Bone metastases in gynaecological epithelial malignancies have been rarely reported and occur in about 0.2% of the patients [1]. Bone metastases have been reported in 1.1% patients with cervical cancer [2], in about 1.2% patients with ovarian cancer [3], in 1-8% patients with endometrial cancer [4–6] and extremely rare in vulvar cancers [7,8] and vaginal cancers [9]. In an autopsy series, performed from 1948 to 1984, bone metastases in gynaecological malignancies were detected in 16.1% of the patients. Out of these patients, 40.8%, 34.7%, 14.3%, 8.2% and 2% patients had cervical, endometrial, ovarian, vulvar and tubal cancers respectively [10]. Though a relatively uncommon occurrence, bone metastases in gynaecological epithelial malignancies is now being increasingly detected due to the prolonged local disease control in these tumours and due to the easy availability of advanced imaging techniques. Pain is the most common clinical presentation in patients of gynaecological cancers with bone metastases, often accompanied with neurological deficit or pathological fracture [2]. The presence of bone metastases is a poor prognostic indicator [11]. Patients with synchronous extra-osseous metastases and symptomatic treatment without any oncological therapy had further poor prognosis [1]. The objective of the current study is to analyse the clinical characteristics and prognostic variables associated with gynaecological epithelial malignancies with bone metastases.
Materials and Methods
The present study was a single institutional retrospective study done from the data available from the Department of Oncology in the institute from January 2008 to January 2015.
There were a total of 1686 patients with epithelial gynaecological malignancies, who were treated in the institution in that period. Out of them 18 patients had bone metastases. The presence of bone metastases in all the patients were proven by histopathological examination. A Technetium 99m bone scan was also performed in all these patients to detect other sites of bone metastases.
Statistical Analysis
Clinico-pathologic characteristics were tabulated in Microsoft Excel 2013 and data were analysed using SPSS software Version 21. On correlating two variables, p-value< 0.05 was taken to be statistically significant in this study. Survival analysis was done by using Kaplan-Meier method and log-rank test was used to investigate the difference in survivals. Institutional ethical committee approval was taken.
Results
Patient Characteristics
There were 18 patients with epithelial gynaecological malignancies with bone metastases in the current study. The primary site of cancer was cervix, ovary, endometrium and vulva in twelve, three, two and one patient respectively. Out of the 18 patients, only four of them were younger than 50 years the youngest being a 32 years old female with vulvar malignancy. The eldest patient was 62 years of age. The mean age of the patients with epithelial gynaecological malignancies having bone metastases was 51.9 years [Table/Fig-1]. Pain was the most common presenting feature, which was present in 17 patients. Only one patient with metastatic bone disease did not have any pain and presented with neurological deficit alone. Two patients had neurological deficit along with pain. Two patients each had pathological fracture along with pain and fatigue along with pain.
Clinico-pathologic characteristics of epithelial gynecological malignancy patients with bone metastases
| CLINICO-PATHOLOGIC VARIABLES | PRIMARY CANCER SITES |
|---|
| CERVIX | OVARY | ENDOMETRIUM | VULVA |
|---|
| Number of Patients | 12 | 3 | 2 | 1 |
| Age of the Patients |
| < 50 years≥ 50 years | 210 | 12 | 02 | 10 |
| Tumor Histopathology |
| Squamous cell carcinomaAdenocarcinoma | 111 | 03 | 02 | 10 |
| Tumor Grade |
| 123 | 057 | 003 | 002 | 100 |
| Tumor FIGO Stage |
| IIIIIIIV | 0390 | 0021 | 0110 | 0010 |
| Time from primary diagnosis to bone metastases (in months) |
| MinimumMaximumMean | 146038.5 | 162114.3 | 323734.5 | 111 |
| Site of bone metastases |
| Lumbar vertebraPelvic bonesExtremetiesThoracic vertebraSacral vertebraRibs | 4 (33.3%)5 (41.7%)4 (33.3%)3 (25%)1 (8.3%)3 (25%) | 3 (100%)2 (66.7%)0000 | 02 (100%)1 (50%)000 | 001 (100%)000 |
| Number of bone lesions |
| Solitary lesionMultiple lesions | 57 | 12 | 11 | 01 |
| Extraosseous metastases |
| PresentAbsent | 84 | 30 | 02 | 10 |
| Local disease status |
| Local disease controlledLocal failure present | 93 | 03 | 11 | 10 |
| Treatment given for the bone metastases |
| Radiotherapy and ChemotherapyRadiotherapy aloneChemotherapy alone | 1020 | 300 | 101 | 100 |
| Patient status at the end of follow up |
| AliveDead | 66 | 03 | 02 | 01 |
| Mean time from detection of bone metastases to death (in months) | 8.2 | 3.7 | 10.5 | 11 |
FIGO = International Federation of Gynecology and Obstetrics
Pathological characteristics
Twelve of the 18 patients had squamous cell carcinomas on histopathological examination, while the rest had adenocarcinomas. Twelve patients had histological grade 3 tumours at diagnosis, while five patients and one patient had grade 2 tumours and grade 1 tumour respectively. Four, thirteen and one patient had FIGO Stage II, III and IV disease respectively at diagnosis [Table/Fig-1].
Bone metastases
Mean time interval between the diagnosis of the primary tumour to the diagnosis of bone metastases in gynaecological epithelial malignancies were 38.5 months, 14.3 months and 34.5 months in patients with cervical cancer, ovarian cancer and endometrial cancer respectively. Only one patient had bone metastases from a primary vulvar cancer, where the time interval between the diagnosis of the primary tumour and diagnosis of bone metastases was only one month [Table/Fig-1].
The most commonly involved bones in cervical and endometrial cancer were pelvic bones, while lumbar vertebra was the most common site of bone metastases in ovarian cancer [Table/Fig-1]. In the patient with vulvar cancer, the sites of bone metastases were left humerus, left femur and the left fibula. All patients had osteolytic bone lesions [Table/Fig-2], apart from only one patient with ovarian primary who had osteoblastic lesion.
(a) Osteolytic bone metastasis in the head of the left humerus from a primary vulvar cancer. (b) An Tc 99m isotope bone scan image of a patient with primary cervical cancer with multiple bone metastases – Right humerus, lumbar spine, left femur. (c) Pathological fracture of right shaft of femur in a patient of primary cervical cancer. (d) Metastasis to the first lumbar vertebrae in a patient of cervical cancer. (e) MRI picture showing osteolytic lesion in L1 vertebrae in a patient with primary cervical cancer.
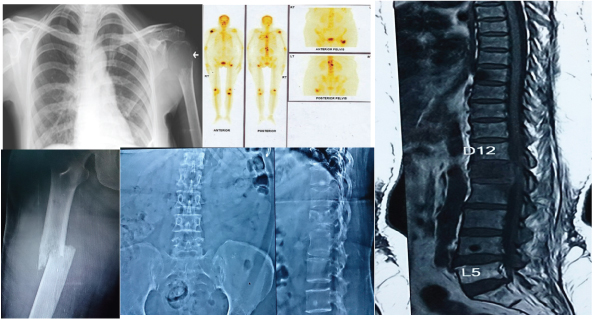
Seven patients had solitary metastatic bone lesion, while the rest had multiple bone metastases [Table/Fig-1]. Twelve of the 18 patients had metastatic disease at sites other than bones also [Table/Fig-1]. Extra-osseous metastases were most commonly seen in lymph nodes. Three of the 12 patients with extra-osseous metastases had lung and liver involvement each. Two patients also had brain metastases [Table/Fig-3]. The extra-osseous metastases were detected along with the presence of bone metastases in 8 out of the 12 patients. Only in three patients with primary cervical cancer and one patient with Stage IV ovarian cancer (with liver parenchymal metastases), extra-osseous metastases was detected before the detection of bone metastases. The mean interval between detection of the extra-osseous metastases and metastases to bones was 4.8 months. Only one patient with cervical cancer had metachronous bone metastases (metastases to bone after 6 months of detection of extra-osseous metastasis). She was detected to have para-aortic lymph node metastasis after 14 months of treatment completion. She was treated with palliative radiotherapy to the para-aortic nodes to a total dose of 54 Gy using 3D conformal treatment. After eight months of this, she developed metastasis to the lumbar vertebra and underwent orthopaedic stabilization. Following screw and plate fixation, she was again treated with palliative radiation of 3 Gy per fraction in 10 fractions over two weeks. She was relieved of her pain following treatment. Local disease control was present in 11 of the 18 patients [Table/Fig-1]. The mean duration for development of bone metastases in these patients with no local failure was 31.45 months. The mean duration for development of metastatic bone disease in patients with local recurrence was 32.7 months.
Sites of extra-osseous metastases in the study patients.
| Site of Extra-osseous metastases | Cervical (n=12) | Ovarian (n=3) | Endometrial (n=2) | Vulvar (n=1) | Total |
|---|
| Lymph Nodes | 6 | 2 | 0 | 1 | 9 |
| Lungs | 2 | 1 | 0 | 0 | 3 |
| Liver | 1 | 2 | 0 | 0 | 3 |
| CNS | 0 | 2 | 0 | 0 | 2 |
| Total patients with extra-osseous metastases | 8 | 3 | 0 | 1 | 12 |
Treatment delivered
Orthopaedic stabilization was done for the metastatic bone disease in five of the study patients. Palliative radiation with 3 Gy per fraction in 10 fractions was delivered to all these five patients postoperatively for pain relief. Three of these five patients also received palliative chemotherapy post-radiation, due to the presence of extra-osseous metastatic disease. Palliative radiation therapy followed by chemotherapy was given in 12 patients. One patient was given chemotherapy alone [Table/Fig-1]. The palliative radiation therapy delivered to the sites of bone metastases was 3 Gy per fraction daily in 10 fractions, weekly five fractions, to a dose of 30 Gy. Palliative chemotherapy was given with single agent Paclitaxel 250 mg/m2 intravenously over three hours every 3 weeks. Patients with brain metastases underwent whole brain irradiation with 3 Gy per fraction, once daily, five fractions per week upto a total dose of 30 Gy. Inspite of some form of treatment, severity of the pain did not improve in four of the 18 study patients (measured by the Visual Analogue Scale). All these four patients had multiple bone metastases and also extra-osseous metastases. All of them were treated with palliative radiation therapy followed by palliative chemotherapy. Performance status assessment of the study patients, before and after treatment was not possible due to the lack of documentation in the case-sheets.
Survival of the patients
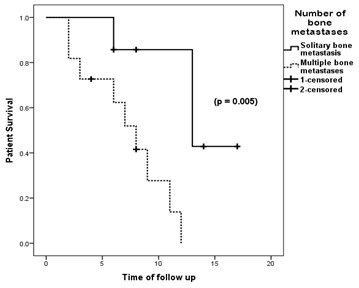
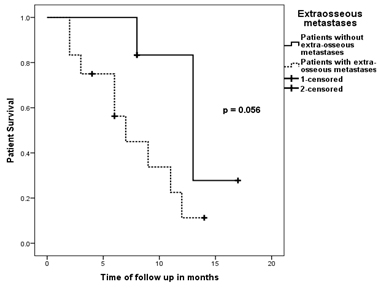
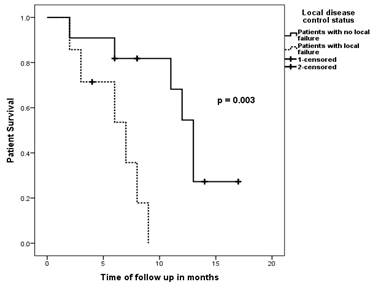
Twelve of the 18 patients in the present study died in the follow up period [Table/Fig-1]. The survival of the epithelial gynaecological malignancy patients with solitary bone metastases had a significantly improved survival (p-value = 0.005) when compared to the patients with multiple metastatic bone lesions [Table/Fig-4]. Also, the survival of the patients without any extra-osseous metastatic disease was near significantly improved (p-value = 0.056) when compared to the patients with extra-osseous metastatic disease [Table/Fig-5]. The survival of the patients with controlled local disease was significantly better (p-value = 0.003) than that of the patients with local failure [Table/Fig-6].
Kaplan-Meier survival curves showing that the survival of the 7 patients with solitary bone metastases (normal line) was significantly higher (log rank test, p-value = 0.005) than the 11 patients with multiple bone metastases (dashed line) with epithelial gynecological malignancies.
Kaplan-Meier survival curves showing that the difference in survival of the 6 patients without extra-osseous metastatic disease (normal line) was near significantly higher (log rank test, p-value = 0.056) than the 12 patients with extra-osseous metastatic disease (dashed line) with epithelial gynecological malignancies.
Kaplan-Meier survival curves showing that the difference in survival of the 11 patients without any local failure (normal line) was significantly higher (log rank test, p-value = 0.003) than the 7 patients with local failure (dashed line) with epithelial gynecological malignancies.
Discussion
Though, there has been few case reportings, the present study is the first Indian retrospective study to report on the characteristics and survival of patients with bone metastases in gynaecological epithelial malignancies. Although bone metastases in gynaecological epithelial malignancies have been infrequently reported in clinical series [1], autopsy series have shown a higher incidence of actual bone metastases in these cancers [10]. Owing to the increased availability of advanced imaging technologies, bone metastases have been increasingly reported nowadays. The mean interval between diagnosis of primary gynaecological malignancy and bone metastases was found to be around 31 months [1]. In the current study, the mean interval between diagnosis of the primary and bone metastases was found to be 31.9 months. In the present study, it was found that most of the bone metastases from gynaecological epithelial malignancies were from primary cervical cancer and rarely from ovarian, endometrial and vulvar cancers. This is in concordance with other clinical and autopsy studies [1,10]. The most common sites of bone metastases were vertebral bodies, with lumbar vertebra being the most commonly involved [1]. Any patient with gynaecological malignancy with unclear bony pains should be suspected to have bone metastases [7] and should undergo plain radiography and radionuclide bone scans [12], as necessary. Petty et al., have stated that technetium bisphosphonate bone scans can detect a metastatic bone lesion approximately 18 months earlier than that detected by a plain radiograph [13]. The metastatic bone lesion is to be then subjected to cytological [14] or histopathological diagnosis [15].
Presence of synchronous non-osseous metastases and symptomatic treatment without any oncological therapy were associated with poor prognosis in these patients [1]. Near significant association of synchronous non-osseous metastatic disease with poor survival was also noted in the present study. But unlike Gottwald et al., the current study demonstrated a significant survival advantage in patients with solitary bone metastases and in patients with controlled local disease [1].
As has been previously validated, some form of oncological therapy results in better survival in these patients [1]. There is no consensus on the treatment of such patients, and treatment should thus be tailored according to the patient’s needs. Although palliative treatment is a cornerstone in the management of these patients, a radical approach may be offered in patients with controlled primary disease, a solitary metastatic bone lesion without any synchronous non-osseous metastases and good performance status. Surgical excision and implantation is a good therapeutic approach in these patients [15]. In the present study, surgical stabilization was performed in five patients along with some other form of treatment. All the patients had pain relief following treatment. Localized radiation therapy in a palliative setting has been delivered in order to prevent impending fracture and for relief of pain in such patients [8]. Palliative radiation therapy was delivered in 17 of the study patients at some point of time. Out of these 17 patients, 14 patients benefitted from the treatment in terms of pain control. As the mechanism of bone metastases is mostly haematogenous, palliative chemotherapy has been given in patients with widespread metastases, mostly along with radiation therapy [7,16]. A comprehensive treatment with radiation therapy and chemotherapy was found to be better than that of radiation therapy or chemotherapy alone [17]. In the present study, radiation therapy and chemotherapy were given in 15 patients, of which 11 patients had relief from pain to some extent. While radiation therapy results in local pain relief, the chemotherapy tackles the systemic spread of the disease. Bisphosphonates have also been used in the treatment of these patients with quite success [18].
Limitations
Due to the rarity of epithelial gynaecological malignancy patients with bone metastases, a large-sample study could not be performed. A multi-institutional large-sample study would be necessary to identify the various prognostic groups in such patients based on survival. Performance status of the study patients, before and after treatment could not be commented upon due to the lack of proper documentation in the case-sheets.
Conclusion
Although rare, bone metastases may be present in patients with epithelial gynaecological malignancies. Lumbar vertebra and pelvic bones are most commonly affected. The probability of bone metastases should be borne in mind in a patient with bony pain and should be investigated accordingly. Multiple sites of bone involvement, failure at the primary tumour site and presence of non-osseous synchronous metastases are poor prognostic indicators. Treatment should be tailored on an individual basis. A radical surgical excision may be a valid option for patients with solitary bone lesion with controlled primary, without any extra-osseous disease. Surgical excision and stabilization followed by radiation therapy with or without chemotherapy produces good relief from pain.