Thyroid hormones are invaluable in their role in maintenance of growth, metabolism and mental development in infants and children. Congenital hypothyroidism is the most common preventable cause of mental retardation with an incidence of 1:2500-2800 live births in India [3,4]. Hypothyroidism, during later years, when brain development is completed, leads to slow growth and delayed skeletal maturation [5]. On the other hand, hyperthyroidism is rare in children [4]. It can induce rapid skeletal growth and maturation and other systemic effects due to increased metabolic activity [5].
The prevalence of thyroid dysfunction depends on gender, age, ethnic and geographical background and most importantly on iodine intake. Therefore, data of thyroid disorders from one population cannot be extrapolated to other. Despite the successful implementation and widespread coverage of National Iodine Deficiency Diseases Control Program (NIDDCP) in India, thyroid disorders are still prevalent in many parts of the country [6]. Highlighting the lack of attention paid to thyroid disorders, Thyroid Research and Practice advocated inclusion of thyroid diseases in the list of non-communicable disease of public health importance [7].
This prospective, observational study was carried out to determine the prevalence, clinical profile, aetiology and associated co-morbidities of thyroid dysfunction in children visiting a tertiary care hospital in Western India. These children were also followed up with appropriate therapy to look for response to therapy and complications of the same.
Materials and Methods
Children below 12 years of age who visited the Pediatric Out-Patient Department (OPD) and in-patient department (IPD) of a tertiary care hospital in wetern India were included if they had clinical suspicion of thyroid dysfunction. Ethical clearance was obtained from the Institute Ethics Committee and a written informed consent was taken from either of the parents/ legal guardian. A total of 23,460 OPD patients and 9,351 IPD patients visiting the hospital over a period of one and half years (December 2008 to June 2010) were screened. Inclusion criteria were newborns and children below 12 years with clinical features suggestive of thyroid disorders like prolonged jaundice in newborn, features of cretinism, goiter, physical and mental growth retardation, obesity, constipation etc. Patients who were on any medications known to alter the thyroid hormone status including patients already diagnosed with thyroid disorder on therapy and patient in whom an informed consent could not be obtained were excluded from the study. A total of 498 patients were recruited, 300 from OPD and 198 from IPD. Demographic data (age, sex, address) including family history of thyroid disorders, use of iodized salt, data related to symptoms suggestive of thyroid hypo- or hyper- function (eg. dry skin, lethargy, developmental delay, prolonged neonatal jaundice, constipation) were recorded. Anthropometric measurements were noted and compared with the CDC references. Vital parameters were recorded and complete systemic examination was done. Examination of the thyroid gland was done as per WHO guidelines [8]. Eye signs of hyperthyroidism, e.g. conjunctival chemosis lid retraction, exophthalmos, lid lag, external opthalmoplegia etc., were also looked for [9].
All the 498 children were tested for thyroid profile consisting of Tri-iodothyronin (T3), Thyroxin (T4) and Thyroid stimulating hormone (TSH) levels. Three ml of blood sample was collected under aseptic conditions in a plain vial in the morning after a light breakfast at the RIA laboratory of the hospital. The sample was allowed to clot and then serum was separated after centrifugation at 2000 rpm/min for 30 minutes. The serum was labeled and stored in a refrigerator at -20oC and assays were performed at a later date. The assays used were as follows: T4 - ARCHITECT Total T4 assay (Chemiluminescent Microparticle Immunoassay; Abbott diagnostics, reagent B7K660); T3 - ARCHITECT Total T3 assay (Abbott diagnostics, reagent B7K640); TSH - ARCHITECT TSH assay (Abbott diagnostics, reagent B7K620).
Thyroid dysfunction (hypothyroidism or hyperthyroidism) was defined as per the standard cut offs of T3, T4 and TSH in different age groups [10]. Subsequently, patients who were found to have thyroid dysfunction (excluding infants) had X-ray of left wrist for bone age estimation (N=44). Children with hypothyroidism were also tested and graded for anaemia with estimation of haemoglobin and RBC indices {Mean corpuscular volume (MCV), mean corpuscular haemoglobin (MCH), mean corpuscular haemoglobin concentration (MCHC) and red cell distribution width (RDW)}. Anaemia was defined and treated as per WHO guidelines [11]. Forty two patients with thyroid dysfunction had undergone ultrasound of thyroid gland and in 35 cases I123 thyroid scan was performed. FNAC of thyroid gland was performed in 8 cases of hypothyroidism presenting with solitary thyroid nodule. All the diagnosed cases were treated as per standard guidelines [12] and they were followed up for next 1 year both clinically and with repeat T3, T4 and TSH measurement at 1 month, 3 months, 6 months and at 1 year.
Results
Out of the 498 children tested, 65 were found to have thyroid dysfunction, 61 with hypothyroidism (93.8%) and 4 had hyperthyroidism (6.1%). The demographic characteristic of patients having thyroid hormone abnormalities reveals that most of the patients belonged to 0-1 year age group followed by 9-12 years. The overall male to female ratio was 1:1.2, but a gradual rise in female preponderance of cases was observed with increasing age of patients and the ratio in 9-12 years age group was 1:2.5. Overall, only 15.38 % of patients with thyroid dysfunction had goiter but it was associated with 50% cases of hyperthyroidism [Table/Fig-1].
Distribution of cases of thyroid dysfunction (hypothyroidism and hyperthyroidism) with and without goiter.
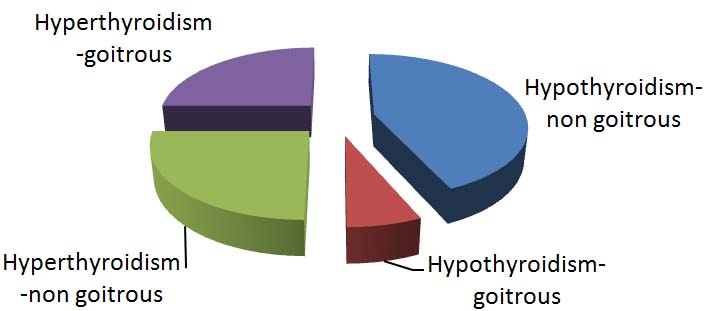
Use of iodized salt was found to be low (68.9%) among children with hypothyroidism, on the other hand, all children having hyperthyroidism were consuming iodized salt. Four out of 61 cases with hypothyroidism had family history of thyroid disorders (6.5%) but none of the hyperthyroid children had positive family history.
There was only 1 patient having secondary hypothyroidism (1.6%) and rest were primary hypothyroidism (98.4%). The most common clinical presentation in patients with hypothyroidism was short stature followed by lethargy. The clinical features of the cases with hypothyroidism are presented in [Table/Fig-2]. All the cases with hyperthyroidism had tachycardia. Other clinical features in these cases are presented in [Table/Fig-3].
Clinical features of cases with hypothyroidism (N=61).
| Clinical features | N (%) |
|---|
| Short stature | 36 (59%) |
| Lethargy | 30 (49.2%) |
| Constipation | 25 (41%) |
| Non pitting edema | 25 (41%) |
| Dry skin | 24 (39.3%) |
| Slow relaxation of deep tendon reflexes | 23 (37.7%) |
| Developmental delay | 23 (37.7%) |
| Pallor | 20 (32.8%) |
| Coarse facies | 18 (29.5%) |
| Delayed closure of anterior fontanel | 13 (21.3%) |
| Prefers heat | 12 (19.6%) |
| Change in voice/ dysphagia | 12 (19.6%) |
| Umbilical hernia | 10 (16.4%) |
| Bradycardia | 9 (14.7%) |
| Prolonged neonatal jaundice | 9 (14.7%) |
| Goiter | 8 (13.1%) |
| Positive family history | 4 (6.5%) |
Clinical features of cases with hyperthyroidism (N=4).
| Clinical features | N (%) |
|---|
| Tachycardia | 4 (100%) |
| Palpitation | 3 (75%) |
| Heat intolerance | 3 (75%) |
| Hyperactivity | 2 (50%) |
| Weight loss | 2 (50%) |
| Increased frequency of stools | 2 (50%) |
| Moist skin | 2 (50%) |
| Polyuria | 2 (50%) |
| Positive family history | 2 (50%) |
| Goiter | 2 (50%) |
| Fatigue | 1 (25%) |
| Tremor | 1 (25%) |
| Ophthalmopathy | 1 (25%) |
There were 10 cases of neonatal hypothyroidism with only one case of transient neonatal hyperthyroidism. The commonest clinical feature in cases with neonatal hypothyroidism was prolonged jaundice (90%) and in 2 cases the mothers were having hypothyroidism on medication. Whereas, the patient with hyperthyroidism had transient neonatal hyperthyroidism presenting with hyperactivity, tachycardia and increased frequency of stools and the mother was a known case of hyperthyroidism on medications.
Bone age estimation was performed in all the cases excluding infants (N=44). In children with hypothyroidism, 32 patients (29.3%) had delayed bone age, while the children with hyperthyroidism had normal bone age. Anaemia was looked for in cases of hypothyroidism (N=61). Total 19 (31.1%) children with hypothyroidism were found to have anaemia, 12 mild and 7 moderate. All the anaemic subjects were having microcytic, hypochromic RBCs, while there was evidence of iron deficiency in 46 (75.4%) hypothyroid children. Thyroid scintigraphy was performed in 35 patients of hypothyroidism and most common finding was dyshormonogenesis (46%) followed by normal uptake of radiotracer (40%). In 8 patients of hypothyroidism in whom FNAC of thyroid gland was performed, 3 patients each had lymphocytic thyroiditis and chronic granulomatous thyroiditis while 2 were diagnosed with benign follicular thyroid. There was no case of malignancy of thyroid gland.
There were a host of co-morbidities identified in 22 children with thyroid dysfunction, the commonest being congenital heart disease. The co-morbidities are presented in [Table/Fig-4].
Co-morbidities in children with thyroid dysfunction.
| Co-morbidities | N |
|---|
| Congenital heart disease | 5 |
| Thalassemia | 4 |
| Cerebral palsy | 4 |
| Down’s syndrome | 2 |
| Diabetes mellitus | 1 |
| Hypopituitarism | 1 |
| Hypoparathyroidism | 1 |
| Extrahepatic portal venous hypertension | 1 |
| Hypophosphatemic rickets | 1 |
| Constrictive pericarditis | 1 |
| Asplenia syndrome | 1 |
All the cases of hypothyroidism (N=61) were treated with L-thyroxin supplementation and 75.4% also received iron supplementation. During follow up over next 1 year, 9 patients (14.8%) of hypothyroidism were lost to follow up. Thyroid function was normalized on therapy in 33 (54%) children, 34 (55.7%) patients achieved normal growth velocity, 4 out of 8 patients with hypothyroidism and goiter had a decrease in their neck swelling. But there was development of features of hyperthyroidism in 2 patients with hypothyroidism requiring frequent follow up and dose adjustment of thyroxine [Table/Fig-5]. Among the 4 cases with hyperthyroidism, one patient had transient neonatal hyperthyroidism and was treated only symptomatically with propranolol alone. Two cases were treated with methimazole and in one of them treatment could be stopped at 1 year. One patient with hyperthyroidism required surgery. All the cases of hypothyroidism with anaemia being treated with iron supplementation were reassessed after 6 months with repeat haemoglobin and RBC indices. Out of 19 patients, 13 were having normal haemoglobin and RBC indices, 3 had haemoglobin within normal limits but still had deranged RBC indices, and 3 were still anaemic, though they all had improvement in their anaemia. All the patients presenting with lethargy had significant improvement in their activity level.
Therapy and treatment outcome of patients with thyroid dysfunction.
| Hypothyroidism |
|---|
| Follow up duration | N | Numberreceivingthyroxine | NumberreceivingIron | Normalthyroidfunctiontest | Normalgrowthvelocity | Featuresof hyperth-yroidism | Decreasein goiter |
|---|
| 1 month | 61 | 61 | 46 | 2 | 0 | 0 | 0 |
| 3 months | 59 | 61 | 45 | 8 | 2 | 1 | 0 |
| 6 months | 56 | 61 | 33 | 16 | 10 | 1 | 1 |
| 9 months | 55 | 61 | 19 | 25 | 19 | 0 | 1 |
| 12 months | 52 | 61 | 08 | 33 | 34 | 0 | 2 |
| Hyperthyroidism |
|---|
| Follow upduration | N | NumberReceivingMethimazole | NumberReceivingpropranolol | NumberRequiringsurgery | Resolution ofGoiter |
|---|
| 1 month | 4 | 2 | 3 | 0 | 0 |
| 3 months | 4 | 2 | 2 | 1 | 0 |
| 6 months | 4 | 2 | 1 | 0 | 1 |
| 9 months | 3 | 2 | 0 | 0 | 0 |
| 12 months | 3 | 1 | 0 | 0 | 0 |
Figures in parentheses are percentages.
Discussion
In this hospital based, prospective study we analyzed the thyroid profile of 498 children aged below 12 years, who were suspected to have thyroid dysfunction on clinical basis. Sixty five children were found to have abnormalities in thyroid profile, 61 had hypothyroidism and 4 were detected to have hyperthyroidism.
Thyroid disorders are well known to be more common in female at all ages [2]. In our study, the overall male to female ratio was 1:1.2, though there was a gradual rise in female preponderance of cases with increasing age of patients and the ratio in 9-12 years age group was 1:2.5. Previously reported studies in Indian children have demonstrated a male: female ratio ranging from 1:2.9 to 1:3.4 [4,13,14], whereas another study from Scotland reported a ratio of 1:2.8 in patients below 22 years of age [15]. Hospital based nature of our study with referral bias could account for this deviation of epidemiology observed. Only 10% of children with thyroid dysfunction in our study had goiter in contrast to an earlier study [4] reporting a prevalence of goiter in 38% of children with thyroid hormone abnormalities. This discrepancy could be accounted for by the differences in local prevalence of iodine deficiency. Out of 65 children with thyroid dysfunction, 93.8% had hypothyroidism which is higher than that reported by previous Indian series in children. In the study of 798 children with thyroid disorder from Mumbai [4], Desai MP et al., have reported 79% of children having hypothyroidism while another small Indian study [14] reported 37% of children having hypothyroidism among the cases with thyroid dysfunction. The possible explanation could be that in our study there were no cases of euthyroid goiter, thereby, making the proportion of hypothyroidism cases disproportionately higher. Among children with thyroid abnormalities, only 6.1% were diagnosed with hyperthyroidism. This is corroborated with previously published reports in Indian children [4,14–16], where a proportion ranging from 2.5-10% was observed.
The clinical features of the cases with hypothyroidism are quite comparable to the previously reported Indian studies [4,17,18]. Only 68.9% of patients with hypothyroidism were found to be using iodized salt historically. Though we did not test the salt samples for iodine content, such low rate of iodized salt intake could account for the hypothyroidism and should arouse concern. Anaemia is known to be associated with hypothyroidism [19]. The commonest type of anaemia found in hypothyroidism is anaemia of chronic disease [20], which manifests with normocytic normochromic blood picture. In our patients, the prevalence of anaemia in hypothyroid children was 31.1%, while the available literature suggests the prevalence to be 20-60% [21]. All our patients had microcytic, hypochromic RBC’s, which could be attributed to hypothyroidism or partly be explained by the high prevalence of iron deficiency in the community [22]. Iron deficiency is observed to be much higher than overt anaemia in hypothyroidism [23] and iron deficiency was even indicated to be associated with subclinical hypothyroidism as well [24]. Treatment of anaemic patients with hypothyroidism responds best when thyroid and iron supplementation is provided together than either given alone [25]. Therefore, all our hypothyroid patients with anaemia were treated with iron supplementation along with thyroxine and the overall response was satisfactory.
Some limitations were noted and must be acknowledged in this study. First, it was a hospital based study, thus the participant may not represent the whole population, and secondly, the number of study subjects was not high, thereby limiting the power of study. Dyshormonogenesis was the commonest abnormality revealed by thyroid scintigraphy. Being autosomal recessive in inheritance, these cases require examination of the sibling and genetic counseling. Despite these limitations, our results indicate a high prevalence of thyroid disorders in children in the community. Moreover, there are very few studies documenting the treatment outcome and follow up in these children [26]. But, we could follow our patients with appropriate therapy to evaluate their response to treatment and outcome which was documented to be satisfactory. Furthermore, a host of co-morbid conditions were seen to be co-existent with thyroid disorders, indicating that, we should be vigilant about thyroid dysfunction while dealing with these conditions. Because disorders of thyroid hormone can compound the outcome of these illnesses, on the other hand these conditions can mask the identification of thyroid disorders. Review of literature reveal that in India less than 10% of cases of congenital hypothyroidism are diagnosed by the age of 3 months and only about 50% by the age of 2 years [27,28]. The poor outcome is reflected in evidence of brain damage in almost half of the children [28]. Lack of awareness amongst the primary health care practitioners and family physicians was one of the important reasons for delayed diagnosis [28].
Conclusion
Thyroid hormones are unique in view of their important role in fetal and early neonatal brain development and also having actions on growth and development during the first two decades of life. The adverse effects of deprivation of thyroid hormone on the rapidly growing infantile brain have prompted the institution of neonatal screening for congenital hypothyroidism thus promoting an early intervention. But in the absence of similar services in India, many cases of hypothyroidism remain unrecognized and may cause havoc in the developing brain. The findings of this study underscore the need to adopt strategies to evaluate children from our community for disorders of thyroid gland.
Figures in parentheses are percentages.