Primary Angiosarcoma of the Breast: An Uncommon Histopathological Subtype
Sunil Vitthalrao Jagtap1, Dhirajkumar Shukla2, Vijay Subhashrao Bonde3, Swati Sunil Jagtap4
1 Professor, Department of Pathology, Krishna Institute of Medical Sciences, University, Karad, Maharashtra, India.
2 Assistant Professor, Department of Pathology, Krishna Institute of Medical Sciences, University, Karad, Maharashtra, India.
3 Assistant Lecturer, Department of Pathology, Krishna Institute of Medical Sciences, University, Karad, Maharashtra, India.
4 Associate Professor, Department of Physiology, Krishna Institute of Medical Sciences, University, Karad, Maharashtra, India.
NAME, ADDRESS, E-MAIL ID OF THE CORRESPONDING AUTHOR: Dr. Sunil Vitthalrao Jagtap, Professor, Department of Pathology, KIMS University, Karad-145539, Maharashtra, India.
E-mail: drsvjagtap@gmail.com
Primary angiosarcoma of the breast is extremely rare malignant tumour of the breast. The cytological and radiological findings are often non specific for diagnosis. Histopathology plays an important role in diagnosis and grading of tumour. Herewith we present a case of 55-year-old postmenopausal woman having history of rapidly enlarging right breast lump with prominent vascularity and associated bluish discolouration of overlying skin. Mammography shows ill defined mass lesion. Fine needle aspiration cytology was done which showed haemorrhagic aspirate with few clusters of highly pleomorphic neoplastic cells. Patient underwent modified radical mastectomy. On histopathological evaluation it showed primary angiosarcoma of the breast, poorly differentiated, grade-III. We are presenting this extremely rare malignancy of breast for its clinical, cytological and histopathological findings.
Breast sarcoma, Histopathology, Mammography
Case Report
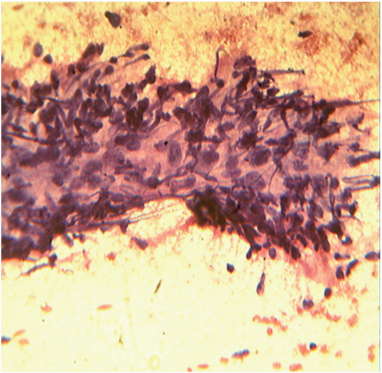
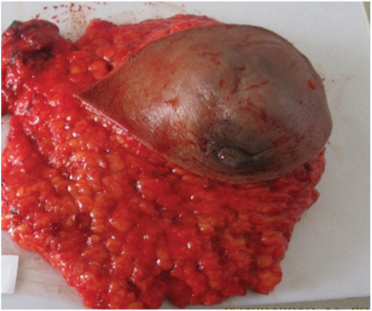
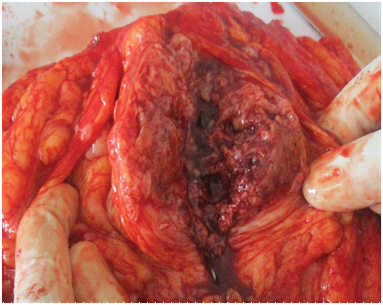
We present a case of 55-year-old woman having rapidly enlarging right breast mass within two months. Mass was situated in right upper inner quadrant of breast. The overlying skin was tense and showed bluish discolouration. The mass was approximately 10x4x3 cm, soft to firm and fixed to underlying tissue. Skin, nipple, areola were unremarkable. Right axillary lymphadenopathy was noted. No history of previous surgery, radiation, any hormone or drug. No contributory family or past history was there. Fine needle aspiration cytology was done from right breast lump, which showed predominantly haemorrhagic material. In areas neoplastic cells were arranged in small sheets, clusters and scattered singly. Individual cells were round to ovoid having moderate to highly pleomorphic hyperchromatic nuclei and scant amount of cytoplasm [Table/Fig-1,2]. Scattered tumour giant cells were noted. The diagnosis was given as positive for high grade malignancy. We received gross specimen of right modified radical mastectomy totally measuring 20x19x6.5cm covered with skin which was shiny, tense and bluish [Table/Fig-3]. On serial cut sections revealed tumour measuring 9x7x4cm located in upper inner quadrant. The tumour was soft to firm in consistency, friable and showed extensive areas of haemorrahge and necrosis [Table/Fig-4]. Many blood filled spaces and friable tissue fragments were noted.
Photomicrograph FNAC showing neoplastic cells with high grade features. (Leishman stain, 400x)
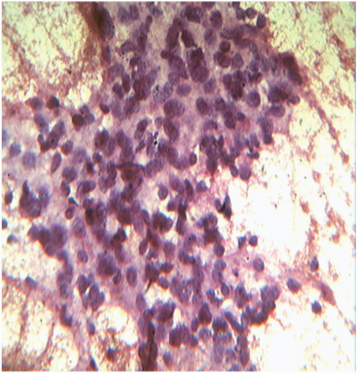
Photomicrograph of FNAC showing high grade neoplastic cells along with spindle cells with malignant features on haemorrhagic background. (Leishman stain,400x)
Gross specimen of right breast totally measuring 20x19x6.5cm covered with skin which was shiny, tense and bluish
Cut section of tumour-soft to firm, friable and shows extensive areas of haemorrahge and necrosis
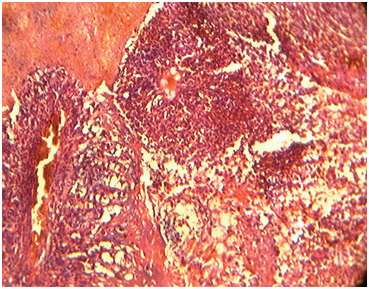
On microscopic examination from multiple sections showed a breast tissue with a tumour. Tumour was composed of predominantly solid areas (70%) of pleomorphic neoplastic cells admixed with spindle cells having compressed or rudimentary lumen formation of blood vessels [Table/Fig-5]. Tumour cells were round to ovoid having highly pleomorphic hyperchromatic or vesicular nuclei with 2-3 prominent nucleoli and moderate amount of eosinophilic cytoplasm [Table/Fig-6]. Numerous mitotic figures are noted. Extensive areas of haemorrhage, necrosis and blood filled with vascular channels are noted [Table/Fig-7]. Reticulin stain showed prominent vascular channels with fibrosis [Table/Fig-8]. The surgical margins, skin, nipple, areola were unremarkable. Section from surrounding breast tissue showed changes of fibrocystic mastopathy with congestion. Right axillary lymph nodes were unremarkable. On histopthological examination diagnosed as features suggestive of high grade malignancy- Primary angiosarcoma of breast poorly differentiated, Grade-III with extensive areas of haemorrhage and necrosis.
Photomicrograph showing tumour is composed of solid areas of pleomorphic neoplastic cells admixed with spindle cells having compressed or rudimentary lumen formation of blood vessels.(H&E stain,100x)
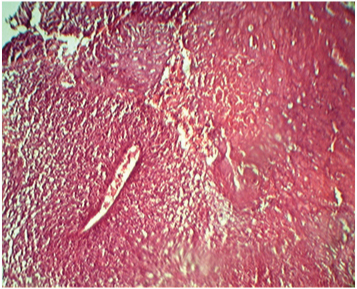
Photomicrograph showing tumour cells having highly pleomorphic hyperchromatic or vesicular nuclei with 2-3 prominent nucleoli and moderate amount of eosinophilic cytoplasm.(H&E, 400x)
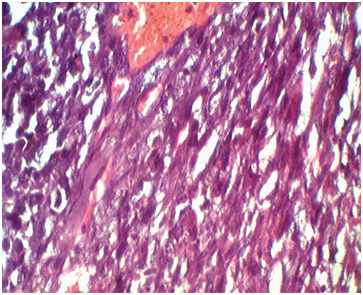
Photomicrograph showing tumour with extensive areas of haemorrhage, necrosis and blood filled vascular channels (H&E stain,100x)
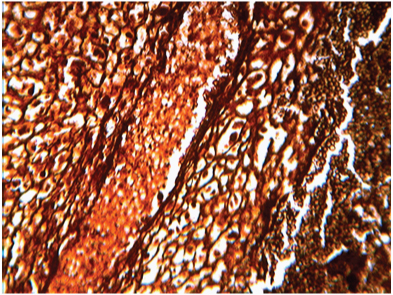
] Reticulin stain showed prominent vascular channels with fibrosis. (400x)
Discussion
Primary angiosarcoma of the breast is extremely rare and accounts for <0.04% of all malignant neoplasms of the breast [1]. The angiosarcoma of the breast is divided into: primary mammary angiosarcoma and cutaneous angiosarcoma.
Primary angiosarcoma of the breast is defined as malignant vascular neoplasm arising within breast parenchyma with or without minor extension into overlying skin. Primary angiosarcoma of breast is rare and arises in patient having no history of treatment for breast cancer, while secondary angiosarcoma occurs mostly after conservation with radiotherapy. Primary angiosarcoma affects relatively younger patients usually at the age of 20-50 years [2]. Patient with primary angiosarcoma may have an insidious onset, usually presents as a palpable mass without pain and with history of rapidly growing size [3]. The tumours which are large and superficial often present with purple blue skin discolouration. In most cases, tumour size at diagnosis is larger than 4cm [4]. Mammographic study of breast usually shows nonspecific changes with ill defined mass without calcification. As many patients are young the dense breast parenchyma may obscure the mass visualization on mammography. The reported incidence showed about 33% of angiosarcomas was not detected on mammography [5]. Sonography with colour doppler and magnetic resonant imaging study may be helpful. The cytological diagnosis on fine needle aspiration cytology is always difficult and reported about 37% of cases with false negative results [6].
In our case on fine neddle aspiration cytology showed predominantly haemorrhagic aspirate with areas showing neoplastic cells having high grade cellular features. So we reported it as positive for malignant cells. The final diagnosis was made on histopatholgy after examining mastectomy specimen. On microscopic evaluation showed predominantly solid pattern of tumour growth with high cellular pleomorphism, high mitotic rate, and extensive areas of haemorrhage and necrosis, which favours the features of poorly differentiated angiosarcoma. However at periphery we noted the tumour with large blood filled spaces with well differentiated areas.
On histopathology angiosarcomas are subclassified into three grades as per classification by Donnel et al., [7]. Grade-I, well differentiated contains open anastomosing vascular channels lined by endothelial cells which dissect through stroma. Grade-II, intermediate grade shows addition to it, foci of solid and spindle cell proliferation. Grade-III, poorly differentiated tumour shows predominantly solid and spindle cell areas, mostly devoid of vascular formations with high mitotic activity. Areas of haemorrages (blood lake), necrosis is extensive. Our case showed features of Grade-III, poorly differentiated angiosarcoma. There was significant axillary lymphadenopathy but on histopathological study, no evidence of nodal metastasis was noted. 0n radiological study there was no evidence of metastasis. The differential diagnosis for grade-I tumour is mostly haemangioma, myoepithelioma, angiolipoma, benign spindle cell proliferation lesion. While for higher grade it should be differentiated from sarcoma- like liposarcoma, fibrosarcoma, malignant phylloides, metaplastic carcinoma etc. Although histopathological findings are conclusive, various immunochemical stains suggested for epithelial markers-(pancytokeratin), for endothelial markers (CD31, CD34, Factor VIII) and other markers for sarcoma will help in finalising the diagnosis.
These cases are mostly treated surgically. The chemotherapy and radiotherapy is as per requirement. The new approach against proliferative endothelium in angiogenesis by agents like bevacizumab or rapamycin [8] will be helpful against this tumour. Our patient underwent surgical excision with chemotherapy. The prognosis in angiosarcoma is poor as it carries moderate risk of local recurrence and high risk of metastasis. The tumour size, presence of residual disease, degree of cellular pleomorphism is the important prognostic factors for angiosarcoma breast [9]. In large series of primary angiosarcoma of the breast, studied by Nascimento AF et al., stated that there is no correlation between histological grade and patient outcome [10]. The overall 5 year survival ranges from 8-50% [11]. Our patient treated with surgical resection followed by chemotherapy and is on regular follow up.
Conclusion
Primary angiosarcoma of the breast is extremely rare malignancy. When breast lesion show large vascular mass, possibility of angiosarcoma should be considered. Although prognosis is bad, surgical excision with histopathological confirmation plays important role in precise diagnosis and further management.
[1]. Vorhurger SA, Xing Y, Hunt KK, Angiosarcoma of the breastCancer 2005 104(12):2682-88. [Google Scholar]
[2]. Scow JS, Reynolds CA, Degnim AC, Peterson IA, Jakub JW, Bonghoy JC, Primary angiosarcoma of the breast: the Mayo Clinic experienceJournal of Surgical Oncology 2010 101(5):401-07. [Google Scholar]
[3]. Georgiannos SN, Sheaft M, Angiosarcoma of the breast: a 30 year perspective study with an optimistic outlookBritish Journal of Plastic Surgery 2003 56(2):129-34. [Google Scholar]
[4]. Yang WT, Hennessy BT, Dryden MJ, Valero V, Hunt KK, Krishnamurthy S, Mammary angiosarcomas: imaging finding in 24 patientsRadiology 2007 242:725-34. [Google Scholar]
[5]. Liberman L, Dershaw DD, Kaufman RJ, Rosen PP, Angiosarcoma of the breastRadiology 1992 183:649-54. [Google Scholar]
[6]. Chen KTK, Kirkegaard DD, Bocian JJ, Angiosarcoma breastCancer 1980 46:268-71. [Google Scholar]
[7]. Donnel RM, Rosen PP, Liberman PH, Angiosarcoma and other vascular tumours of the breastAm J Surg Pathol 1981 5:629-42. [Google Scholar]
[8]. Vemulapalli S, Mita A, Alvarado Y, The emerging role of mammalian target of rapamycin inhibitors in the treatment of sarcomasTargeted Oncology 2011 6(1):29-39. [Google Scholar]
[9]. Bousquet G, Confavreux C, Magne N, Outcome and prognostic factors in breast sarcoma: a multicenter study from the rare cancer networkRadiother Oncol 2007 85:355-61. [Google Scholar]
[10]. Nascimenta AF, Raut CP, Fletcher CDM, Primary angiosarcoma of the breast- Clinicopothologic Analysis of 49 casesAm J Surg Pathol 2008 32(12):1896-904. [Google Scholar]
[11]. Merino MJ, Carter D, Berman M, Angiosarcoma of the breastAm J Surg Pathol 1983 7:53-60. [Google Scholar]