Introduction
Langerhan’s Cells (LCs) are dendritic cells of the oral epithelium which play a role in a series of oral lesions from gingivitis to oral cancer. Oral Lichen Planus (OLP) is an oral mucosal T-lymphocyte mediated immunologic reaction to an unidentified putative antigen or allergen.
Aim
The aim of this study was to quantify the presence of immature LCs in OLP comparing them with normal epithelium.
Materials and Methods
A retrospective study using 30 of OLP cases were conducted. Immunohistochemistry was performed using polyclonal anti-CD1a antibodies to identify LCs in 10 cases of normal tissue and 30 samples of OLP. The distribution of LCs among lesional tissue and normal mucosa was analysed using Mann-Whitney U test.
Results
LC population in OLP was significantly higher when compared to the normal epithelium (p<0.001).
Conclusion
The increase in LCs indicates the active role played during the antigen detection in OLP and subsequent presentation to T-lymphocytes.
Antigens, Antigen presentation, Dendritic cells, Mouth mucosa
Introduction
Oral cavity is one of the target areas of the human body to assess the general health status of an individual. The histological revelations rather than the clinical findings are focus of attention in diagnosis of mucosal diseases as biologically diverse lesions resemble each other. Lichen Planus (LP), with its clinical resemblance to lichenoid lesions and histological features mimicking chronic inflammatory conditions, poses a challenge to both clinicians and pathologists [1].
LP is a fairly distinctive mucocutaneous disorder that affects the skin, mucous membrane, nails and hair [2]. Oral Lichen Planus (OLP) most frequently involving the buccal mucosa symmetrically, is a common chronic inflammatory disease of unknown etiology, affecting 0.1-4% of various populations [3]. OLP is more common in females, mean age of onset is between 40-50yrs [4,5].
OLP is an oral mucosal T-lymphocyte mediated immunologic reaction to an unidentified putative antigen or allergen [6]. It represents an autoimmune response to basal cell antigens [7]. The Antigen Presenting Cells (APC) of oral epithelium i.e., the Langerhan’s Cells (LCs) have a crucial role in antigen presentation to T-cells in OLP [8].
The classical histopathological features of OLP are hyperorthokeratosis or hyperparakeratosis, acanthosis, thickening of the granular cell layer, basal cell liquefaction, "saw-tooth" configuration of the rete pegs and a band-like dense inflammatory cellular infiltrate in the upper lamina propria [2]. The inflammatory infiltrate in OLP usually consists of T-cells, including both CD4+ and CD8+ lymphocytes [6].
The frequency of malignant transformation ranges from 0.4% to 3.3%. The most important complication of OLP is development of oral squamous cell carcinoma [9].
The nature of antigen in OLP has not yet been established and many studies have been conducted by various authors but still it remains an enigma. The role of APCs like LCs in OLP cannot be disregarded and with the advent of immunohistochemistry, specific markers for LCs have been identified and have found wide usage to define the role of these cells.
CD1a, a membrane protein expressed by dendritic cells like LCs, is used as a marker for immature LCs in the normal oral epithelium. Its expression in OLP was studied and compared with that of normal oral mucosa but results obtained were contradictory [9]. The process of antigen presentation to T-cells in OLP by LCs involves both immature and mature LCs where in the immature LCs undergo a process of maturation by activation of numerous co-stimulatory pathways. The mature LCs then migrates into the connective tissue to reach the lymph nodes to present antigen to the T-cells [9].
Previous immunohistochemical studies have shown the role of immature LCs in OLP. They found an increased number of CD1a expressing LCs in OLP compared to healthy controls [10].
Aim
The purpose of this study was to determine the immunohistochemical expression of immature LCs in OLP with a greater sample size and thus assess the role of LCs in OLP which in turn may be helpful in better understanding of the pathogenesis of OLP.
Materials and Methods
The present study was undertaken to observe the expression of CD1a by LCs in OLP. It was a retrospective study comprising of 30 cases of OLP obtained by retrieving previous paraffin embedded tissue blocks of 15 OLP cases from the Department of Oral Pathology and Microbiology, Sri Sai College of Dental Surgery, Vikarabad, India and 15 new cases of OLP reporting to the Department of Oral Medicine and Radiology, Government Dental College, Hyderabad over a period of six months. The staining was carried out in ACTREC Lab, Kharghar, Mumbai, India.
Total 30 cases of OLP were selected out of which 14 were females and 16 were males. Ten samples of normal oral mucosa served as controls which were taken from five females and five males subjects.
Inclusion criteria: Patients with clinically and histopathologically diagnosed OLP and normal healthy subjects were considered as controls.
Exclusion criteria: Patients with OLP on medication for other systemic diseases and pregnant women with OLP.
In patients suspected to be suffering from OLP, case history was recorded as per the proforma and biopsies were taken from lesions occurring on buccal mucosa as part of the diagnostic procedure. All these cases were diagnosed by routine haematoxylin and eosin staining. These were subjected to immunohistochemical staining for CD1a. Control specimens were obtained from healthy oral mucosa of patients undergoing oral surgery for removal of impacted third molars. A written and informed consent was obtained from all the patients for the biopsy procedures.
Immunohistochemistry (IHC): Sections of 4μm thickness were taken onto the polylysine coated slides. Routine immunohisto-chemical technique was followed which included deparaffinisaton of slides followed by rehydration and after which antigen retrieval was done in microwave oven at 450°c and then peroxide block and power block was done to prevent the background staining then followed by application of primary antibody (Monoclonal Mouse Anti-Human CD1a : Dako, Glostrup, Denmark) and then application of poly Horse Radish Peroxidase reagent followed by addition of substrate chromogen (DAB) was done and H&E counterstaining was finally done for evaluation
Evaluation of the staining: Quantitative analysis was performed on one section per biopsy. Criteria followed for identification of LCs was [11]:
Positive (brown) staining by chromogen.
Cell body with at least two well-visualized dendrites.
CD1a positive LCs were counted manually in randomly selected six High Power Fields (HPF). The slide was moved in a single direction (from right to left) to avoid repetition of already examined fields. Number of CD1a-labelled LCs was counted as mean number of positive cells/HPF.
Statistical Analysis
All calculations were performed using IBM SPSS statistical software package (SPSS 19.0 for Windows, SPSS Inc, Chicago, IL, USA). The obtained data was analyzed by relevant statistical tests and the p-value less than 0.05 was considered to be statistically significant.
Results
An immunohistochemical study was conducted to observe expression of CD1a by LCs in OLP. For this study 30 tissue samples of buccal mucosa of OLP patients were taken. Ten samples of normal oral mucosa (five males and five females) were obtained which served as controls. The data obtained from the present study was compiled, tabulated and subjected to relevant statistical analysis. The results were presented in the following manner: The mean CD1a expression by LCs in OLP was estimated by analyzing the obtained data by Wilcoxon Signed Rank Test. A p-value less than 0.05 was considered significant. It was noted that mean CD1a expression was higher in epithelium than connective tissue in OLP and this increase was statistically significant (p<0.05) [Table/Fig-1].
Mean CD1a expression by LCs in OLP.
| Antibody | Epithelium | Connective tissue | p-value |
|---|
| CD1a | 7.04 | 2.37 | p < 0.001 |
Wilcoxon Signed Ranks Test p < 0.001
In the control group, the mean CD1a expression by LCs was analysed statistically and the mean value was higher in epithelium than connective tissue of controls and this increase was statistically significant (p<0.05) [Table/Fig-2].
Mean CD1a expression by LCs in controls.
| Antibody | Epithelium | Connective tissue | p-value |
|---|
| CD1a | 4.31 | 0.93 | p = 0.005 |
Wilcoxon Signed Ranks Test p = 0.005
The expression of CD1a by LCs in OLP was then compared to the corresponding mean values of CD1a in controls. The obtained data was analyzed by Mann Whitney U Test and p-value less than 0.05 was considered as significant.
The mean CD1a expression by LCs in epithelium of OLP was much higher than that of epithelium of the controls and this increase was statistically significant (p<0.05). Also in the connective tissue, the mean CD1a expression of LCs in OLP showed a significant increase than that of connective tissue of controls (p<0.05) [Table/Fig-3].
Mean CD1a expression by LCs in controls and OLP.
| Component | Control | OLP | p-value |
|---|
| Epithelium | 4.31 | 7.04 | p = 0.001 |
| Connective tissue | 0.93 | 2.37 | p = 0.011 |
Mann Whitney U Test p=0.001 (Epithelium), p=0.011 (Connective tissue)
Discussion
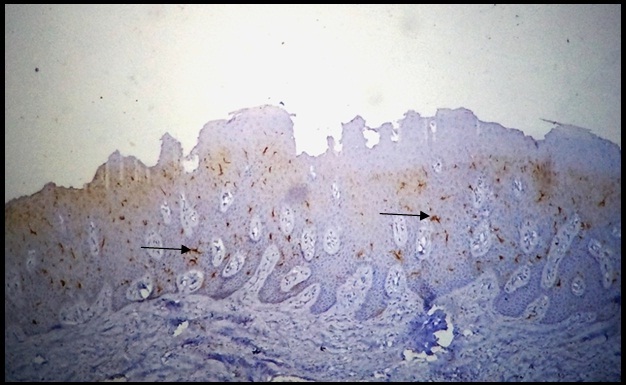
LCs are APC’s present in skin and normal oral mucosa, which are derived from bone marrow and finally reside in these peripheral tissues. In oral mucosa they are present just above the basal layer of stratified squamous epithelium with dendritic processes directed towards the epithelium [Table/Fig-4]. In case of antigenic challenge, before the T-cell activation begins, DCs must process antigen-derived peptides and present them together with major histocompatablity complex molecules to T-cells [12].
Normal oral mucosa showing CD1a-positive Langerhan’s cells in the epithelium (10X).
The presence of LCs and their expression in epithelium of OLP has been widely investigated after advent of immunohistochemistry. A wide variety of monoclonal antibodies are now available with shortened procedure time and greater shelf life to detect LCs in both immature and mature form. The immature LCs in oral mucosa express CD1a molecule more than any other CD1 isoforms, which is highly capable of presenting various types of microbial lipid and self antigens to T-cells [13].
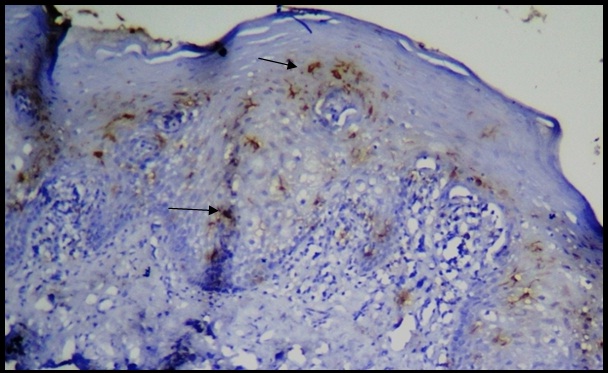
CD1a in OLP and compared with that of normal oral epithelium and thus, were utilized for the study and immunohistochemical staining [Table/Fig-5] was carried out and results obtained were statistically analyzed and tabulated.
CD1a-positive cells in OLP in the epithelium and connective tissue (40X).
The present study showed that in normal oral mucosa the CD1a stained LCs were abundantly seen in the epithelium just above the basal cell layer while in connective tissue the cells were sparse. Our results are in accordance with the results obtained by Chou [14], Pitigala-Arachi [15], Van Loon [16], Lombardi [17], Hasseus [18], Dorrego [19], Santoro [20], Gustafsson [10] and Mitamura [21] who had also noted increase CD1a+ LCs in epithelium than in the connective tissue. This highlights the known fact that the LCs are immune sentinels present more in normal epithelium and are always ready for antigen capture.
In addition, Hasseus [18] and Gustafsson [10] suggested that the CD1a stained LCs in epithelium of the normal mucosa formed an intraepithelial network for better coordination in antigen capture and this feature was also noted in our study. CD1a antibody was chosen as it is exhibited by LCs at exceptionally higher levels compared to other subsets of dendritic cells occurring in the mucosa [22].
Our study revealed that CD1a expressing LCs were more abundant in the epithelium of OLP lesions than in connective tissue. Moreover, on comparison with normal oral epithelium there was an increase in mean number of CD1a molecule expressing LCs per high power field in OLP epithelium. Similar findings have been reported by many other authors also namely Pitigala-Arachi [15], Hasseus [18], Dorrego [19], Santoro [20], Gustafsson [10] and Mitamura [21]. This increase can probably be due to the capture of unknown antigen which is present in excessive amount in OLP leading to mobilization of numerous LCs to the site and thus preventing the destructive epithelial changes in OLP to some extent.
CD1a-positive LCs were also seen in connective tissue of OLP in our study. This finding is in accordance with studies conducted by Santoro [20] and Gustafsson [10] who found an increased numbers of CD1a-positive cells in connective tissue along with the epithelium of OLP when compared to connective tissue of normal oral mucosa. The presence of CD1a-stained LCs in connective tissue may signify the fact that after the antigen capture, the activated LCs are in the intermediate stage of maturation wherein they migrate to regional lymph nodes through connective tissue [10,18].
This increase in CD1a-stained LCs in OLP highlights the crucial role played by these cells in antigen capture and their subsequent role in antigen presentation to T cells.
Our results differed, however, from those of Sloberg [8], Chou [22], and Farthing [23] who reported no change in number of CD1a-positive LCs in OLP compared with that of healthy oral epithelium. This deviation in the results probably reflects differences in criteria for identification of LC or sensitivity of reagents and standardization of staining techniques or the counting procedures employed by various authors.
However, a study conducted by Mukae [24] gave completely different results wherein they found decreased number of CD1a-positive cells in OLP epithelium compared to healthy mucosa. The recruitment of LC’s in OLP is confirmed [25]; but the discrepancy in the result can be partly explained by anatomical specificity and environment of oral mucosa, which is exposed to various stimuli from food intake, mechanical stress and to some unknown factors [26].
The LCs present the antigenic peptides through MHC molecules which are recognized by the T cell receptors present on the membrane of the both helper and cytotoxic T lypmhocytes. Hence, a close contact of LCs with T cells is a necessary step in carrying out the secondary immune response leading to antigenic destruction.
Finally, the immunopathological changes seen in OLP thus show that LCs are key immune regulators in oral epithelium. They play a decisive role in the pathogenesis of OLP by priming the naive T-cells and inducing the secondary immune response through antigen-specific T cell clones [27]. Similar studies and there results have been tabulated in [Table/Fig-6].
Comparison of the results obtained in the present study with that other similar published literature.
| Component | Control | OLP |
|---|
| LCs in healthy oral mucosa. | Present | Present as showed by Chou [14], Pitigala-Arachi [15], Van Loon [16], Lombardi [17], Hasseus [18], Dorrego [19], Santoro [20], Gustafsson [10] and Mitamura [21] |
| Network of LCs in epithelium. | Present | Also seen in studies conducted by Hasseus [18] and Gustafsson [10] |
| Increase in LCs in OLP compared to normal mucosa. | Present | Similar results by Pitigala-Arachi [15], Hasseus [18], Dorrego [19], Santoro [20], Gustafsson [10] and Mitamura [21] |
| No change in LCs compared to OLP and normal mucosa. | Not seen | Studies by Sloberg [8], Chou [22], and Farthing [23] no change in number of LC’s in OLP compared to normal mucosa. |
| Less number of LCs in OLP compared to normal mucosa. | Not seen | Study by Mukae [24] showed this feature. |
Limitation
The limitations of the study being that the role of LCs in connective tissue region and their subsequent migration and maturation to lymph nodes has not been evaluated and needs a follow-up cellular and molecular level with markers for mature LCs. The active role played by LCs in OLP in antigen presentation throws light on type of cellular interactions involved which can help in better understanding of pathogenesis of this disease which subsequently helps formulating better therapeutic methods which could be developed to act as an adjunct to the role of LCs in antigen presentation and capture.
Conclusion
This study highlights the role played by LCs in antigen capture and presentation to T-cells in OLP characterized by a quantitative increase thus shows importance of this pathogenesis of OLP. The presence of band of T-lymphocytes in connective tissue reveals ongoing interactions in sub-epithelial area. Hence, studies highlighting the role of LCs in connective tissue (mature LCs) and role of antigen specific T-cells should be evaluated for better understanding of pathogenesis of OLP.
Wilcoxon Signed Ranks Test p < 0.001
Wilcoxon Signed Ranks Test p = 0.005
Mann Whitney U Test p=0.001 (Epithelium), p=0.011 (Connective tissue)
[1]. Eisenburg E, Oral lichen planus: a benign lesionJ Oral Max Surg 2000 68:1278-85. [Google Scholar]
[2]. Scully C, El-Kom L, Lichen planus: review and update on pathogenesisJ Oral Pathol 1985 14:431-58. [Google Scholar]
[3]. Xia J, Li C, Hong Y, Yang L, Huang Y, Cheng B, Short-term clinical evaluation of intralesional triamcinolone acetonide injection for ulcerative oral lichen planusJ Oral Path Med 2006 35:327-31. [Google Scholar]
[4]. LoMuzio L, Mignogna MD, Favia G, Procaccini M, Testa NF, Bucci E, The possible association between oral lichen planus and oral squamous cell carcinoma: a clinical evaluation on 14 cases and a review of the literatureOral Oncol 1998 34:239-46. [Google Scholar]
[5]. Kanwar AJ, Handa S, Ghosh S, Kaur S, Lichen planus in childhood: a report of 100 casesClin Exp Dermatol 2009 8:1-5. [Google Scholar]
[6]. Eversole LR, Dam J, Ficarra G, Hwang C-Y, Leukocyte adhesion molecules in oral lichen planus: a T cell-mediated immunopathologic processOral Microbiol Immunol 1994 9:376-83. [Google Scholar]
[7]. Bramanti TE, Dekker NP, Lozada-Nur F, Sauk JJ, Regezi JA, Heat shock (stress) proteins and gamma delta T lymphocytes in oral lichen planusOral Surg Oral Med Oral Pathol Oral Radiol Endod 1995 80:698-704. [Google Scholar]
[8]. Sloberg K, Jonsson R, Jontell M, Assesment of Langerhan’s cells in oral lichen planus using monoclonal antibodiesJ Oral Pathol 1984 13:516-24. [Google Scholar]
[9]. Scully C, Beyli M, Ferreiro MC, Ficarra G, Gill Y, Griffiths M, Update on oral lichen planus: etiopathogenesis and managementCrit Rev Oral Biol Med 1998 9:86-122. [Google Scholar]
[10]. Gustafson J, Eklund C, Wallstrom M, Zellin G, Magnusson B, Hasseus B, Langerin-expressing and CD83-expressing cells in oral lichen planus lesionsActa Odontologica Scandinavica 2007 65:156-61. [Google Scholar]
[11]. Barret AW, Cruchley AT, Williams DM, Oral mucosal langerhans cellsCrit Rev Oral Biol Med 1996 7(1):36-58. [Google Scholar]
[12]. Sugerman PB, Savage NW, Walsh LJ, Zhao ZZ, Zhou XJ, Khan A, The pathogenesis of oral lichen planusCrit Rev Oral Biol Med 2002 13:350-65. [Google Scholar]
[13]. Roopashree MR, Gondhalekar RV, Shashikanth MC, George J, Thippeswamy SH, Shukla A, Pathogenesis of oral lichen planus – a reviewJ Oral Path Med 2010 39:729-34. [Google Scholar]
[14]. Chou LL, Epstein J, Cassol SA, West DM, He W, Firth JD, Oral mucosal Langerhan’s cells as target, effector and vector in HIV infectionJ Oral Path Med 2000 29:394-402. [Google Scholar]
[15]. Pitigala-Arachchi A, Crane IJ, Scully C, Prime SS, Epithelial dendritic cells in pathological human oral tissuesJ Oral Pathol Med 1989 18:11-16. [Google Scholar]
[16]. Van Loon LAJ, Krieg SR, Davidson CL, Bos JD, Quantification and distribution of lymphocyte subsets and Langerhan’s cells in normal human oral mucosa and skinJ Oral Path Med 1989 18:197-201. [Google Scholar]
[17]. Lombardi T, Hauser C, Budtz-Jorgenscn E, Langerhans cells: structure, function and role in oral pathological conditionsJ Oral Pathol Med 1993 22:193-202. [Google Scholar]
[18]. Hasseus B, Jontell M, Brune M, Johansson P, Dahlgren UI, Langerhans cells and T cells in oral graft versus host disease and oral lichen planusScand J Immunol 2001 54:516-24. [Google Scholar]
[19]. Dorrego M, Correnti M, Delgado R, Tapia FJ, Oral lichen planus: immunohistology of mucosal lesionsJ Oral Pathol Med 2002 31:410-14. [Google Scholar]
[20]. Santoro A, Majorana A, Rovers L, Gentili F, Marelli S, Vermi W, Recruitment of dendritic cells in oral lichen planusJ Pathol 2005 205:426-34. [Google Scholar]
[21]. Mitamura J, Onodera K, Ooya K, Histopathological and immunohistochemical study of oral lichen planus in the buccal mucosa: relationship between clinicopathological features and hsitometric analysisOral Med Pathol 2008 13:1-6. [Google Scholar]
[22]. Chou MJ, Daniels TE, Langerhans cells expressing HLADQ, HLA-DR and T6 antigens in normal oral mucosa and lichen planusJ Oral Pathol Med 1989 18:573-76. [Google Scholar]
[23]. Farthing PM, Matear P, Cruchley AT, The activation of Langerhan’s cells in oral lichen planusJ Oral Pathol Med 1990 19:81-85. [Google Scholar]
[24]. Mukae S, Okazaki Y, Tsuda H, Nagai K, Matsumoto N, Ochiai T, Detection of fascin and CCR-7 positive mature dendritic cells in oral lichen planusJ Oral Pathol Med 2009 38:334-42. [Google Scholar]
[25]. Gueiros LA, Gondak R, Junior JJ, Coletta RD, Carvalho AA, Leao JC, Increased number of Langerhans cells in oral lichen planus and oral lichenoid lesionsOral Surg Oral Med Oral Pathol Oral Radiol 2012 113:661-66. [Google Scholar]
[26]. Upadhyay J, Upadhyay RB, Agrawal P, Jaitley S, Shekhar R, Langerhan’s cells and their role in oral mucosal diseasesNorth Am J Med Sci 2013 5:505 [Google Scholar]
[27]. Mizumoto N, Takashima A, CD1a and langerin: acting as more than Langerhan’s cell markersJ Clin Invest 2004 113(5):658-60. [Google Scholar]