and thus visited a local dentist 10 days back and got his 45 and 46 extracted. He was prescribed antibiotics and anti-inflammatory drugs (Amoxycillin and Aceclofenac). However, after the extraction, the pain aggravated and he developed a swelling. The patient had also undergone a surgery on the superficial part of right parotid gland two months back for right parotid swelling and it was diagnosed as non-specific sialadenitis and non-specific lymphadenitis.
He was a known diabetic for five years and was under medication (Metformin) and his family history was non-contributory. The patient was an occasional alcoholic and did not give any history of trauma.
On general physical examination, the patient was moderately built and nourished and his bilateral submandibular lymph nodes were enlarged measuring about 1cmX1.5cm in size, tender, firm in consistency and were fixed to the underlying tissue. Upper and middle cervical lymph nodes on the right side were also enlarged, measuring about 2cmX2cm in size, tender, firm in consistency and were fixed to the underlying tissue.
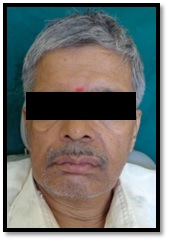
On extra-oral examination, slight facial asymmetry and a diffuse swelling was noted over right lower 1/3rd of the face in mandibular body region [Table/Fig-1]. Drooping of upper and lower eyelids and slight obliteration of naso-labial fold was noted on the right side indicating Bell’s palsy which might have occurred after the surgery on right parotid gland two months back.
Extra-oral photograph showing gross facial asymmetry and swelling on right side of patient’s face.
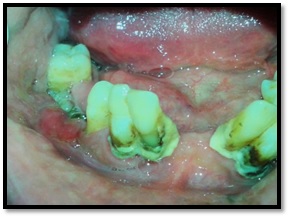
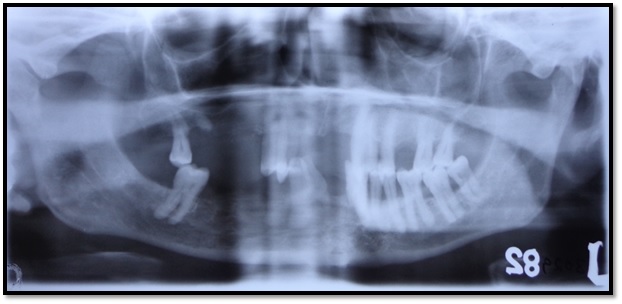
On intra-oral examination, non-healing extraction sockets covered with whitish slough were noted in relation to 45 and 46 that mimicked a chronic non-healing ulcer [Table/Fig-2]. The surrounding alveolar mucosa in this region was swollen and erythematous in appearance. On palpation, the swelling was tender and soft in consistency. OPG showed ill-defined radiolucency in relation to 45 [Table/Fig-3]. Routine blood investigations and TRI-DOT test were performed and were non-contributory [Table/Fig-4]. A provisional diagnosis of non-healing healing extraction socket was given and squamous cell carcinoma was considered in the differential diagnosis.
Intra-oral photograph showing non-healing extraction socket and inflamed surrounding mucosa in relation to 45, 46 region.
OPG showing ill-defined radiolucency in relation to 45.
| INVESTIGATION | RESULT | NORMAL VALUES |
|---|
| Hemoglobin | 14.8 g% | 12-18g% |
| Total Leucocyte Count | 7900 cells/mm3 | 5000-10000 cells/mm3 |
| ESR(Erythrocyte Sedimentation Rate) | 22 mm/1st hour | 0-20 mm/1st hour |
| PCV(Packed Cell Volume) | 42% | 35-50% |
| Bleeding Time | 1 min | 1-5 min |
| Clotting Time | 4 min | 1-8 min |
| Differential Count | Neutrophils – 65% | -- |
| Lymphocytes – 30% |
| Eosinophils – 3% |
| Monocytes – 2% |
| Basophils – 0% |
| Blood Group | ‘O’ positive | -- |
| HIV 1 & 2(TRI-DOT) | negative | -- |
| HB S Ag(HEPASCAN) | negative | -- |
| Random Blood Sugar | 176 mg% | 70-110 mg% |
| Serum Urea | 12.5 mg% | 13-45 mg% |
| Serum Creatinine | 0.85 mg% | 0.6-1.4 mg% |
| SGOT | 23.69 IU/L | 5-34 IU/L |
| SGPT | 23.05 IU/L | 0-40 IU/L |
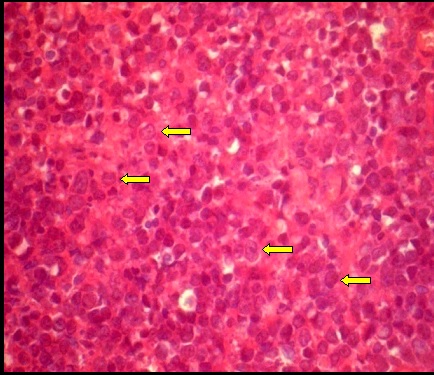
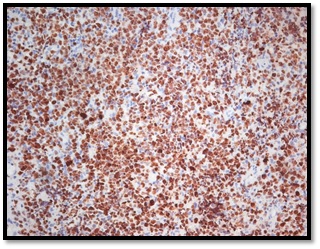
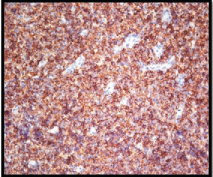
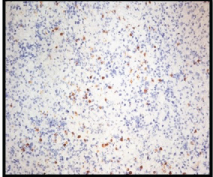
Curettage of the extraction socket was performed and a biopsy was taken from the edge of the ulcer. On histopathological examination, the biopsy specimen revealed moderately dysplastic epithelium and the sub-epithelium showed diffuse proliferation of large cells with round to oval vesicular nucleus and a scant amount of cytoplasm resembling atypical lymphoid cells [Table/Fig-5,6]. Mature lymphocytes and extensive areas of necrosis were seen. Mitotic activity was sparse. Muscle invasion and perineural invasion by the atypical lymphoid cells was also evident. The features were suggestive of Non-Hodgkin’s lymphoma. Immunohistochemistry revealed positivity for Ki-67, CD-20, CD-10 and Bcl-6 markers and stained negative for CD-3 and MUM-1 markers [Table/Fig-7,8,9,10,11 and 12]. Correlating the histopathological and immunohistochemical features, a final diagnosis of Diffuse Large B-cell Lymphoma (Germinal Centre B-cell type, High grade) was given [Table/Fig-8]. The patient was referred to the higher centre for further treatment.
The photomicrograph of the H & E stained section shows dysplastic epithelium and diffuse proliferation of cells in underlying connective tissue[10X].
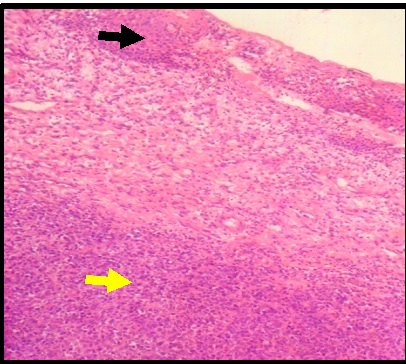
The photomicrograph of the H & E stained section shows large atypical lymphoid cells having round to oval vesicular nucleus and scant amount of pale cytoplasm [40X].
Atypical lymphoid cells showing positivity for ki-67 marker.
Atypical lymphoid cells showing positivity for CD-20 marker.
Atypical lymphoid cells stained negative for CD-3 marker.
Atypical lymphoid cells showing positivity for CD-10 marker.
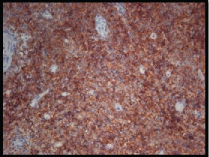
Few atypical lymphoid cells showing positivity for Bcl-6 marker.
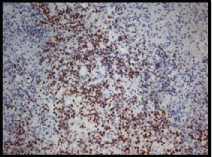
Atypical lymphoid cells stained negative for MUM-1 marker.
Discussion
Lymphomas are the second most common malignant neoplasms involving the soft tissues of head and neck region next only to squamous cell carcinoma [1]. It has been noted that the incidence of head and neck Non Hodgkin’s Lymphoma (NHL’s) has increased in the last few years and older adults were most commonly affected [2]. They usually affect the lymph nodes and Waldeyer’s ring is the most commonly involved site in the oro-facial region [3]. About 20-30% of NHL’s arise from extra-nodal sites [4]. Involvement of the oral cavity is not so common (0.1%-5%) and among the reported cases most of them were noted in the maxilla than in mandible and the most common site involved has been reported to be palate and gingiva [1,2,4].
The clinical presentation of lymphomas in the oral region is not characteristic [1]. They may present as localized swelling with the destruction of hard and soft tissues, painless inflammation of the mucosa with or without ulcerations, tooth mobility, cavitations or as a sessile soft tissue mass [1,5].
Few case reports have been reported where NHL has been diagnosed following tooth extraction. In a report 30-year-old female patient with a growth in the left upper jaw at the site of extraction wound was finally diagnosed as DLBCL [1]. In another case a 57-year-old male with a swelling which increased in size after a tooth was extracted due to mobility and bleeding gums in right posterior region of lower jaw was diagnosed as DLBCL after performing incisional biopsy of the swelling [6].
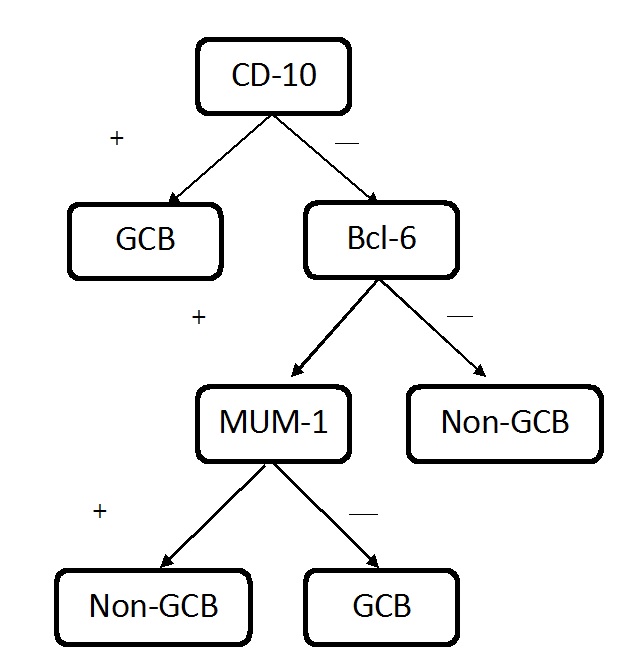
The most frequent type NHL of the oral cavity is Diffuse Large B-Cell Lymphoma (DLBCL) [5]. The aetiology of DLBCL remains unknown and they may originate de novo or because of immune deficiencies which are a significant risk factor [4]. The commonly observed risk factors for NHL include chromosomal translocations and molecular rearrangements, viruses (Epstein - Barr virus, Human T-Cell Leukaemia Virus Type 1, Hepatitis C, and Kaposi’s Sarcoma-Associated Herpes Virus), Helicobacter pylori infection, environmental factors (pesticides, herbicides, solvents and organic chemicals), Congenital and acquired immunodeficiency states and autoimmune disorders. It is characterized by a diffuse proliferation of large neoplastic B cells with nuclear size equal to or exceeding normal macrophage nuclei, or more than twice the size of a normal lymphocyte [5]. It has been broadly divided into two types, Germinal Center B-Cell-Like (GCB) and non-Germinal Center (non-GCB) or Activated B-Cell-Like (ABC) based on gene expression profiles and by immunohistochemical expression of CD10, Bcl-6 and MUM-1. GCB lymphomas have exhibited a better prognosis than non-GCB lymphomas [5]. In normal B-cell differentiation, the B cells go through the pre-germinal center, germinal center and post-germinal center stages [5]. Cells comprising the GC consist of small blast cells, centroblasts, centrocytes and occasionally plasma cells. The B-cells that leave the germinal centre and enter the post-germinal centre phase, differentiate and either become the memory cells or plasma cells. Normal GC B cells express CD10 and Bcl-6. Bcl-6 may be lost by late GC B cells, which in turn acquire MUM-1 expression [5].
In the present case, Ki-67, CD-20, CD-3, CD-10, Bcl-6 and MUM-1 markers were used and the tumor cells were seen to be strongly immunoreactive for CD20, CD10 and Bcl-6 and immunonegative for MUM-1 (10% cells immunopositive) and CD3. The Ki-67 proliferative index was around approximately 90%. These observations indicated that the lesion is highly proliferative and thus is a high-grade lesion and the positivity for CD-10, Bcl-6 markers and negativity for MUM-1 marker indicated it to be a GC B-cell type of DLBCL [Table/Fig-13].
Flow chart for classifying DLBCL into GCB and Non-GCB types using immunohistochemical markers [5].
International Prognostic Index (IPI) developed five clinical parameters in early 1990’s which has developed into the most successful tool in predicting survival in lymphoma patients to date. The parameters are age, stage of disease, extra-nodal site, performance status and serum Lactate Dehydrogenase (LDH) level. Patients with a low-risk IPI score had an approximate 5-year survival rate of 73% and for high-risk group it was 26% [5]. It has been stated that prognosis was better in GC B-cell type than the non GCB-cell type of lesion which was the main reason for further classification of the DLBCL.
The exact cause of the lesion in the present case is unknown but there has been considerable evidence that lymphomas at specific sites are preceded by the presence of a local inflammatory process [6]. It has been stated that inflammation increases the rate of cell division of lymphocytes, thereby increasing the chance of a malignant clone developing [6].
Current treatment of DLBCL includes multi-agent chemotherapy; typically CHOP (cyclophosphamide, hydroxydoxorubicin, oncovin and prednisone). It has been noted that although patients have responded to the initial therapy, more than half the patients have succumbed to the disease. In the early stage, the treatment involves either chemotherapy alone or a combination of chemotherapy and radiotherapy. The chemotherapy usually involves three cycles of CHOP. Bone marrow transplant is to be considered if remission is not maintained. The role of surgery is severely limited in the treatment of DLBCL. Other drugs used in multi-agent chemotherapy for advanced stage disease include various combinations of methotrexate, bleomycin, doxorubicin, vincristine, dexamethasone and leucovorin, etoposide, mechlorethamine, procarbazine, cytarabine [5].
Conclusion
An innocent face can have a wild side. This quote truly applies to the present case as a simple non-healing extraction socket has finally turned out to be diagnosed as High-Grade Diffuse Large B-cell Lymphoma (Germinal Centre B-cell type). Hence, it is important to look at the cases with suspicion even though they appear to be not harmful as some of the life-threatening diseases have got innocuous symptoms. The immunohistochemistry carried out in the present case helped us to distinguish between the types of DLBCL which has got a prognostic importance as GCB-cell type of DLBCL has got a better prognosis.
[1]. Agrawal MG, Agrawal SM, Kambalimath DH, Non-Hodgkin’s lymphoma of maxilla: a rare entityNatl J Maxillofac Surg 2011 2(2):210-13. [Google Scholar]
[2]. Alirezaei S, Baharvand M, Tavakoli B, Sarikhani S, Mafi AR, Advanced primary lymphoma of oral cavity: report of a CaseOpen Journal of Stomatology 2014 4:109-14. [Google Scholar]
[3]. Parihar S, Garg RK, Narain P, Primary extra-nodal non-Hodgkin’s lymphoma of gingiva: a diagnostic dilemmaJ Oral Maxillofac Pathol 2013 17(2):320 [Google Scholar]
[4]. Vinoth PN, Selvan SM, Sahni L, Krishnaratnam K, Rajendiran S, Anand CV, Primary extra nodal non-Hodgkin’s lymphoma of the oral cavity in a young girlNatl J Maxillofac Surg 2012 3(2):187-89. [Google Scholar]
[5]. Bhattacharyya I, Chehal HK, Cohen DM, Samer Z, Al-Quran. Primary diffuse large B-cell lymphoma of the oral cavity: germinal center classificationHead and Neck Pathol 2010 4:181-91. [Google Scholar]
[6]. Parrington SJ, Moorthy AP, Primary non-Hodgkin’s lymphoma of the mandible presenting following tooth extractionBritish Dental Journal 1999 187(9):468-70. [Google Scholar]