Osteoporosis is a serious, metabolic bone disorder that leads to increased susceptibility to fractures and decreased mechanical strength of the bones. Osteoporosis is a silent disease that is developed in millions of the people worldwide. Since osteoporosis is asymptomatic unless a fracture occurs, it is called silent thief [1–3].
It was estimated that over 200 million people suffer from osteoporosis around the world [4]. Osteoporosis-associated fractures impose a considerable economic burden on health system and this disease, if left untreated, results in serious and grueling outcomes [5]. It was reported that the worldwide annual incidence of hip fracture was about 1.7 milion in 1992 [4]. Approximately 30% of all postmenopausal women have had osteoporosis in the United States and Europe. It was stated that the risk of hip fracture in these countries was even more than the risk of breast cancer in 2001 [6]. Studies on Asian women have indicated that bone density is lower in these women compared to European and the U.S women [7]. The highest prevalence of osteoporosis is observed in the elderly, particularly women, such that the investigations have demonstrated that women develop osteoporosis four times higher than men, but sometimes this disease has occurred in skeletal growth period and even adolescence [8].
Osteoporosis is a multifactorial disease and is related to interaction between genetic and environmental factors. The study of potential genetic predisposition to osteoporosis is highly important. Several works have reported that osteoporosis is associated with the polymorphisms of some genes [9,10]. Analyses have revealed that vitamin D receptor (VDR), calcitonin receptor, Estrogen receptor (ER) gene, Type I collagen (COLIAI) gene, and Lactase (LCT) gene have the highest contribution to osteoporosis expansion [11]. Mineral homeostasis and bone metabolism are totally dependent on vitamin D. This vitamin plays its role through its receptor and vitamin D receptor (VDR) gene determines receptor structure. Therefore, VDR gene could be an important candidate gene for osteoporosis [12]. Gene mutation could influence function and be associated with potential development of osteoporosis [13]. Although a strong association has been offered between VDR genotypes and bone mineral density (BMD) in osteoporosis [14], inconsistent findings have been obtained on the possible association between postmenopausal bone loss and VDR genotype [12,15]. VDR gene is located on long arm of chromosome 12 (12q12-14). 10 exons and 8 interons have been determined on this gene’s structure [16]. VDR polymorphisms consist of FokI, ApaI, BsmI, TaqI, Cdx2, etc [17].
The ApaI polymorphism (rs7975232) is located on interon 8. This polymorphism causes guanine nucleotide to be substituted with thymine (G→T) [18]. In the TaqI polymorphism, the nucleotide at position 352 is converted from thymine into cytosine (T352C). Therefore, ATT codon which encodes isoleucine is converted into the codon of another isoleucine, i.e. ATC [19,20]. In the VDR gene, the ability to cut by the ApaI and TaqI enzymes leads to development of other genotypes as follows:
The genotypes of ApaI: AA (absence of the position identifying the enzyme); Aa (heterozygous position of identifying the enzyme), and aa (existence of the position identifying the enzyme) and the genotypes of TaqI: TT (absence of the position identifying the enzyme); Tt (heterozygous position of identifying the enzyme), and tt (existence of the position identifying the enzyme).
The BsmI polymorphism (rs1544410) is located on interon 8 of VDR gene. In this polymorphism, the nucleotide adenine is substituted with guanine (A→G) [18].
The relationship between the VDR genotypes and BMD may be different in various racial and geographical populations [21,22]. This study was conducted to investigate the association between the ApaI, TaqI, and BsmI polymorphisms and bone density in 45 and over 45 year-old women in southwestern Iran.
Materials and Methods
In this analytical study, blood samples were taken from 45- and over 45-year-old women referring centers for bone density measurement in southwestern Iran based on convenience sampling. In this study, based on Cockran formula 200 participants (130 patients and 70 healthy individuals) were investigated after their consent was obtained. Exclusion criteria were curtin consumption history, ovariectomization or premature ovarian failure, thyroid disease, calcium intake disorders, gastrointestinal and renal diseases and other diseases demonstrated to be associated with osteoporosis.
The bone density rate in lumbar vertebrae and femoral neck was measured by dual-energy X-ray absorptiometry (DEXA) method using Hologic QDR (Germany) and the values were determined by t-score. Then a 5 ml complete blood was taken for genetic examinations and collected in the tubes containing EDTA Anti-coagulation. The samples were kept at 20°C till the day of tests.
The genomic DNA was extracted using phenol-chloroform method. The polymorphisms of ApaI, TaqI and BsmI were amplified from VDR gene using the primers designed by Gene Runner Software. The used primers are shown in [Table/Fig-1].
| polymorphism | The size of fragment | Primer type | 5’-3’ Primer sequences |
|---|
| ApaI | 737 | F | CAGAGCATGGACAGGGAGCAA |
| R | TCATGGCTGAGGTCTCAAGGG |
| TaqI | 737 | F | CAGAGCATGGACAGGGAGCAA |
| R | TCATGGCTGAGGTCTCAAGGG |
| BsmI | 358 | F | GGGAGACGTAGCAAAAGG |
| R | AGAGGTCAAGGGTCACTG |
The materials in PCR reaction were as follows: 0.2 ml 10X buffer, 1.5 μl 50mM MgCl2, 10 mM dNTP (2’-deoxynucleoside 5’-triphosphate) Mix, 0.7 μliter 10 pmol/ml forward primer, 0.7 μl10 pmol/ml reverse primer, 0.2 μl of Taq polymerase (5 U/μl), 3.2 μliters of template DNA, and ddH2O till the reaction volume reaches 25 μl.
PCR reactions were done for ApaI and TaqI using the temperature gradient as follows:
95°C for 5 min as initial denaturation followed by 95°C for 50 seconds, 68.7°C for 45 seconds as the connection point, 72°C for 1 min as a 36-cycle extension, and finally 72°C for 5 min as final extension.
PCR reaction was done for BsmI per touchdown PCR using the temperature gradient as follows: 95°C for 3 min as initial denaturation followed by 95°C for 30 seconds, 56°C for 40 seconds and 72°C for 30 seconds for 8-cycle proliferation, and then 95°C for 40 seconds, 49°C for 40 seconds and 72°C for 30 seconds for 26-cycle amplification, and finally 72°C for 5 min as final extension. Thermocycler (ASTEC, PC818 Japan) was used.
After amplification, the fragments were digested using ApaI, TaqI, and BsmI restriction enzymes (Fermentas).
The RFLP reaction for each of the polymorphisms (for 30-μl volume) were as follows: 17.5μl sterile water, 2μl 10X buffer, 10μl PCR product, and 0.5μl restriction ApaI, TaqI and MVa12691 (BsmI) enzymes. Then, they were left at 37°C. The obtained products were reviewed on an 8% polyacrylamide gel and the genotype of ApaI, TaqI and BsmI were determined.
Statistical Analysis
The data on bone density and PCR-RFLP were analysed by chi-square and ANOVA. Also, triad combination of the genotypes was statistically analysed. For each genotype combination, chi-square was run between the patients and control group and p-value was calculated.
Results
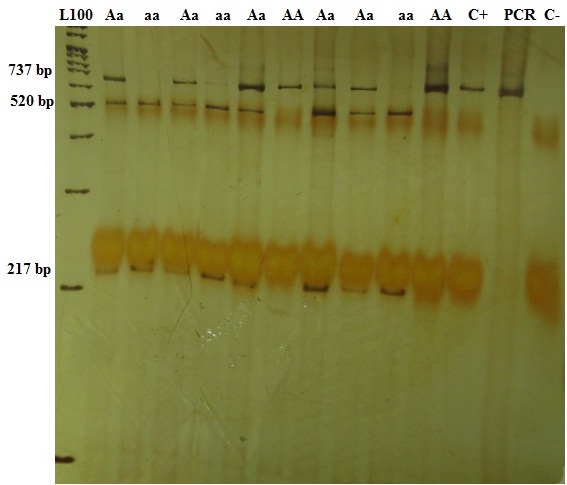
In 200 samples there were 737 bp amplified fragments for ApaI polymorphism, which produced two 520-bp and 217-bp fragments after digestion with ApaI enzyme if the position of restriction site was present [Table/Fig-2].
The association of ApaI, TaqI, and BsmI polymorphisms with t-score in the patients.
| polymorphism | The patients’ genotype | Number | Femoral neck’s T-score Mean±SD | Lumbar vertebrae’s T-score Mean±SD |
|---|
| ApaI polymorphism | AA | 50 | -1.29±0.34 | -2.75±0.83 |
| Aa | 79 | -1.3±0.55 | -2.83±0.50 |
| aa | 1 | -2.1±0 | -2.9±0 |
| p-value | 0.269 | 0.797 |
| TaqI polymorphism | TT | 57 | -1.43±0.48 | -2.80±0.47 |
| Tt | 66 | -1.2±0.48 | -2.80±0.68 |
| tt | 7 | -1.27±0.35 | -2.80±1.38 |
| p-value | 0.025 | 0.999 |
| BsmI polymorphism | BB | 31 | -1.14±0.26 | -2.92±0.78 |
| Bb | 70 | -1.3±0.52 | 2.68±0.61 |
| bb | 29 | -1.48±0.53 | -2.94±0.53 |
| p-value | 0.025 | 0.091 |
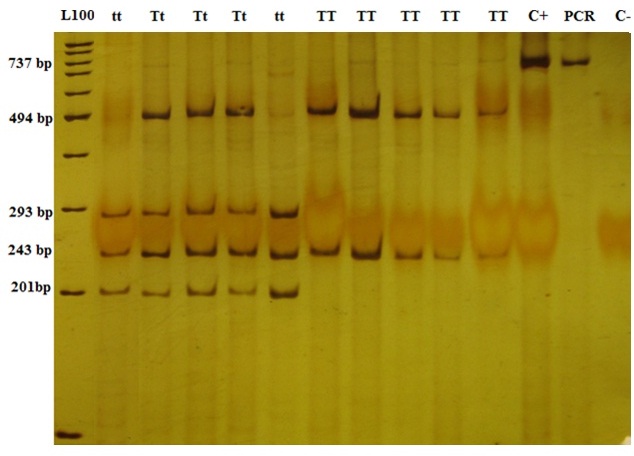
The length of amplified fragment in TaqI was 737 bp and the fragments below were produced due to the presence of a mandatory secondary position of identifying the enzyme in normal conditions: the TT genotype produced two 494-bp and 243-bp fragments, Tt four 494-bp, 243-bp, 201-bp, and 293-bp fragments, and tt three 243-bp, 201-bp, and 293-bp fragments [Table/Fig-3].
The comparion of the genotypic combinations between the control group and the patients.
| Genotypic combination | T- score of the patients’ femoral neck | T- score of the patients’ lumbar vertebrae | The observed number/ patients | The expectednumber/ patients | The observed number/ controls | The expected number/ controls | Chi-Square | p-value |
|---|
| AABBTt | -1.1±0.19 | -3.02±0.56 | 18 | 12 | 6 | 12 | 6 | p<0.05 |
| AABBtt | -1.27±0.49 | -2.9±1.92 | 4 | 4 | 4 | 4 | 0 | p<0.05 |
| AABbTT | -1.6±0.43 | -2.61±0.44 | 7 | 6 | 5 | 6 | 0.32 | p<0.05 |
| AABbTt | -1.41±0.32 | -2.35±0.86 | 15 | 10.5 | 6 | 10.5 | 3.86 | p<0.05 |
| AAbbTT | -1.24±0.34 | -3.04±0.67 | 5 | 4 | 3 | 4 | 0.5 | p<0.05 |
| AaBBTt | -1.16±0.17 | -2.76±0.41 | 6 | 4 | 2 | 4 | 2 | p<0.05 |
| AaBbTT | -1.34±0.38 | -2.64±0.34 | 19 | 15 | 11 | 15 | 2.14 | p<0.05 |
| AaBbTt | -1.14±0.68 | -2.9±0.59 | 27 | 21 | 15 | 21 | 3.42 | p<0.05 |
| AaBbtt | -1.3±0 | -2.9±0 | 1 | 1.5 | 2 | 1.5 | 1.4 | p<0.05 |
| AabbTT | -1.51±0.56 | -2.92±0.52 | 23 | 16.5 | 10 | 16.5 | 5.2 | p<0.05 |
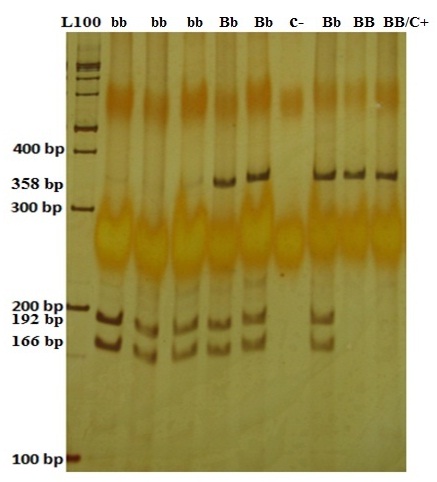
The BsmI enzyme cut the samples with b allele (the bb genotype) and produced two 192-bp and 166-bp fragments. The samples with BB genotype remained unchanged and had a 358-bp band, and those with Bb genotype had 192-bp, 166-bp, and 358-bp fragments [Table/Fig-4].
Polyacrylamide gel of the PCR-RFLP relevant to ApaI genotypes. The 737 bp fragment was seen for AA and Aa genotypes. The 520-bp and 217-bp fragments were seen after digestion with ApaI enzyme for Aa and aa genotypes. C+ and C- are abbreviation for control positive and control negative, respectively. Control positive and PCR show the 737 bp original amplified fragment. Control negative does not show any band as expected.
In the patients, the frequency distribution of ApaI genotypes was 79 (60.8%) individuals with Aa, followed by AA in 50 (38.5%) and aa in one (0.7%). In the control group it was 41 (58.6%) individuals with Aa, followed by AA in 26 (37.1%) and aa in 3 (4.3%). By statistical analyses, there was no significant association between ApaI polymorphism and bone density rate in femoral neck and lumbar vertebrae (p<0.05). In the patients, the frequency distribution of TaqI genotypes was 66 (50.8%) individuals with Tt, followed by TT in 57 (43.8%) and tt in seven (5.4%). In the control group it was 34 (48.5%) individuals with TT, followed by Tt in 30 (42.9%) and tt in 6 (8.6%). By statistical analyses, there was a significant association between TaqI polymorphism and bone density rate in femoral neck (p<0.05) and no significant association between TaqI polymorphism and bone density rate in lumbar vertebrae (p>0.05). The individuals with the homozygous dominant TT genotype had the lowest femoral neck’s bone density rate and lumbar vertebrae’s was the same in all genotypes.
In the patients, the frequency distribution of BsmI genotypes was 70 (53.8%) individuals with Bb, followed by BB in 31 (23.8%) and bb in 29 (22.3%). In the control group it was 39 (55.7%) individuals with Aa, followed by AA in 14 (20%) and aa in 17 (24.3%). By statistical analyses, there was a significant association between BsmI polymorphism and bone density rate in femoral neck (p<0.05) and no significant association between this polymorphism and bone density rate in lumbar vertebrae (p>0.05). The individuals with the homozygous recessive Bb genotype had a lower bone density rate in femoral neck and lumbar vertebrae compared to the genotypes Bb and BB [Table/Fig-5].
Polyacrylamide gel of the PCR-RFLP relevant to TaqI genotypes. The TT genotype produced two 494-bp and 243-bp fragments. Tt genotype produced four 494-bp, 243-bp, 201-bp, and 293-bp fragments, and tt one produced three 243-bp, 201-bp, and 293-bp fragments. C+ and C- are abbreviation for control positive and control negative, respectively. Control positive and PCR show the 737 bp original amplified fragment. Control negative does not show any band as expected.
The results of sequence of polymorphic position are shown in [Table/Fig-6,7 and 8]. For ApaI polymorphism, substitution of guanine with thymine removes the restriction site completely in AA genotype. For TaqI polymorphism, conversion of thymine into cytosine leads to generation of a restriction site in tt genotype. For BsmI polymorphism, substitution of adenine with guanine produces a restriction site in bb genotype.
Polyacrylamide gel of the PCR-RFLP relevant to BsmI genotypes. The bb genotype produced two 192-bp and 166-bp fragments. The BB genotype had a 358-bp band, and the Bb genotype had 192-bp, 166-bp, and 358-bp fragments. C+ is abbreviation for control positive. Control positive shows the 358 bp original amplified fragment.
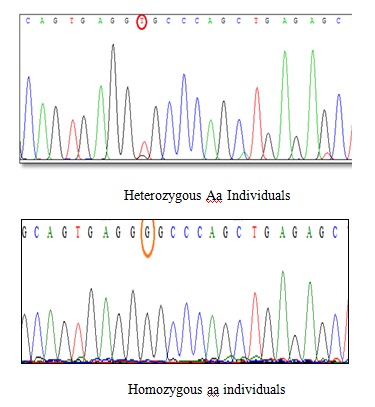
The results of DNA sequencing for ApaI polymorphism The results of sequence determination indicated that G→T occurred in heterozygous Aa individuals (GT), but this conversion did not occur in homozygous aa individuals.
The results of DNA sequencing for TaqI polymorphism. The results of sequence determination indicated that T→C occurred in heterozygous Tt individuals (TC), but this conversion did not occur in homozygous tt individuals (TT).
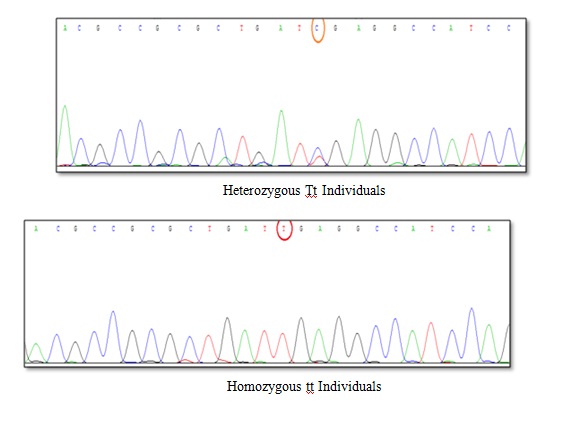
The results of triad combination: 11 genotypic combinations were common between the patients and the control group. In view of p-value, there was a significant difference in frequency of only AABBTt, AABbTt, and AabbTT between the control group and the patients (p<0.05). The individuals with AABbTt genotype had the highest bone density rate of lumbar vertebrae in the patients and those with AabbTT had the least bone density rate of femoral neck in the patients [Table/Fig-9].
The results of sequence for BsmI polymorphism. (The results of sequence determination indicated that G→A occurred in both heterozygous Bb individuals (GA) and homozygous BB individuals (AA).
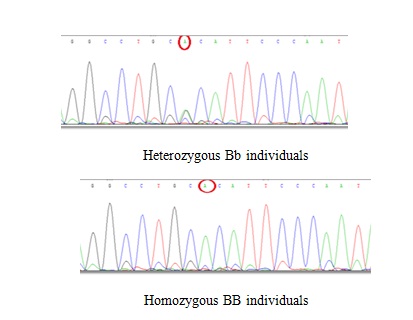
Discussion
Osteoporosis is a serious, metabolic bone disorder. Osteoporosis expansion can be related to VDR gene polymorphisms, including ApaI, TaqI and BsmI polymorphisms. This study indicated that there was no significant association between ApaI polymorphism and bone density rate in lumar vertebrae and femoral neck; TaqI and BsmI polymorphisms were significantly associated with femoral neck’s bone density rate, but not significantly associated with lumbar vertebrae’s. In addition, the patients with homozygous recessive bb genotype had a lower bone density rate in femoral neck and lumbar vertebrae compared to the genotypes Bb and BB.
In similar works by Qin et al., on the association of different polymorphisms of VDR gene and estrogen receptor alpha with bone density in postmenopausal women, no significant association was noted between ApaI polymorphism and bone density in femoral neck and lumbar vertebrae [23]. Zajickova et al., investigated the association of BsmI, ApaI and TaqI polymorphisms with bone density in 114 Czech postmenopausal women with mean age of 62.5±8.9 years. They argued that there was no significant association between these polymorphisms and bone density in any skeletal site and no significant difference in genotypic distribution was observed between the patients and the control group [24]. Uysal et al., also examined the association between BsmI, ApaI, and TaqI and osteoporosis in 246 postmenopausal women in Turkey and found no significant association between ApaI and TaqI polymorphisms and osteoporosis [25]. Shen et al., indicate that ApaI, BsmI, TaqI and FokI polymorphisms may be not associated with the risk of fracture in postmenopausal women [26]. But, Mitra et al., examined 246 Indian postmenopausal women with mean age of 54.2±3.4-year-old and reported mean bone density in spine and femoral neck of the women with homozygous recessive AA genotype and homozygous dominant TT genotype was 10% higher than that of the women with the genotypes AA and tt [21].
Tamulaitiene et al., reported similar findings to those on BsmI polymorphism obtained in the present study. They stated there was no significant difference in the frequency of BsmI genotypes between the patients and the control group. Also, the BsmI polymorphism of VDR gene had a non significant, weak association with BMD and severe postmenopausal osteoporosis, which needed further investigations. In the patients with severe osteoporosis, the bone density of the individuals with the BB genotype was higher compared to those with the genotypes Bb and bb [27]. But, Ji et al., in a meta-analysis found that in the Caucasian cases with osteoporosis-related fracture, the frequency of homozygous recessive bb genotype was significantly lower [28]. Also, Garnero et al., indicated an association between the increase in B allele and fracture frequency. The relative risk of such fractures in the women with heterozygous Bb genotype was 1.5 times higher and in the women with the homozygous dominant BB genotype was 2.1 times higher compared to the women carrying homozygous recessive bb genotype [29].
Langdahl et al., investigated the BsmI, ApaI, and TaqI polymorphisms in 192 patients with osteoporosis and 207 healthy controls in Denmark and found the genotypes BB and Bb were higher. Femoral neck’s BMD was higher in the individuals with Bb genotype. They concluded that B allele of BsmI polymorphism was associated with decrease in bone density and the fractures due to osteoporosis [30]. Thakkinstian et al., did an extensive analysis on the association between BsmI polymorphism of VDR gene and bone density and found that B allele was significantly associated with lumbar vertebrae’s bone density and also the individuals with the BB genotype had a lower bone density compared to those with the genotypes Bb and bb [31].
When comparing genotypes in the patients individually, the Aa genotype of ApaI polymorphism had a bone density approximately similar to that of the AA genotype. For TaqI polymorphism in the patients, Tt genotype was similar to the two other genotypes in view of lumbar vertebrae’s bone density and between the two other genotypes (TT, tt) in view of femoral necks. Therefore, since frequency of the genotypic combination AABbTt is low (15 of 130) in the patients, this combination could represent less likelihood of fracture in the individuals with this genotype compared to other patients.
In addition, of the three above-mentioned genotypic combinations, the individuals with genotypic combination of AabbTT had the lowest bone density of femoral neck and this genotypic combination was observed in 23 patients. When comparing the genotypes of each polymorphism individually, we observed that the Bb genotype of BsmI polymorphism and TT genotype of TaqI polymorphism had the lowest bone density of femoral neck compared to other relevant genotypes. Therefore, simultaneous study of two polymorphisms is more valuable than studying them separately, but the genotypes related to ApaI polymorphism seem to yield little data on genotypic combinations. The same could be concluded for examining this polymorphism separately.
Conclusion
The study of genotypic combination of three polymorphisms does not yield the data inconsistent with those of studies on individual polymorphisms and simultaneous study of three polymorphisms could yield better data compared to study of individual polymorphisms. TaqI and BsmI polymorphisms could be good markers in identifying the women predisposed to osteoporosis in the region and even Iran, however larger and/or other populations need to be investigated to confirm these findings.