A Case of Hepatopulmonary Syndrome
P.B. Sriram1, R. Sindhuja2, M. Natarajan3, P.S. Arul Rajamurugan4, B. Palanikumar5
1 Postgraduate Student, Department of General Medicine, Madurai Medical College, Madurai, Tamil Nadu, India.
2 Postgraduate Student, Department of General Medicine, Madurai Medical College, Madurai, Tamil Nadu, India.
3 Professor, Department of General Medicine, Madurai Medical College, Madurai, Tamil Nadu, India.
4 Assistant Professor, Department of General Medicine, Madurai Medical College, Madurai, Tamil Nadu, India.
5 Assistant Professor, Department of General Medicine, Madurai Medical College, Madurai, Tamil Nadu, India.
NAME, ADDRESS, E-MAIL ID OF THE CORRESPONDING AUTHOR: Dr P.B. Sriram, No.127, Oasis Postgraduate Hostel, Madurai Medical College, Panagal Road, Madurai-625020, Tamil Nadu, India.
E-mail: srirampb@gmail.com
We report the case of a 52-year-old female, a known case of Chronic Liver Disease with portal hypertension. She presented with dyspnoea, platypnoea, melena, cyanosis, clubbing and orthodeoxia. She had oesophageal varices and splenomegaly indicating portal hypertension. Her arterial blood gas revealed hypoxaemia and orthodeoxia. From this clinical background and investigation, a diagnosis of hepatopulmonary syndrome was made. Patient was managed conservatively as she was not willing for liver transplantation.
Arterial hypoxaemia, Cirrhosis, Contrast enhanced transthoracic echocardiography, Liver transplantation, Platypnoea-orthodeoxia
Case Report
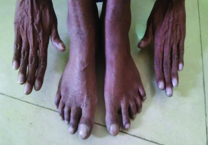
A 52-year-old female, a known case of Chronic Liver Disease with portal hypertension for the past 2 years, presented with dyspnoea on exertion for past 1 month which increases during exertion and also on assuming upright posture. She also had history of abdominal pain & black coloured stools for the past 4 days. On physical examination, the patient had Grade III pan digital clubbing [Table/Fig-1] and central cyanosis [Table/Fig-2]. Her vitals were normal except SpO2 which was 86% in lying posture and became 80% in standing posture after 3 min (Orthodeoxia positive). On abdominal examination spleen was palpable 4cm below left costal margin. Her cardiovascular, respiratory and central nervous system were found to be normal.

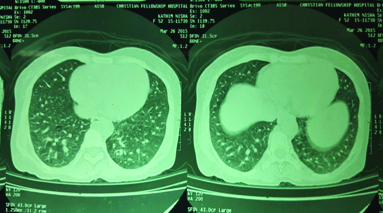
Laboratory investigations showed RBS of around 89 mg/dl with normal blood urea and creatinine. Liver function test revealed SGOT – 38 IU/L(5-35 IU/L); SGPT – 40 IU/L(10-40 IU/L); ALP – 45 IU/L(35-130 IU/L); Total Bilirubin – 0.8; PT – 15 sec, INR-1.4(1.2 control); Total protein – 4.5; S.Albumin – 2.1(3 -5 g); S.Globulin – 2.4(2-3.5 g). Complete hemogram showed high haemogloblin of around 16.8gm%, with total count of 3000 cells/cu.mm and platelet count of 30000 cells/cu.mm. In peripheral smear, RBCs looked normochromic normocytic, WBC count and platelet count were diminished. Viral serological markers were negative and serum ceruloplasmin was 19.3 mg% (25-65mg/dl). Arterial blood gas analysis showed characteristic decrease in pO2 more than 5 mmHg from supine to upright posture indicating opening of intrapulmonary shunting during upright posture resulting in arterial hypoxaemia [Table/Fig-3]. Ultrasound abdomen showed liver span of 10.7cms with dilated portal vein and spleen was enlarged upto 15.9cm. Collaterals were noted in epigastrium. Chest X ray showed reticulonodular pattern on bilateral basal zones[Table/Fig-4]. HRCT of thorax [Table/Fig-5] showed bilateral patchy ground glass opacities in the dependent position. Endoscopy showed small grade I varices and gastropathy (snake skin appearance) with normal duodenum. Echocardiography showed ejection fraction of around 60% with normal left ventricular systolic function. Then we proceeded with contrast echocardiography with agitated saline which showed bubble appearance in left atrium and left ventricle after 3rd cardiac cycle suggestive of right to left intrapulmonary shunting [Table/Fig-6,7]. CT-pulmonary angiography was normal. She was managed with nasal oxygen, diuretics, laxatives, prophylactic antibiotics, β blocker and nitrate to reduce portal hypertension. She was also given 1 unit whole blood transfusion. Her blood counts improved. But her SpO2 remained in the same level. Hence she was prepared for liver transplantation. However she was not willing for transplantation and was managed conservatively.
This Arterial Blood Gas analysis shows characteristic decrease in po2 more than 5 mmHg from supine to upright posture indicating opening of intrapulmonary shunting during upright posture resulting in arterial hypoxaemia.
| Blood gas analysis | Lying posture (37 deg C) | upright posture after 3 mins (37 deg C) | Normal range |
|---|
| PH | ↑ 7.49 | 7.44 | 7.35-7.45 |
| pCO2 | ↓ 22.mmHg | ↓ 24.7 mmHg | 32-48 |
| pO2 | ↓ 60 mmHg | ↓ 55 mmHg | 96-108 |
| cHCO3 | ↓ 16.6 mmol/L | ↓ 16.7 mmol/L | 24-28 |
| Anion Gap | 18.6 mmol/L | 16.8 mmol/L | 10-12 |
| pO2 (A) | 122 mmHg | 119 mmHg | |
| pO2 (A-a) | 62 mmHg | 63 mmHg | |
Chest X-ray showing reticulonodular pattern in the basal zones bilaterally.
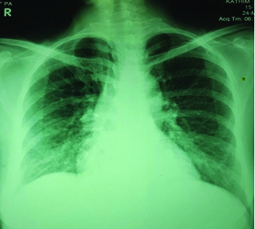
This HRCT shows Bilateral patchy ground glass opacities in the dependent position and evidence of cardiomegaly. No evidence of ILD in this film.
Contrast echocardiography with agitated saline showed bubble appearance in left atrium and left ventricle after 3rd cardiac cycle suggestive of right to left intrapulmonary shunting.

Discussion
Hepatopulmonary syndrome is defined as the triad of liver disease, pulmonary gas exchange abnormalities leading to arterial deoxygenation and evidence of intrapulmonary vascular dilatations [1,2].
Diagnostic criteria for Hepatopulmonary Syndrome consist of the following [3–5]:
The presence of liver disease and / or portal hypertension
An elevated room air alveolar-arterial oxygen gradient {P(A-a)O2 gradient} > 15mmHg or > 20mmHg when age > 65years.
Evidence of intrapulmonary vascular dilatations in the basal parts of the lungs
Absence of other significant cardio pulmonary disease.
Our patient had all the four criteria.
Hepatopulmonary syndrome is an important complication of chronic liver disease, especially cirrhosis with portal hypertension. However there are case reports of patients presenting with hepatopulmonary syndrome due to non- cirrhotic portal hypertension because of inherited thrombophilias (protein C and protein S deficiency) [6]. Prevalence of hepatopulmonary syndrome among patients with chronic liver disease ranges from 4 to 32 percent [2].
Upregulation of endothelial nitric oxide synthase (eNOS) in the intralobar pulmonary arteries, and enhanced eNOS-derived NO production were associated with the development of HPS [7]. The enhanced eNOS-derived NO production may have resulted from pulmonary endothelial endothelin B (ETB) receptor overexpression and increased circulating endothelin-1 (ET-1)-induced vasodilation [8]. The precise trigger for these events remains unclear; however, one possibility is that nitric oxide production is stimulated by absorbed endotoxin (intestinal endotoxaemia), which is less efficiently cleared due to portal-systemic shunting and decreased reticuloendothelial cell function in cirrhosis [9].
Hypoxaemia, platypnoea and orthodeoxia are the other characteristic findings seen in a case of hepatopulmonary syndrome, as present in our case. Intrapulmonary vascular dilatations may be identified by contrast enhanced echocardiography, macroaggregated albumin scanning, and pulmonary arteriography. Contrast enhanced echocardiography is generally preferred because it is more sensitive than macro aggregated albumin and less invasive than pulmonary arteriography [10]. Contrast echocardiography is performed by injecting a contrast material, usually agitated saline and then performing echocardiography. Under normal circumstances, the contrast opacifies only the right heart chambers because it is filtered by the pulmonary capillary bed. However, contrast may opacify the left heart chambers if a right to left intracardiac or intrapulmonary shunting is present. With an intracardiac shunt, contrast appears in the left heart within three heart beats after injection. However, with an intrapulmonary shunt, contrast generally appears in the left heart three to six heart beats after its appearance in the right heart [11] as in our case. Pulmonary arteriography is generally reserved for excluding alternative causes of hypoxaemia, pulmonary hypertension and large direct arteriovenous communications. Three angiographic patterns were described - type 1 minimal pattern which is characterised by normal to finely diffuse, spidery abnormalities. It is associated with severe hypoxaemia, orthodeoxia and a good response to 100% inspired oxygen. The type 1 minimal pattern may evolve into a type 1 advanced pattern, which is characterized by a diffuse spongy or blotchy angiographic appearance. It is less responsive to 100% inspired oxygen. Type 2 discrete pattern was characterized by localized arteriovenous communications and is associated with poor response to supplemental oxygen [12,13]. Prognosis is poor with development of hepatopulmonary syndrome. One observational study reported a higher mortality in patients with HPS compared with case-matched controls (78 versus 43 percent) [14]. Long-term supplemental oxygen is the most frequently recommended therapy for symptoms related to hypoxaemia. Transjugular Intrahepatic Portosystemic Shunt (TIPS) placement is a medical intervention that has been associated with improvement of HPS in several case reports [15]. At present, the most effective and only radical treatment is liver transplantation. Liver transplantation is indicated for patients with incapacitating hypoxaemia due to HPS. Several studies showed decreased shunting with a striking improvement in oxygenation following liver transplantation in some patients [16]. Additionally, observational studies have found that, among patients with and without HPS, the survival is similar in both groups after liver transplantation [17]. These observations led to the acceptance of HPS as an indication to liver transplantation.
Cirrhotic patients who are on the waiting list for a liver transplantation have a shorter survival period if they develop hepatopulmonary syndrome. Therefore, it is suggested that all cirrhotic cases should be followed closely for hepatopulmonary syndrome and should have priority in the waiting list.
Conclusion
Hepatopulmonary syndrome is an important complication of cirrhosis, which causes dyspnoea and cyanosis in patients with liver cirrhosis. There is no relation between the development of hepatopulmonary syndrome and the severity of cirrhosis. Contrast enhanced echocardiography is the simplest and most sensitive way of demonstrating intrapulmonary shunting, thereby confirming the diagnosis of hepatopulmonary syndrome.
Consent
A written informed consent was obtained from the patient.
[1]. Murray KF, Carithers RL, AASLD practice guidelines: evaluation of patient for liver transplantationHepatology 2005 41:1407-32. [Google Scholar]
[2]. Palma DT, Fallon MB, The hepatopulmonary syndromeHepatology 2006 41:617-25. [Google Scholar]
[3]. Rodríguez-Roisin R, Krowka MJ, Hervé P, Pulmonary-Hepatic vascular Disorders (PHD)Eur Respir J 2004 24:861-80. [Google Scholar]
[4]. Colle I, Van Steenkiste C, Geerts A, VanVlierberghe H, Hepatopulmonary syndrome and portopulmonary hypertension: what’s new?Acta Gastroenterol Belg 2007 70:203-09. [Google Scholar]
[5]. Rodríguez-Roisin R, Krowka MJ, Hepatopulmonary syndrome—a liver-induced lung vascular disorderN Engl J Med 2008 358:2378-87. [Google Scholar]
[6]. Maya OGM, Santos OM, Case report of hepatopulmonary syndrome in patients with non-cirrhotic portal hypertensionRev Col Gastroenterol 2012 27(4):319-22. [Google Scholar]
[7]. Fallon MB, Abrams GA, Luo B, The role of endothelial nitric oxide synthase in the pathogenesis of a rat model of hepatopulmonary syndromeGastroenterology 1997 113:606-14. [Google Scholar]
[8]. Tang L, Luo B, Patel RP, Modulation of pulmonary endothelial endothelin B receptor expression and signaling: implications for experimental hepatopulmonary syndromeAm J Physiol Lung Cell Mol Physiol 2007 292(6):L1467-72. [Google Scholar]
[9]. Zhang HY, Han DW, Zhao ZF, Multiple pathogenic factor-induced complications of cirrhosis in rats: a new model of hepatopulmonary syndrome with intestinal endotoxaemiaWorld J Gastroenteroly 2007 13:3500-07. [Google Scholar]
[10]. Abrams GA, Jaffe CC, Hoffer PB, Diagnostic utility of contrast ECHO and lung perfusion scan in patients with hepatopulmonary syndromeGastroenterology 1995 109:1283-38. [Google Scholar]
[11]. Khabbaza JE, Krasuski RA, Tonelli AR, Intrapulmonary shunt confirmed by intracardiac echocardiography in the diagnosis of hepatopulmonary syndromeHepatology 2013 58:1514-15. [Google Scholar]
[12]. Krowka MJ, Dickson ER, Cortese DA, Hepatopulmonary syndrome. Clinical observations and lack of therapeutic response to somatostatin analogueChest 1993 104:515-21. [Google Scholar]
[13]. Krowka MJ, Clinical management of hepatopulmonary syndromeSemin Liver Dis 1993 13:414-22. [Google Scholar]
[14]. Swanson KL, Wiesner RH, Krowka MJ, Natural history of hepatopulmonary syndrome: Impact of liver transplantationHepatology 2005 41:1122-29. [Google Scholar]
[15]. Chevallier P, Novelli L, Motamedi JP, Hepatopulmonary syndrome successfully treated with transjugular intrahepatic portosystemic shunt: a three-year follow-upJ Vasc Interv Radiol 2004 15:647-48. [Google Scholar]
[16]. Gupta S, Castel H, Rao RV, Improved survival after liver transplantation in patients with hepatopulmonary syndromeAm J Transplant 2010 10:354-63. [Google Scholar]
[17]. Deberaldini M, Arcanjo AB, Melo E, Hepatopulmonary syndrome: morbidity and survival after liver transplantationTransplant Proc 2008 40:3512-16. [Google Scholar]