Case Reports
Case 1
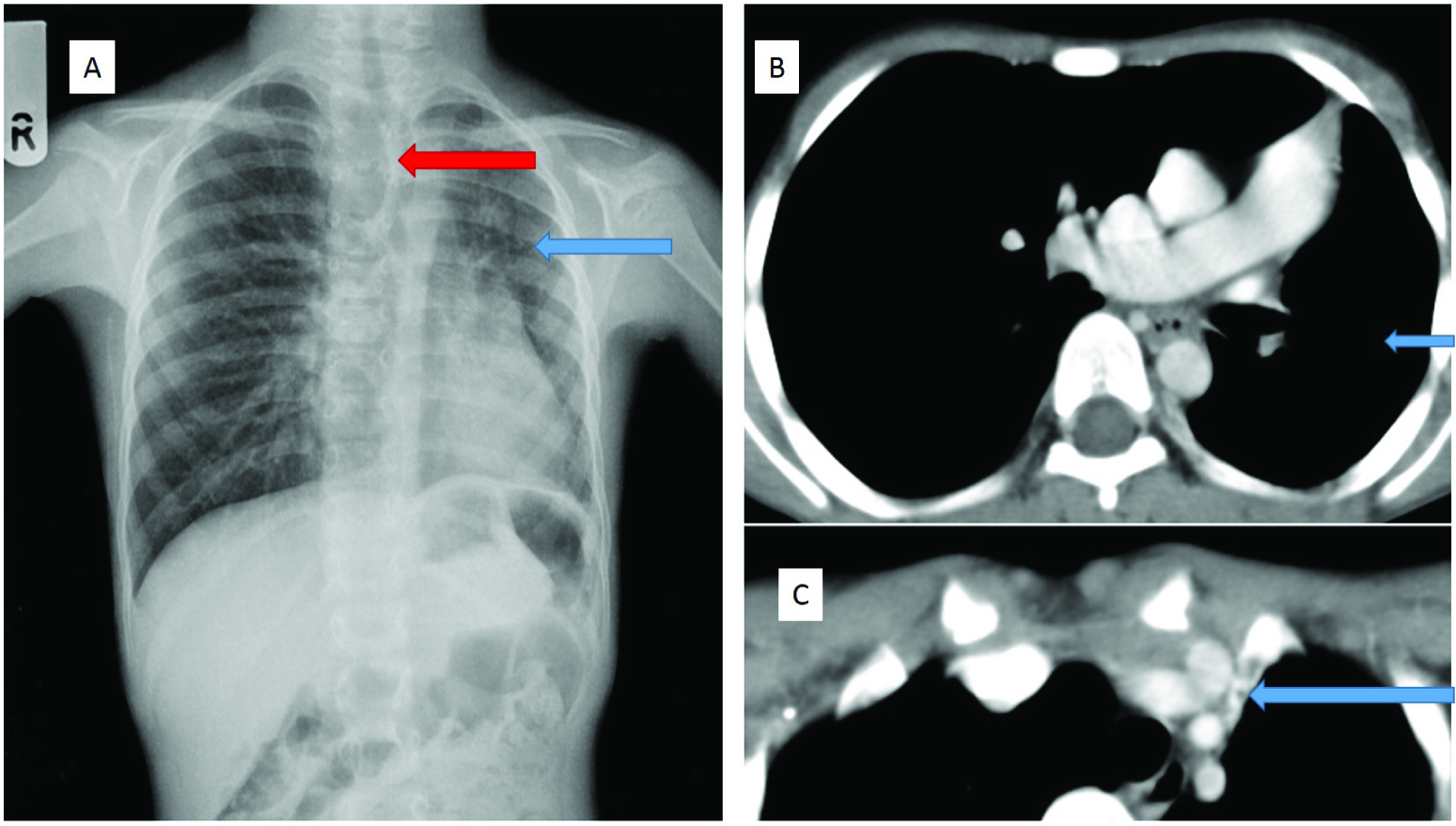
A seven-year-old boy, presented with recurrent respiratory tract infection. The chest radiograph [Table/Fig-1a] showed reduced left lung volume, small left hilum, and hyperlucent contralateral lung herniated across the midline. A diagnosis of left pulmonary hypoplasia was considered initially. Contrast CT thorax confirmed volume loss in the left haemithorax with hyperinflation of the right lung [Table/Fig-1b]. There was non-visualization of the entire left pulmonary artery [Table/Fig-1b], with paucity of pulmonary vasculature, mild bronchiectasis, subpleural and mediastinal systemic collaterals, pleural thickening and subpleural scarring [Table/Fig-1c]. The patient underwent pneumonectomy and the gross specimen showed hypoplastic, pale looking left lung with a leash of telangiectatic vessels and absent left pulmonary artery. Microscopy revealed mild bronchiectasis and interstitial inflammation within the hypoplastic lung.
(a) Chest radiograph PA view shows hypoplastic left lung (blue arrow), with small left hilum and herniation of the right lung (red arrow). (b) Contrast enhanced CT thorax shows non-visualization of the left pulmonary artery, with volume loss in the left lung (blue arrow). (c) Contrast enhanced CT thorax showed a collateral in the mediastinum, which was seen to arise from the left subclavian artery (blue arrow)
Case 2
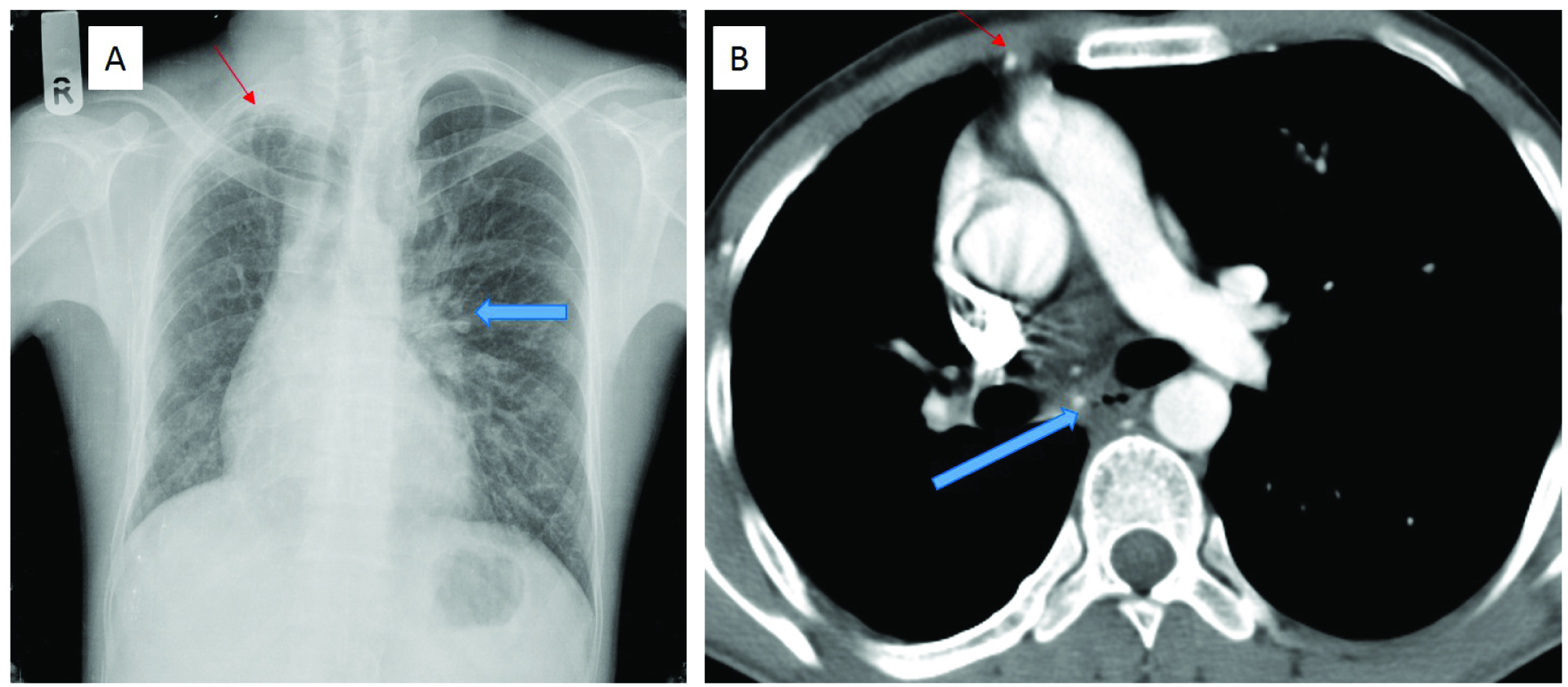
A 27-year-old male patient, with an underlying congenital heart disease (subaortic VSD, mild aortic regurgitation and mild mitral regurgitation) diagnosed on echocardiography, presented with haemoptysis of 3 months duration. Chest radiograph [Table/Fig-2a] showed loss of right lung volume, with paucity of markings. There were linear opacities and pleural thickening in the right upper zone. The left lung showed increased transradiancy and hyperinflation, with prominence of the left hilum. Contrast enhanced CT thorax demonstrated loss of right lung volume, non-visualization of the right pulmonary artery and its branches, and collaterals from bronchial and non-bronchial systemic circulation in the subpleural and intercostal spaces [Table/Fig-2b]. There were discrepancies in the calibre of the intraparenchymal pulmonary arterial branches in the right and left lungs. Right pleural thickening and parenchymal scarring were present. AFB smear was negative and sputum culture showed multiple gram-positive bacteria. The patient was treated with antibiotics and had no haemoptysis at first follow up. He was advised embolization, if haemoptysis recurred.
(a) Chest radiograph PA view shows loss of right lung volume, with paucity of vascular markings and right upper zone linear opacities and pleural thickening (thin red arrow). The left lung is hyperinflated with prominent left hilum (blue arrow). (b) Contrast enhanced CT thorax shows loss of right lung volume, with non-visualization of the right pulmonary artery. Few systemic collaterals are seen (blue arrow). Internal mammary artery is larger than on the uninvolved side (thin red arrow)
Case 3
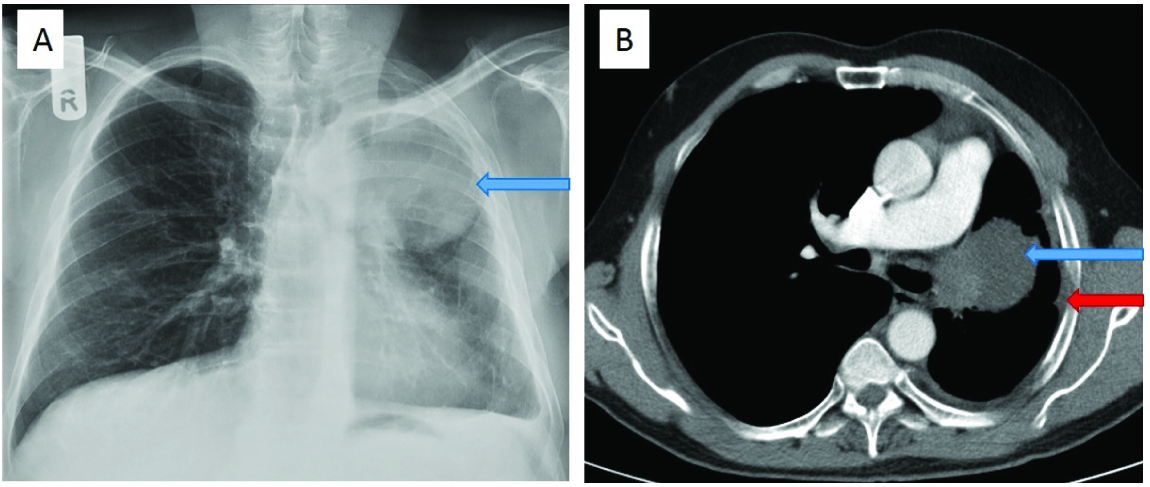
A 57-year-old male patient, diagnosed elsewhere to have non-small cell carcinoma lung, came for a follow up CT after chemotherapy. Chest radiograph demonstrated volume loss of the left lung, and a large mass with a well-defined inferior margin in the left upper zone [Table/Fig-3a]. Contrast enhanced CT thorax showed a left upper lobe mass occluding the corresponding bronchus, with mediastinal invasion. There was complete absence of the left pulmonary artery, which was not due to vascular invasion by the mass or thrombus [Table/Fig-3b]. The patient had no symptoms related to interruption of the left pulmonary artery and the finding was incidental.
(a) Chest radiograph shows volume loss in the left lung, with a mass in the left upper zone (blue arrow). There is paucity of vascular markings on the left side. (b) Contrast enhanced CT thorax demonstrated a left upper lobe mass, occluding the bronchus and invading the mediastinum (blue arrow). Interruption of the left pulmonary artery is seen. Reduced left lung volume and left pleural thickening is seen (red arrow).
Discussion
Proximal Interruption of the Pulmonary Artery is an uncommon developmental anomaly [1]. It occurs more commonly in association with a cardiovascular anomaly, than as an isolated entity. In a review by Pool et al., the isolated form was seen in only 33% of cases [2], with a prevalence of one in 200,000 [3]. The median age at diagnosis of isolated interruption of the pulmonary artery is 14 years (range, 0.1 to 58 years) [4]. The condition is also known as Unilateral Absence of the Pulmonary Artery (UAPA). Three major reviews of world literature on this condition have been published between 1962 and 2002 [2,4,5].
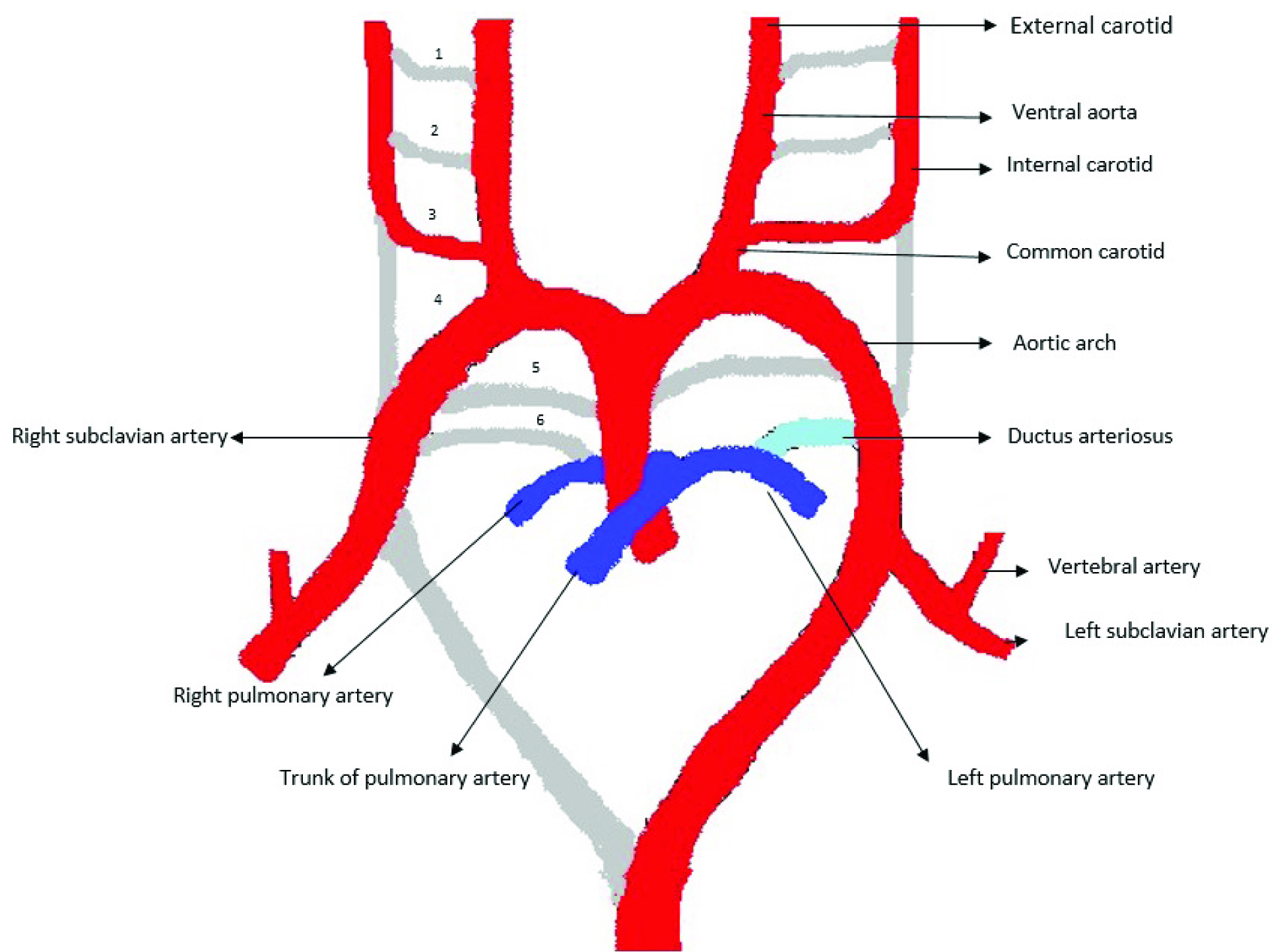
“Interruption” is the term preferred to absence of a pulmonary artery, since the intrapulmonary vascular network is intact [6]. With pulmonary arterial interruption, intrapulmonary vessels continue to develop independently and a well-developed pulmonary arterial tree is present in the lung, with a blind end proximally at the hilum [7]. These vessels receive oxygenated blood through systemic collateral vessels, such as bronchial, internal mammary, innominate and subclavian vessels [8,9]. Normally, the lung develops as a bud from the ventral aspect of the oesophagus, carrying with it blood supply from the splanchnic plexus surrounding the embryonic oesophagus. This network of small blood vessels (the post branchial pulmonary plexus) joins with the proximal limb of the sixth aortic arch. In the fetus, the proximal portion of the left and right 6th aortic arch form the main, right and left pulmonary arteries. Later, the distal portion of the sixth aortic arch atrophies on the right side and persists as the ductus arteriosus on the left, which normally closes after birth [10]. [Table/Fig-4] is a schematic representation of the embryological development of the major neck and thoracic vessels.
Schematic representation of the embryological development of the major vessels in the neck and thorax
One of the three cases we present had an underlying cardiovascular anomaly, known to be associated with Interruption of the Pulmonary Artery. Harkel et al., in their review of world literature, found associations with tetralogy of Fallot, septal defects, co-arctation of the aorta (isolated or in combination with a ventricular septal defect), subvalvular aortic stenosis, transposition of the great arteries (isolated or in combination with ventricular septal defect), pulmonary stenosis, Taussig-Bing malformation and co-arctation, congenitally corrected transposition, pulmonary stenosis and scimitar syndrome [4]. Interruption was an isolated anomaly in the other two cases. [Table/Fig-5,6 and 7] are other cases of pulmonary artery interruption with associated congenital cardiac anomalies.
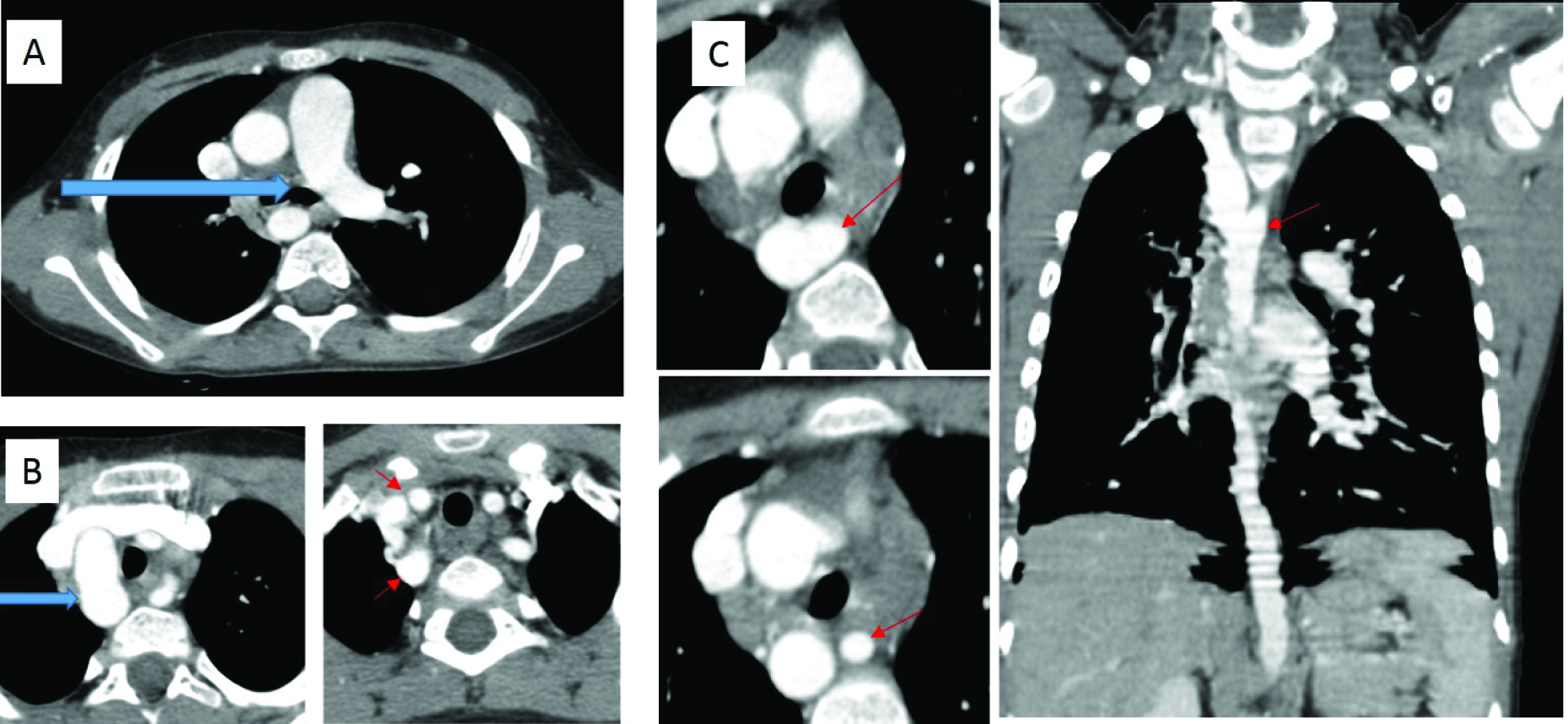
(a) A 5-year-old boy with history of recurrent respiratory tract infections and poor growth since infancy, with subaortic VSD on ECHO. (a) Contrast enhanced CT Thorax shows interrupted right pulmonary artery (blue arrow). (b) Contrast enhanced CT Thorax shows right sided aortic arch (blue arrow) with separate origins of the right subclavian artery and right common carotid artery from the aortic arch (small red arrows). (c) Axial and coronal reconstructed images show the abnormal origin of the left subclavian artery from the proximal descending thoracic aorta (small red arrow)
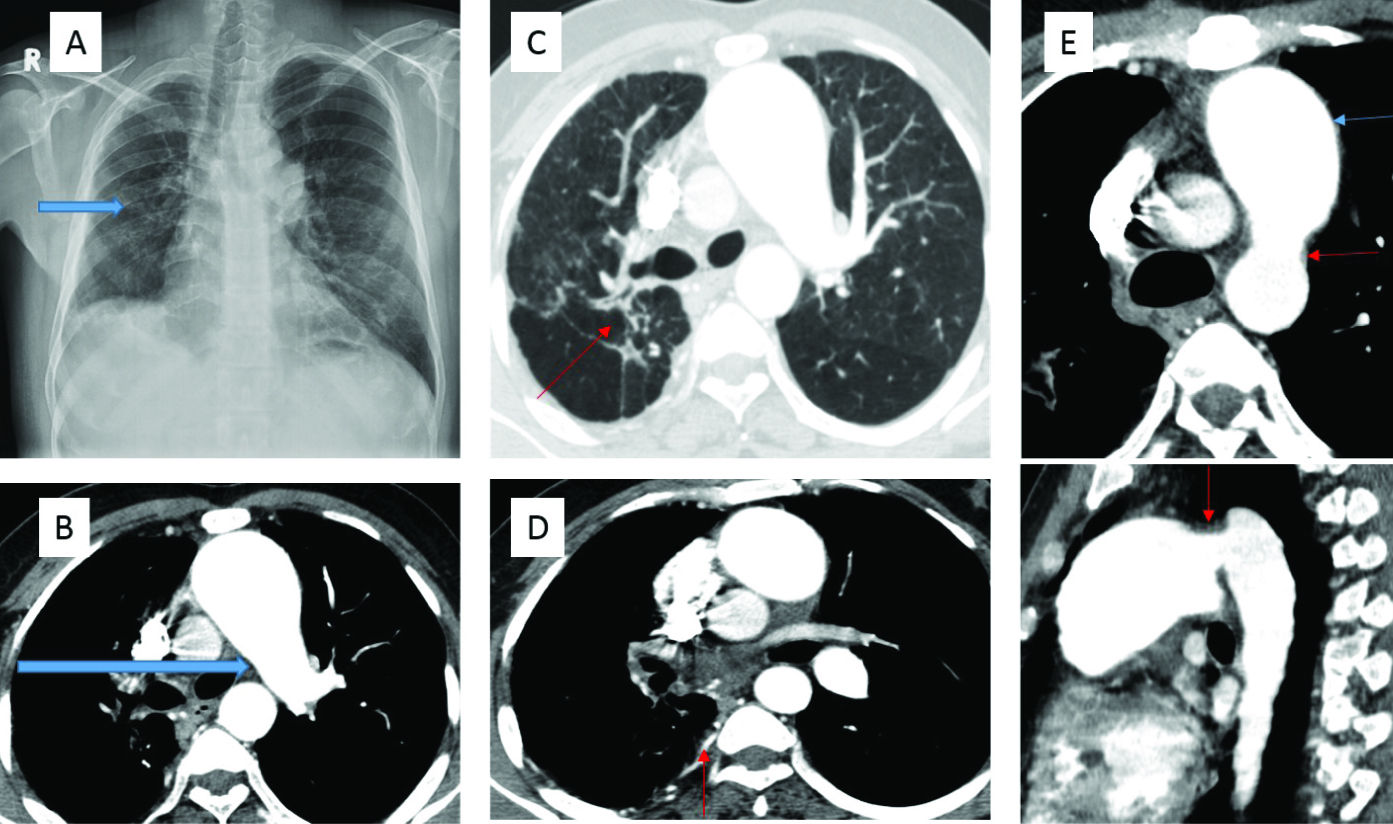
A 36-year-old man with polycythaemia, pulmonary arterial hypertension and bronchiectasis(a) Chest radiograph shows volume loss in the right lung with areas of parenchymal scarring (blue arrow). (b) Contrast enhanced CT Thorax shows interrupted right pulmonary artery (blue arrow). (c) Contrast enhanced CT Thorax in lung window shows parenchymal scarring with bronchiectatic changes in the right lung (small red arrow). (d) Contrast enhanced CT Thorax shows hypertrophied intercostal and bronchial arteries (small red arrow). (e) Axial and sagittal reconstructed images demonstrate the patent ductus arteriosus (small red arrows) with dilated main pulmonary artery (small blue arrow).
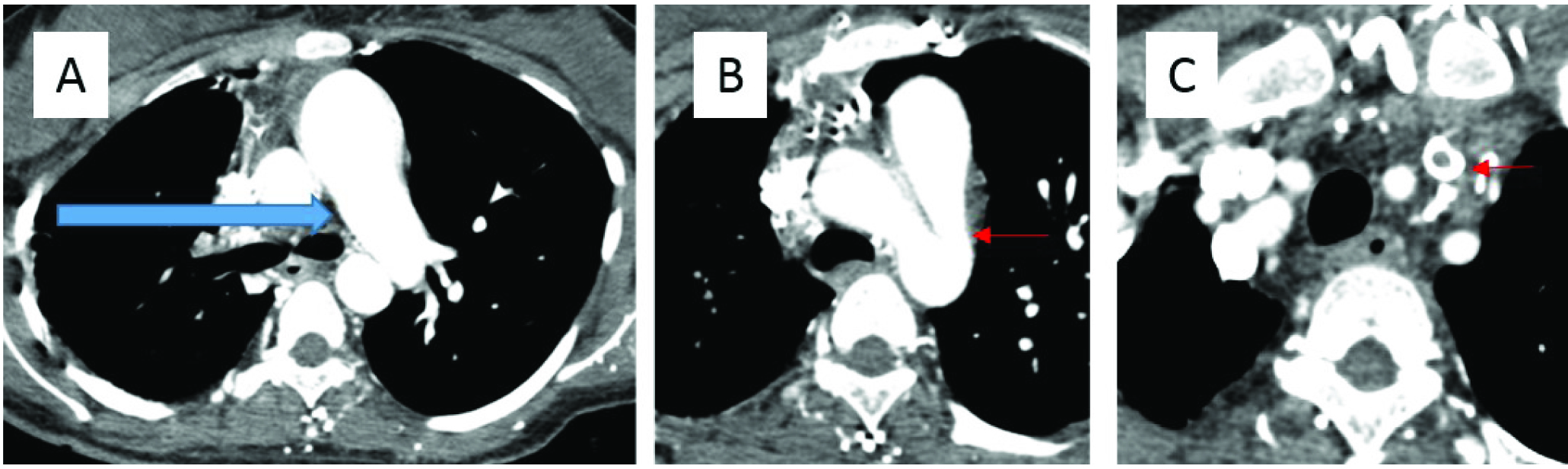
A 22-year-old woman with nephrotic syndrome, presented with cerebral venous thrombosis and dyspnoea. (a) Contrast enhanced CT Thorax shows interrupted right pulmonary artery (blue arrow). (b) Patent ductus arteriosus is demonstrated on contrast enhanced CT Thorax (small red arrow). (c) Contrast enhanced CT Thorax shows a thrombus in the left internal jugular vein (small red arrow).
The symptomatic patient in our series presented at the age of seven, with recurrent respiratory infection. The reasons for this are unclear and include bronchoconstriction as a result of alveolar hypocapnea, decreased mucociliary clearance and defective delivery of appropriate inflammatory cells [3]. Most patients are symptomatic, with recurrent pulmonary infections (37%), dyspnea or limited exercise tolerance (40%), or haemoptysis (20%) [4]. Haemoptysis is secondary to ruptured collaterals between systemic and pulmonary vasculature [11]. It is usually self-limiting, rarely requiring embolization or pneumonectomy [4,6]. The reported incidence of pulmonary hypertension varies from 19% to 44% [2,4].
The asymptomatic patient was detected at the age of 57 years, while on follow up for carcinoma lung. Asymptomatic patients were often diagnosed incidentally due to an abnormal chest radiograph [12]. Harkel et al., in a retrospective analysis of 108 cases (between 1978 and 2000) of isolated interruption of the pulmonary artery from the database of the National Library of Medicine (MEDLINE), found only 13% were asymptomatic [4].
Chest radiographs of patients with interruption of pulmonary artery depict ipsilateral volume loss with mediastinal shift, elevated ipsilateral haemi-diaphragm, paucity of vascular markings, hyperinflation and herniation of contralateral lung. Hypolucent, small lung is attributed to the lung containing fewer and smaller alveoli, due to low oxygen tension. Inapparent or hypoplastic ipsilateral hilum, in contrast to prominent contralateral hilum (secondary to uninterrupted pulmonary artery receiving entire output of right ventricle) is a consistent finding [6]. Fine reticular opacities seen in the periphery of the affected lung are due to collaterals between systemic and pulmonary vasculature and post inflammatory fibrosis. This can be mistaken for Tuberculosis [13]. Pleural thickening and rib notching due to collaterals may be seen [6].
A small haemithorax with small hilum on a chest radiograph may suggest other diagnoses, such as pulmonary hypoplasia, hypogenetic lung syndrome and Swyer-James-Macleod syndrome. Hypogenetic lung syndrome is characterized by a scimitar shaped shadow representing the anomalous draining vein, while Swyer-James-Macleod syndrome shows a small or normal hyperlucent haemithorax, and air trapping on expiratory radiographs or CT [14]. Normal caliber of the pulmonary vessels with normal architecture rules out possibility of interrupted pulmonary artery.
Contrast enhanced CT of the thorax is essential to confirm the diagnosis, and in particular to differentiate it from pulmonary hypoplasia. Pulmonary vessel termination may occur at or within 1cm of the origin. In the cases we present, CT showed complete absence of right or left pulmonary artery. Small ipsilateral haemithorax with herniation of contralateral lung was confirmed. Paucity of normal pulmonary vasculature was striking, and good vascular opacification delineated bronchial and non-bronchial systemic collaterals in the mediastinum, intercostals and subpleural regions, as reported [15]. The MPA was normal in dimension in the two cases not associated with congenital heart disease. In the patient with lung carcinoma, mediastinal invasion occurred in the region of the AP window, and could have been misinterpreted as left pulmonary artery invasion. Other causes of pulmonary hypertension like chronic pulmonary thromboembolism and Takayasu arteritis involving the pulmonary circulation would demonstrate thrombotic pulmonary vessels, increased thickness of vessel wall and caliber variations.
In both cases of isolated interruption of the pulmonary artery in this series, an unusual finding of interruption of the pulmonary artery on the same side as the aortic arch was present. The review by Harkel et al., showed that interruption is more common on the right, seen in 63% of cases. In this review, in only three of 108 cases, was the aortic arch on the same side as the absent pulmonary artery [3,4].
Most of the patients with IPA can remain asymptomatic for a long time. Patients are symptomatic, with recurrent pulmonary infections (37%), dyspnea or limited exercise tolerance (40%), or haemoptysis (20%) [4]. Haemoptysis is usually self-limiting, rarely requiring embolization or pneumonectomy [4,6].
One of the three patients in our series underwent pneumonectomy. Harkel et al., in their review found that surgical procedures were performed in 17% of patients and included pneumonectomy and revascularization surgeries. The overall mortality rate was 7% [4].
Conclusion
When a chest radiograph shows a hypoplastic lung with small ipsilateral hilum, and contralateral hyperinflation of the lung, in asymptomatic patients or those with recurrent respiratory infections or haemoptysis, the possibility of interruption of the pulmonary artery should be considered. Contrast CT allows concurrent evaluation of the bronchial tree and lung parenchyma, and differentiates this condition from pulmonary hypoplasia, hypogenetic lung syndrome, Swyer-James-Macleod syndrome, thromboembolism and arteritis. CT is diagnostic, and obviates the need for angiography, unless embolization is required for control of haemoptysis.
[1]. Fraentzel O, Ein Fall von Abnormer Communication der Aorta mit der Arteria PulmonalisVirchows Arch [A] 1868 43:420-26. [Google Scholar]
[2]. Pool PE, Vogel JH, Blount SG, Congenital unilateral absence of the pulmonary artery: Importance of flow in pulmonary hypertensionAm J Card 1962 10:706-32. [Google Scholar]
[3]. Bouros D, Pare P, Panagou P, Tsintiris K, Siafakas N, The varied manifestation of pulmonary artery agenesis in adulthoodChest 1995 108:670-76. [Google Scholar]
[4]. Ten Harkel AD, Blom NA, Ottenkamp J, Isolated Unilateral Absence of a Pulmonary Artery: A Case Report and Review of the LiteratureChest 2002 122:1471-77. [Google Scholar]
[5]. Shakibi JG, Rastan H, Nazarian I, Paydar M, Aryanpour I, Siassi B, Isolated unilateral absence of the pulmonary artery: review of the world literature and guidelines for surgical repairJpn Heart J 1978 19(3):439-51. [Google Scholar]
[6]. Castaner E, Gallardo X, Rimola J, Pallardo Y, Mata JM, Perendreu J, Congenital and Acquired Pulmonary Artery Anomalies in the Adult: Radiologic OverviewRadiographics 2006 26:349-71. [Google Scholar]
[7]. Anderson RC, Char F, Adams P, Proximal Interruption of Pulmonary arch (absence of one pulmonary artery): case report and new embryological interpretationDis Chest 1958 34:73-86. [Google Scholar]
[8]. Grainger RG, The pulmonary circulation: the radiology of adaptationClin Radiol 1985 36:103-16. [Google Scholar]
[9]. Kieffer SA, Amplatz K, Anderson RC, Lillehei CW, Proximal interruption of a pulmonary arteryAm J Roentgenol Radium Ther Nucl Med 1965 95(3):592-97. [Google Scholar]
[10]. Davies M, Guest PJ, Developmental abnormalities of the great vessels of the thorax and their embryological basisBr J Radiol 2003 76:491-502. [Google Scholar]
[11]. Scheuch RW, Simon-Gabor M, Weinberg HR, Eisenberg H, Left pulmonary artery agenesisN Y State J Med 1988 88(4):200-1. [Google Scholar]
[12]. Werber J, Ramilo JL, London R, Harris VJ, Unilateral Absence of a Pulmonary ArteryChest 1983 84:729-32. [Google Scholar]
[13]. Oakley C, Glick G, McCredie RM, Congenital absence of a Pulmonary Artery: a report of a case with special reference to the bronchial circulation and review of the literatureAm J Med 1963 34:264-70. [Google Scholar]
[14]. Fraser RS, Muller NL, Colman N, Pare PD, Diagnosis of diseases of the chest 1999 Philadelphia, PennsylvaniaW.B Saunders:637-675. [Google Scholar]
[15]. Morgan PW, Foley DW, Erickson SJ, Proximal interruption of a main pulmonary artery with transpleural collateral vessels: CT and MR appearancesJ Comput Assist Tomogr 1991 15(2):311-13. [Google Scholar]