Hajdu-Cheney Syndrome is a rare genetic disorder characterised by progressive focal bone destruction. It is known to be an autosomal dominant disorder but there have been reports of sporadic cases as well. Although the disease manifestation is found to begin from birth, it is most often not diagnosed until adolescence or adulthood. It could be due to the rarity of the condition and the variation of the disease manifestation at different age groups. We report a case of Hajdu-Cheney Syndrome in a 26-year-old male who presented with severe periodontitis and premature loss of teeth. The other characteristic features included craniofacial dysmorphism, abnormalities of the digits and dental anomalies. Patients with craniofacial dysmorphism along with dental abnormalities should be thoroughly examined for any underlying systemic disorder. A team of specialists may be able to diagnose this condition before the disease is advanced.
Acroosteolysis, Craniofacial dysmorphism, Periodontitis
Case Report
A 26-year-old male presented to the dental outpatient clinic with concerns of loose teeth and early loss of teeth. He was born to non-consanguinous parents and his postnatal period was uneventful. He was noted to have global developmental delay and hypotonicity since infancy. There was a history of recurrent upper respiratory tract infections and progressive enlargement of the finger tips. There was no history of any delay in eruption of teeth as well as no visual or hearing impairment. There was no similar history in the family.
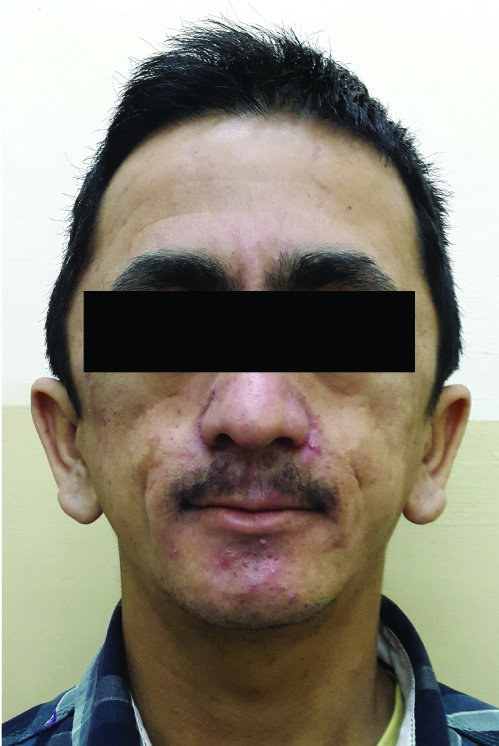
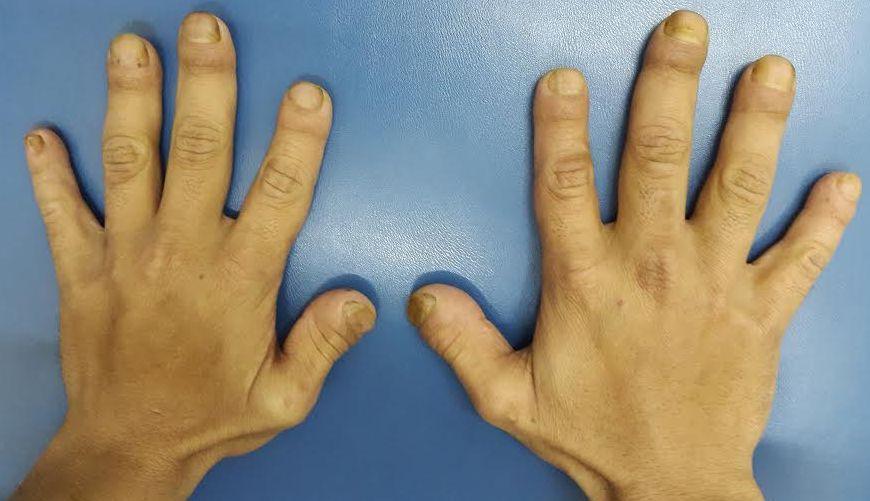
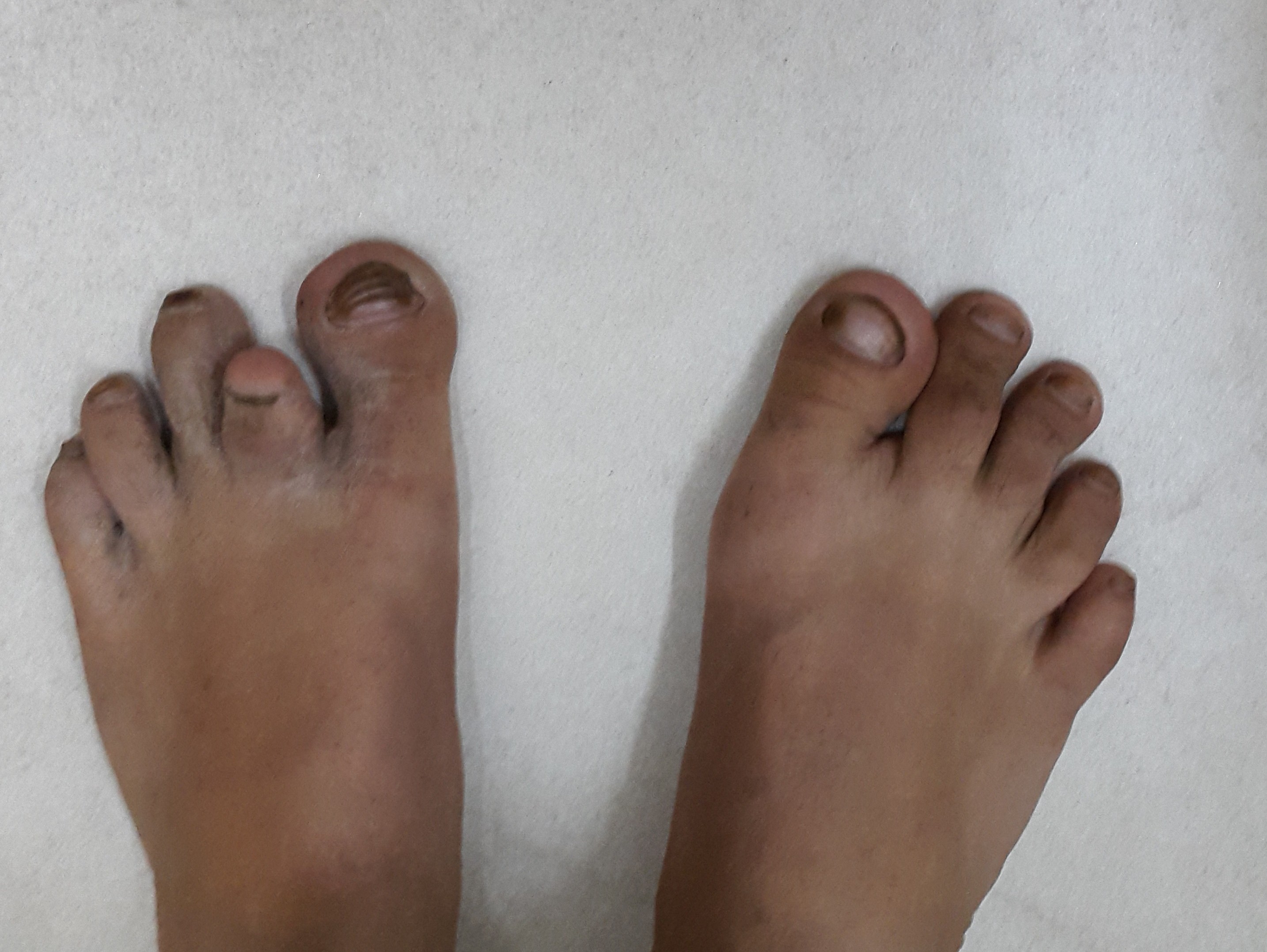
On examination, his height was 165 cm and body weight was 45 kg. His cranial features included a prominent occiput and bi-parietal bossing with a head circumference of 56 cm. He was noted to have thick bushy eyebrows, down slanting palpebral fissures, severe malar flattening, hypoplastic maxilla and low set ears [Table/Fig-1]. He had a hypernasal speech which was difficult to comprehend. The oral findings included a thin upper lip, long philtrum, micrognathia, and high arched palate with bifid uvula. His teeth were malaligned with generalized mobility and few posterior teeth were absent. His finger tips were bulbous with clubbing, toes discoloured and the nails dysplastic [Table/Fig-2a&b]. The joints were found to be lax and hyper flexible. The thoracic cage was narrowed with wide spaced nipple and had mild scoliosis to left.
Facial features showing thick bushy eyebrows, down slanting palpebral fissures, severe malar flattening, hypoplastic maxilla and low set ears
Hands showing bulbous finger tips, clubbing and the dysplastic nails
Feet showing discoloured toes, short left 2nd toe and the dysplastic nails
The biochemical investigations included serum calcium 9.5 mg/dL (8.3–10.4), phosphorus 3.3 mg/dL (2.5– 4.6) serum creatinine 0.9 mg/dL (0.5– 1.4), total alkaline phosphatase 144 U/L (40–125 U/L), serum parathyroid hormone 47.7 pg/mL (8–72 pg/mL) and 25 hydroxyvitamin D 22.8 ng/mL (30–75 ng/mL).
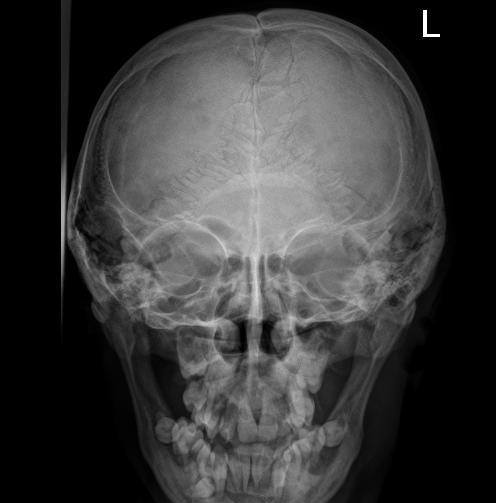
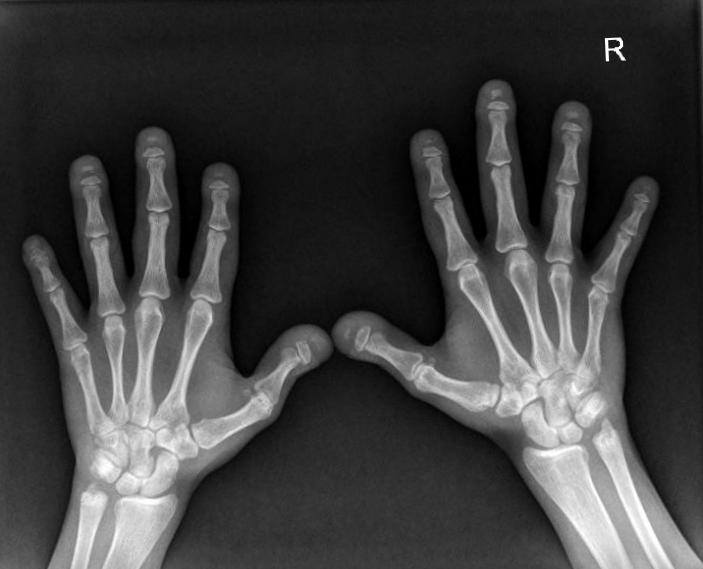
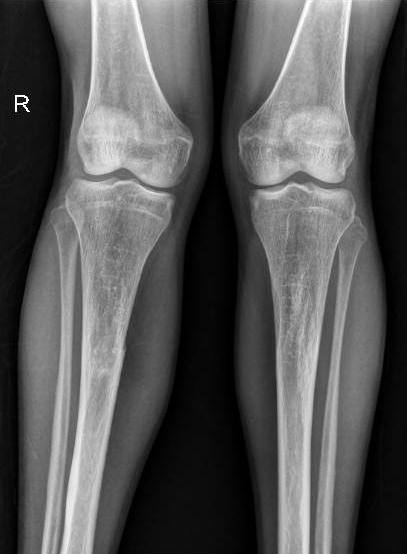
Skull radiography displayed findings of multiple wormian bones along the lambdoid suture, non-fusion of the coronal and lamboid suture, abnormal basi-occiput (bathrocephaly) and thickening of skull base [Table/Fig-3]. Terminal acroosteolysis with normal bone density was noted in hand X-ray [Table/Fig-4]. Radiograph of the leg showed decreased bone density with anterior bowing of tibia as well as transverse metaphyseal and diaphyseal bands [Table/Fig-5].
Skull radiograph displaying findings of multiple wormian bones along the lambdoid suture and non-fusion of the coronal and lamboid suture
Hand radiograph showing terminal acroosteolysis with normal bone density.
Radiograph of the leg showing decreased bone density with anterior bowing of tibia as well as transverse metaphyseal and diaphyseal bands
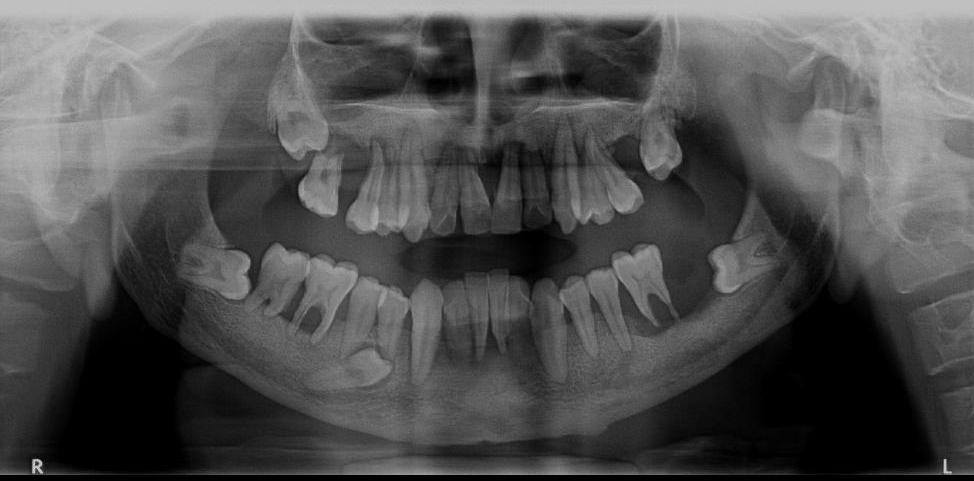
Orthopantomogram revealed hypoplasia of mandibular ramus, generalised atrophy of maxillary and mandibular alveolar bone with absence of a few permanent molars. All the third molars except maxillary right were found to be impacted. An impacted supernumerary tooth was also noted inferior to the mandibular right premolars. Root resorption of multiple teeth was significantly evident [Table/Fig-6]. The characteristic clinical features along with the skeletal radiological findings were diagnostic of Hajdu-Cheney Syndrome.
Orthopantomogram displaying hypoplasia of mandibular ramus, generalised atrophy of maxillary and mandibular alveolar bone with absence of a few permanent molars.
Treatment with bisphosphonates (alendronate) was initiated in addition to calcium and cholecalciferol supplementation. The dental treatment had to be deferred as patient declined consent for dental extractions.
Discussion
Hajdu-Cheney Syndrome is a rare condition characterised with acroosteolysis of the distal phalanges, craniofacial dysmorphism and abnormalities of the teeth and digits. In 1948, Hajdu and Kauntze reported this condition as cranioskeletal dysplasia and Cheney in 1965 included the acroosteolytic feature to this disorder.
Brennan and Pauli in 2011 extensively reviewed 57 cases with Hajdu-Cheney syndrome and noted age specific features of this condition. The characteristic features become more evident with an advancing age although the disease begins to manifest from birth or infancy. They listed out the characteristic clinical craniofacial features such as abnormal dentition, micrognathia, coarse facial features, thick bushy eyebrows, occipital prominence, long philtrum, flat/broad nasal base, telecanthus, mid-facial flattening, short neck, synophrys, coarse hair, low set ears, down slanted eyes [1,2]. The other clinical manifestations included digit abnormalities, joint hypermobility, abnormal speech pattern, coarse skin and recurrent infections. The radiological craniofacial findings include acroosteolysis, decreased bone density, scoliosis, wormian bones, open sutures, bathrocephaly, thickened skull and atrophy of alveolar bone [3,4].
Most of the clinico-radiological craniofacial features listed by Brennan and Pauli were seen in our case. One of the most striking features of this condition is the skeletal involvement. This includes acroosteolysis, fibular deformities, compression fractures, joint laxity, short and broad digits, osteoporosis with fracture and vertebral deformities such as kyphosis or scoliosis [5]. “Pseudo clubbing” of the finger digits is a marked feature and the nails are usually discoloured and dystrophic. Cardiovascular defects, recurrent respiratory infections as well as involvement of renal system have also been reported in patients with HCS. Ramos et al., has described Hajdu Cheney Syndrome and Serpentine fibula-polycystic kidney syndrome as a single entity with distinct phenotype resemblance [6]. Neurological complications such as platybasia and basilar invagination are found to be the associated with this disease [5].
Although the skeletal abnormalities of this disorder are more pronounced, the exact mechanism of the osteolytic nature is yet unclear. The osteolytic lesion in the distal phalanges could be due to the localised increase in bone resorption even though the cause of generalised osteoporosis is unknown [5]. Neovascularisation, inflammation and fibrosis are also frequently seen along with focal osteolysis. Nunziata et al., has reported a marked increase in osteoclastic bone resorption with decreased bone formation and severe osteoporosis in the unaffected radius bone [7].
Dental abnormalities and premature loss of teeth are the usual features seen in association with HCS. Structural changes in the cementum and dentin along with hypoplastic roots are the hard tissue changes that result in premature tooth exfoliation [8]. The possible mechanism for the periodontal disease and early tooth loss is less known [5]. Atrophy of the alveolar bone with an underlying periodontitis is also a common feature. Vingerhoedt et al., noted dental malformation such as fusion of primary incisors, taurodontic molars along with alveolar bone atrophy [9]. An interesting dental finding in our case was the presence of an impacted supernumerary tooth. He was noted to have severe periodontits in advanced stage with early loss of few teeth. It has been recommended that all invasive procedures should be avoided and tooth structure must be preserved as far as possible as the resorption of the alveolar process may continue [10,11]. Currently the skeletal manifestations of patients with Hajdu-Cheney Syndrome has been treated with bisphosphonate therapy alone or in combination with anabolic therapy with teriparatide, although there is no clear evidence that either therapy is beneficial [12]. Invasive dental treatments should be avoided in patients on bisphosphonate therapy as they are known to cause osteonecrosis of the jaw [13].
Conclusion
The diagnosis of HCS is delayed although it has many characteristic features and it could be due its high variability in the disease manifestation. A team of specialists may help in early diagnosis of such a rare condition and treatment can be initiated before the disease has progressed. It is also important to rule out any underlying bone disorder in patient who present with early loss of teeth and periodontitis.
[1]. Brennan AM, Pauli RM, Hajdu-Cheney syndrome: evolution of phenotype and clinical problemsAm J Med Genet 2001 100:292-310. [Google Scholar]
[2]. Gupta SR, Gupta R, Hajdu-Cheney syndrome with osteomyelitis of mandible, calcification of falx cerebri and palatal grooveCleft Palate Craniofac J 2014 51:722-28. [Google Scholar]
[3]. Palav S, Vernekar J, Pereira S, Desai A, Hajdu-Cheney Syndrome: A case report with review of literatureJ Radiol Case Rep 2014 8(9):1-8. [Google Scholar]
[4]. Simpson Michael A, Mutations in NOTCH2 cause Hajdu-Cheney syndrome, a disorder of severe and progressive bone lossNat Genet 2011 43:303-05. [Google Scholar]
[5]. Canalis E, Zanotti S, Hajdu-Cheney syndrome: a reviewOrphanet J Rare Dis 2014 9:200 [Google Scholar]
[6]. Ramos FJ, Kaplan BS, Bellah RD, Zackai EH, Kaplan P, Further evidence that the Hajdu-Cheney syndrome and the serpentine fibula-polycystic kidney syndrome are a single entityAm J Med Genet 1998 78:474-81. [Google Scholar]
[7]. Nunziata V, di Giovanni G, Ballanti P, Bonucci E, High turnover osteoporosis in acroosteolysis (Hajdu-Cheney syndrome)J Endocrinol invest 1990 13:251-55. [Google Scholar]
[8]. Bazopoulou-Kyrkanidou E, Vrahopoulos TP, Eliades G, Vastardis H, Tosios K, Vrotsos IA, Periodontitis associated with Hajdu-Cheney syndromeJ Periodontol 2007 78:1831-38. [Google Scholar]
[9]. Vingerhoedt E, Bailleul-Forestier I, Fellus P, Schoenaers J, Frijns JP, Carels C, Syndrome of Hajdu-Cheney: three case reports of orofacial interestCleft Palate Craniofac J 2010 47:645-53. [Google Scholar]
[10]. Allen CM, Claman L, Feldman R, The acroosteolysis (Hajdu-Cheney) syndrome. Review of the literature and report of a caseJ Periodontol 1984 55:224-29. [Google Scholar]
[11]. Van den Houten BR, Ten Kate LP, Gerding JC, The Hajdu-Cheney syndrome: a review of the literature and report of 3 casesInt J Oral Surg 1985 14:113-25. [Google Scholar]
[12]. Galli-Tsinopoulou A, Kyrgios I, Giza S, Giannopoulou EM, Maggana I, Laliotis N, Two-year cyclic infusion of pamidronate improves bone mass density and eliminates risk of fractures in a girl with osteoporosis due to Hajdu-Cheney syndromeMinerva Endocrinol 2012 37:283-89. [Google Scholar]
[13]. McKiernan FE, Integrated anti-remodeling and anabolic therapy for the osteoporosis of Hajdu-Cheney syndrome: 2-year follow-upOsteoporos Int 2008 19:379-80. [Google Scholar]