FNAC under USG guidance can be utilized for practically all intra-abdominal space occupying lesions which are clearly delineated by ultrasonography. It has gained tremendous popularity since it can play a vital role in patient management and in many instances, preclude diagnostic surgical procedures or hasten therapeutic planning. With the real-time imaging by USG, there is no ionizing radiation, puncture is possible in any direction and needle position can be constantly monitored [1]. USG guided FNAC is a fairly safe procedure in trained hands and it gives reasonably accurate results, thereby helping the clinician to plan the best line of treatment. However occasionally exact diagnosis is not possible with information obtained by FNA cytological material and the possibility of false negative and indeterminate diagnosis always exists [2]. Thus a negative result should always be correlated with the clinical findings and a repeat aspiration may be necessary. An attempt to obtain additional information can be made via the preparation of CB from any residual material after the conventional smears are prepared. CB analysis can obviate the need for repeat needle passes. Also, CB sections are suited to ancillary techniques like histochemical stains and immunocytochemistry [2,3].
The present study was undertaken to assess the diagnostic accuracy of USG guided FNAC in the diagnosis of intra-abdominal masses, as well as to assess the usefulness of routine use of CB technique on cytological specimens.
Materials and Methods
Sixty two patients with clinically and/or radiologically detected intra-abdominal masses diagnosed on FNAC in the Department of Pathology during a 21 month duration in a tertiary care institution form the material for this study. Consent for this study was taken from the ethical committee of the institute. The intra-abdominal site of the lesions sampled included the liver, the retroperitoneum, abdominal lymph nodes, intestine, pancreas, stomach, kidney, omentum and mesentery. Pelvic masses were excluded from the study. Clinical data with emphasis on history, physical examination and relevant investigations were analysed thoroughly and discussed with the referring physician. Patients with severe coagulopathy and cases like haemangioma and hydatid disease of liver were considered as contraindications for FNAC in view of possible serious complications. The procedure of FNA was explained to the patient seeking an informed consent and complete co-operation for the procedure. The mass under investigation was evaluated by USG and the path of FNA providing the safest and easiest access to the lesion was chosen. Under aseptic precautions, FNAC was performed using 20 or 22 gauge, 90 mm spinal needle attached to a 10 ml disposable syringe. The prepared smears were stained with Papanicolaou and Haematoxylin and Eosin stains (H&E) stains. Periodic acid-Schiff, Gram’s and Ziehl-Neelsen stains were done wherever required. For CB analysis, all macroscopically visible tissue fragments in the aspirated material were collected in 10% formaldehyde solution and sedimented by centrifugation. The packed sediment was processed for CB from which H&E histologic sections were prepared. The aspirate smears and CB sections were evaluated and the final cytomorphologic diagnosis offered was correlated with histopathologic diagnosis (obtained from biopsy and/or surgically resected specimen), follow-up and response to therapy information. Statistical data pertaining to true positive, true negative, false positive and false negative rates as well as sensitivity, specificity and accuracy rates of USG guided FNAC as a diagnostic procedure was assessed in the 56 cases in which correlation was available. For statistical purposes, non malignant included benign tumours and non-neoplastic lesions.
Results
The 62 cases were categorized into four groups based on the final cytologic interpretation: non- neoplastic (16 cases), benign (2 cases), malignant (42 cases) and unsatisfactory for interpretation (2 cases).
Out of 62 patients, 42 (67.74%) were males and 20 (32.26%) were females [Table/Fig-1]. Among 42 lesions in the male patients, a majority were malignant accounting for 30 (71.43%) lesions and 11 (26.19%) lesions were non-neoplastic, and one (2.38%) was benign. Among the 20 lesions in the female patients, 12 (60%) were malignant, five (25%) were non-neoplastic, one (5%) was benign and two (10%) were unsatisfactory for evaluation. Overall the diagnostic yield from the USG guided FNAC was 96.77%. Majority of the lesions were located in the liver [Table/Fig-2] and most of them were malignant lesions comprising 11 Hepatocellular Carcinoma (HCC), seven metastatic carcinoma and one case of Cholangiocarcinoma (CC). An attempt to grade the HCC was made, nine cases were well to moderately differentiated and two were poorly differentiated [Table/Fig-3a,b]. All the smears were moderate to highly cellular and were evaluated for three microscopic patterns namely trabecular, pseudoglandular and compact/solid. They were also compared with the pattern in the corresponding CB preparations [Table/Fig-3c,d]. In trabecular pattern the smears and cell block had two sub-patterns namely central sinusoidal and peripheral sinusoidal. Among the seven cases of metastatic carcinoma [Table/Fig-4a,b], the clinical history and radiological findings suggested stomach as the primary site in one case but the primary site was not ascertained in other cases. However, in all the seven cases CB preparations supported the diagnosis. The CB had insufficient material and no biopsy confirmation was available in the case of CC.
Age-gender distribution of cases
| Gender | No. of cases | 1-10 years | 11-20 years | 21-30 years | 31-40 years | 41-50 years | 51-60 years | 61-70 years | 71-80 years | 81-90 years |
|---|
| Male | 42 | 02 | 02 | 06 | 07 | 14 | 05 | 02 | 03 | 01 |
| Female | 20 | 01 | 02 | 03 | 07 | 03 | 02 | 01 | Nil | Nil |
| Total | 62 | 03 | 04 | 09 | 14 | 17 | 07 | 03 | 03 | 01 |
Organ distribution of different intra-abdominal masses
| Organ | Total (%) | Non-neoplastic | Benign | Malignant |
|---|
| Liver | 24 (40) | 05 | Nil | 19 |
| Retroperitoneum | 07 (10) | Nil | Nil | 07 |
| Abdominal lymphnode | 08 (13.3) | 03 | Nil | 05 |
| Intestine | 11 (18.3) | 07 | 01 | 03 |
| Pancreas | 04 (06.7) | 01 | Nil | 03 |
| Stomach | 02 (03.3) | Nil | Nil | 02 |
| Kidney | 02 (03.3) | Nil | Nil | 02 |
| Omentum | 01 (01.7) | Nil | Nil | 01 |
| Mesentery | 01 (01.7) | Nil | 01 | Nil |
| Total | 60 (100) | 16 | 02 | 42 |
(a) FNAC Hepatocellular carcinoma, well differentiated. Round central nuclei, large nucleoli & intranuclear inclusion (arrow) (H&E,x400). (b) FNAC Hepatocellular carcinoma. Characteristic atypical hepatocytic nuclei (arrow) (H&E,x400) (c) Cell block Hepatocellular carcinoma. Trabecular growth pattern.(H&E,x400) (d) Cell block Hepatocellular carcinoma. Pseudoglandular pattern.(H&E,x400)
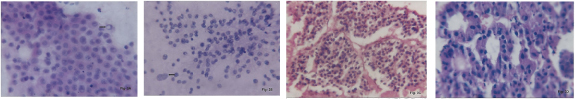
(a) FNAC Liver. Metastatic poorly differentiated carcinoma. Cells with scant cytoplasm & pleomorphic nuclei. Single normal hepatocyte is seen (arrow). (H&E,x400)(b) Cell block Liver. Metastatic poorly differentiated carcinoma. (H&E,x100).
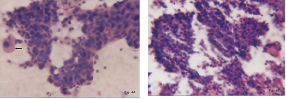
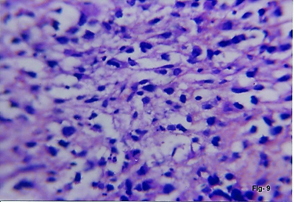
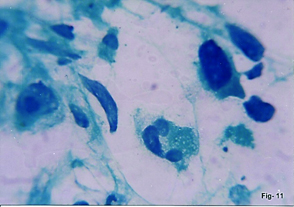
The next most common site of malignant lesion was retroperitoneum comprising four sarcomas, two germ cell tumours (GCT) and one metastatic carcinoma in para-aortic lymph node. The sarcomas included one case each of Leiomyosarcoma (LMS), Neurofibrosarcoma (NFS), Pleomorphic Malignant Fibrous Histiocytoma (MFH) and Pleomorphic Liposarcoma (LS) [Table/Fig-5,6,7 and 8]. Aspirate yielded sufficient material for CB study in MFH and LS [Table/Fig-9]. Surgically resected specimens were available in all the cases. The patient of NFS was a 33-year-old female with typical clinical features of neurofibromatosis. Cytological diagnosis of primary gonadal GCT was made in two patients, both with bilateral undescended testis. One case was seminoma and the other was embryonal carcinoma, both showed characteristic cytological features in smears as well as CB sections [Table/Fig-10a&b,11]. FNAC of para-aortic lymph node mass showed metastatic adenocarcinoma.
FNAC Retroperitoneal leiomyosarcoma. Cells with indistinct cytoplasmic borders & elongated, blunt ended nuclei. (H&E,x400)
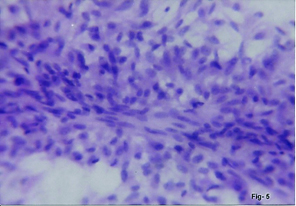
FNAC Retroperitoneal neurofibrosarcoma. Atypical cells with elongated buckled nuclei (arrows). (H&E,x400)
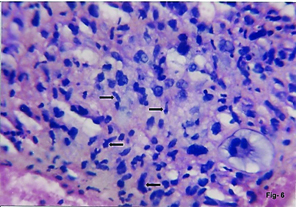
FNAC Retroperitoneal malignant fibrous histiocytoma. Sheet of bizarre cells with abundant eosinophilic cytoplasm. (H&E,x400).
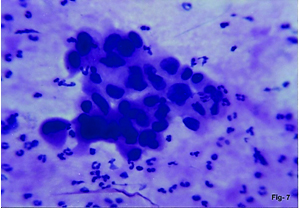
FNAC Retroperitoneal liposarcoma. Poorly cohesive pleomorphic cells. Multinucleate giant cell (arrowhead) and univacuolate lipoblast with indentation of nucleus (arrow). (H&E,x400).
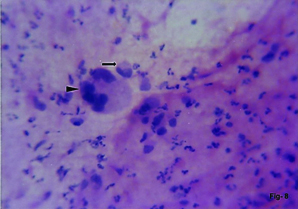
Cell block Retroperitoneal liposarcoma. Univacuolate and multivacuolate lipoblasts. (H&E,x400).
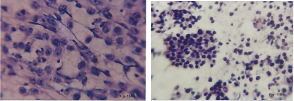
(a) FNAC Cryptorchid testis. Seminoma. Dispersed cells with fragile cytoplasm with vesicular nuclei with multiple nucleoli. Lymphocytes & eosinophils are seen. (H&E,x400). (b) Cell block. Cryptorchid testis. Seminoma. Uniform cells with clear fragile cytoplasm. Prominent lymphocytic infiltrate.(H&E,x400)
FNAC Cryptorchid testis. Embryonal carcinoma. Pleomorphic cells having hyperchromatic nucleus with thick & uneven nuclear membrane & huge nucleoli.(Pap,x1000)
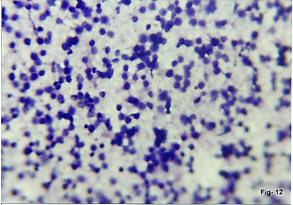
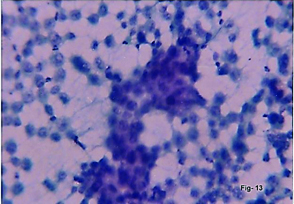
Mesenteric lymph node aspirates were malignant in five cases consisting of three cases of non-Hodgkin’s lymphoma [Table/Fig-12] and two metastatic carcinoma. No CB material was available in any of these cases. The other malignant lesions included adenocarcinoma of intestine (2) [Table/Fig-13], pancreas (3) [Table/Fig 14a&b], stomach (2) [Table/Fig-15] and omentum-metastatic (1), intestinal lymphoma (1), Wilm’s tumour (1) and renal cell carcinoma (1) [Table/Fig-16a&b].
FNAC Mesenteric lymph node. Non-Hodgkin lymphoma. Monomorphic cell population showing coarse chromatin & prominent nucleoli (H&E,x100)
FNAC Colon. Adenocarcinoma. Malignant cells with vesicular nuclei and prominent nucleoli. (Pap,x400)
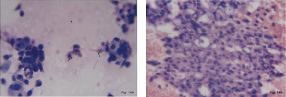
(a) FNAC Pancreas. Adenocarcinoma poorly differentiated. Pleomorphic cells with fragile cytoplasm. (H&E,x400). (b) Cell block Pancreas. Adenocarcinoma, poorly differentiated. Epithelial cells with hyperchromatic nucleus and granular to vacuolated cytoplasm
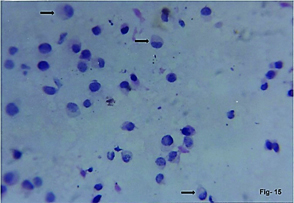
FNAC stomach. Adenocarcinoma showing partial signet ring formation (arrows). (H&E,x400)
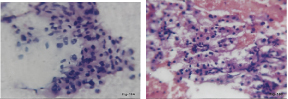
(a) FNAC. Renal cell carcinoma. Pleomorphic cells with prominent nucleoli and abundant granular to vacuolated cytoplasm(H&E,x400). (b) Cell block. Renal cell carcinoma (H&E,x400)
The two benign tumours consisted of a rare case of mesenteric lipoma and a case of benign spindle cell tumour of intestine. The non-neoplastic cases were constituted by three each of supppurative lesions (two hepatic and one pancreatic), cirrhosis, dysplasia (intestine) and non-specific inflammatory lesion (appendicular lump). There were two cases of tuberculosis mesenteric lymphadenitis, one case of intestinal tuberculosis and one case of non specific lymphadenitis.
CB material was available in 45 out of 60 cases with satisfactory aspirate smears. In all these cases the cytomorphologic opinion was compatible with that of the aspirate smears. However in seven cases including one metastatic carcinoma (lymph node), one poorly differentiated carcinoma (pancreas), two poorly differentiated HCC and three cases of tuberculosis (TB), CB cytomorphologic features substantially reinforced the cytological diagnosis on aspirate smears.
The histopathologic material in the form of biopsy material and/or resected specimen was available for correlation in 46 cases including 41 malignant, two benign and three non-neoplastic cases. Cytodiagnosis corroborated with histologic diagnosis in 44 cases. Two false negative cases were noted comprising benign spindle cell tumour of intestine and cirrhosis of liver which were histologically proven as LMS and HCC respectively. Follow up information of response to therapy available in 10 non-neoplastic cases consisting of suppurative lesions, non-specific inflammatory lesions, non-specific lymphadenitis and tuberculosis lesions was considered conclusive of cytodiagnosis.
Discussion
Abdominal masses are a clinical enigma and always pose a dilemma for the surgeon. Differentiation between non-malignant and malignant lesions is vital, especially in advanced unresectable malignant cases to avoid an exploratory laparotomy. FNAC is considered to be more sensitive and accurate than needle core biopsy [4]. The use of USG guidance for needle placement allows aspiration of representative material for precise cytological diagnosis [5]. The residual material in the needle hub after preparation of routine smears may contain diagnostic material. Studies have evaluated the utilization of this material in the form of CB’s which may also help in reducing the number of re-aspirations which are uncomfortable for the patient [2,3,6–8]. USG guided FNA procedure involves the presence of both pathologist and radiologist thereby facilitating clinical correlation and appropriate handling and allocation of aspiration material for routine and ancillary tests [4]. The value of CB is well established as an adjunct to cytological assessment in FNA specimens from many anatomical sites including abdominal lesions.
Ultrasonography was the only guidance technique used in the present and few other studies [5,9–11]. Sidhalingreddy and Andola used USG guided aspiration for 164 out of 245 lesions studied [12]. In the present study, the diagnostic yield was high (96.77%) when compared to the study by Islam et al., who obtained 87.7% diagnostic yield and Sidhalingreddy and Andola who observed higher diagnostic yield with direct aspiration (95%) than with USG guided FNAC (92.7%) [5,12]. Male patients predominated over female patients in our study as well as in few other studies [5,9,11]. The most common organ sampled in the present study was liver which was comparable to that in other studies [4,5,11,12]. Malignant lesions constituted the most common diagnostic category in our study which is comparable to other studies [4,5,9–12] [Table/Fig-17]. Similar to our study, authors [4,11,12] have reported hepatic malignancy as the commonest malignancy. However, a vast majority of hepatic malignancies studied by Stewart et al., were metastatic carcinomas in contrast to predominance of HCC in our study [4]. Stewart et al., prepared CB in a few cases for subsequent ancillary investigations [4]. They quoted that additional cytology preparations lowered the unsatisfactory rate and improved the overall diagnostic sensitivity by 8%. In our study in seven cases the cell blocks were marginally more successful than the routine smears.
Categories of Final Cytologic Diagnosis-Comparative Analysis
| Authors | Stewart et al., [4] | Sidhaling reddy & Andola [12] | Islam et al., [5] | Hemalatha et al., [10] | Mishra et al., [9] | Bolde SA et al., [11] | Present Study |
|---|
| Pelvic mass included | 6.4% | 21% | 0% | 32.8% | 7.5% | 0% | 0% |
| Guidance technique | Image guided | USG/Direct/CT | USG | USG | USG | USG/Direct | USG |
| Nonneoplastic | 7.80% | 10.2% | 37.2% | 20% | 37.5% | 22% | 25.8% |
| Benign | 0% | 22.4% | 15.5% | 0% | 03.2% |
| Malignant | 78.2% | 60.3% | 52.6% | 64.5% | 62.5% | 48% | 67.7% |
| Suspicious | 0% | 0.6% | 0% | 0% | 0% | 18% | 0% |
| Unsatisfactory | 13.5% | 6.5% | 10.3% | 0% | 0% | 12% | 3.2% |
| Total No. cases | 141 | 245 | 78 | 90 | 80 | 50 | 62 |
In an investigative sequence of 72 patients with hepatic lesions [13], the diagnostic accuracy, sensitivity and specificity of USG guided FNAC were 92.82%, 96.8% and 100% respectively when compared to 95.65%, 94.73% and 100% respectively in our study. Wee has provided an algorithmic proposition to the cytological diagnosis of HCC, its variants and their differentiation from imitations [14]. The author further describes the following invaluable biopsy-like architectural features which can be appreciated in CB’s: trabecular-sinusoidal pattern formed by broad trabeculae (> 2 cells thick), pseudoacini, and unpaired arteries and deficient/virtually absent reticulin framework. Mohmmed et al., quote that diagnostic and sub-typing accuracy of liver malignancies significantly improved if the conventional smear and CB were combined together [15]. This was also noted in our study. One of the two false negatives in our study was the case of cytologically diagnosed cirrhosis which was histopathologically confirmed as HCC. This was similar to that noted by Swamy et al., in their study [13]. In cirrhotic livers, the early well-differentiated HCC present as equivocal nodular lesions on imaging techniques; the differentiation of these from benign hepatocellular nodules poses a diagnostic problem in both histological and cytological material [14].
Gangopadhyay et al., diagnosed eight cases of retroperitoneal soft tissue pleomorphic sarcomas; their sub-typing was possible only on histopathology [16]. Mishra et al., also noted the same difficulty in these tumours [9]. They opined that failure to specifically typify the soft tissue tumours is a pitfall in cytology. In our study the characteristic cytological features in the routine smears along with cell block study help to accurately type the tumours. Knowledge of the corresponding histological features is mandatory for differentiating cytologically between various soft tissue sarcomas. Additionally in our case of NFS the typical clinical features of neurofibromatosis in the patient was contributory. The sensitivity, specificity, predictive value and accuracy for the soft tissue tumours were all 100% in our study, in agreement with that noted by Mangal et al., [17]. It is also noted that these tumours usually attain a large size before they become symptomatic and cause difficulty in thoroughly sampling all the different areas of tumour which may have variable morphology [10].
The tumours arising in undescended testis account for 10% of the testicular tumours, the risk of malignancy proportionately increasing with higher position in the path of descent. Males with cryptorchidism are 4-6 times more likely to be diagnosed with testicular cancer and in 80% of the cases true undescended testis is unilateral [18]. In the present study we found the rare occurrence of bilateral undescended testis with bilateral tumours in both the patients. Rakesh et al., reported a unilateral huge retroperitoneal mixed GCT in cryptorchid testis [18]. Pandey et al., has emphasised the value of cell block in diagnosis of testicular tumours [19]. They used the CB to confirm the diagnosis suggested on FNAC and also did tumour markers on CB sections.
For a retroperitoneal lymph node mass with suspected metastatic malignancy as in our case, an aspiration needle can be used. However in the likelihood of the mass being a lymphoma and because of the difficulty in diagnosing and typing of lymphomas with skinny needles, it is often necessary to use cutting needles to obtain a definite diagnosis [1].
USG is usually the preferred guidance technique for renal masses. The easy accessibility of most renal lesions and the generally unequivocal cytomorphology of renal lesions contribute to a higher diagnostic accuracy approaching 100% [1]. The characteristic cytological findings in our cases of Wilm’s tumour and RCC were similar to that noted by other authors [16,17]. Cytological diagnosis of Wilm’s tumour facilitates pre-operative debulking of the tumour with chemotherapy and may prevent operative rupture and spillage [16].
A study quotes the difficulties in the diagnosis of abdominal non-Hodgkin lymphomas due to the close resemblance to any other round cell tumours on cytology [10]. In our cases stringent analysis of cytomorphology in the aspirate smears along with the complete clinical profile of the patient helped us suggest diagnosis of NHL which was confirmed on biopsy. This was in agreement with the observation by Hemalatha et al., [10].
In the absence of any specific clinical, radiological, laboratory findings abdominal TB is a diagnostic problem to the treating surgeons. Abdominal TB being misdiagnosed is often reported in the literature [20]. Similar to that noted by Ghalige et al., in our cases of mesenteric lymph node and intestinal TB there was no pulmonary involvement [20]. The cytological smears showed granulomas, AFB stain positivity was confirmed on CB’s and the patients were treated with anti-tuberculosis therapy. They showed complete recovery on follow up.
Often cytological samples alone cannot provide accurate cell typing in pancreatic carcinoma, multiple needle passes and/or cytohistological material in the form of cell block may provide better tissue yield and improve diagnostic accuracy [1]. In our study, CB morphology aided in the final cytological diagnosis of the poorly differentiated carcinoma.
Mesenteric lipomas are rare, sometimes causing abdominal pain by pressing upon the intestinal loops [21]. They can pose diagnostic difficulties due to absence of abnormal laboratory findings and vague nonspecific symptoms as in the case reported by Cha et al., and in our case of a 13-year-old female with abdominal pain [21]. The FNAC and CB were diagnostic and were confirmed by histopathology of resected specimen.
Potential complications of FNAC of abdominal lesions are described in the literature varying between 0% and 12% depending on the organ sampled in the procedure [1]. But majority of the studies [12] including ours support the safety of the procedure.
In different studies the sensitivity of USG guided FNAC ranged from 86% to 94.1% [4,5,10,12] while specificity was 100% [4,10,12], [Table/Fig-18]. The diagnostic accuracy ranged from 82.5% to 96.5% in literature [4,9–12]. In the present study, we noted a high sensitivity (95.35%) and diagnostic accuracy (96.43%) with specificity of 100%.
Statistical Results –Comparative Analysis
| Studies | Sensitivity | Specificity |
|---|
| Stewart et al., [4] | 86% | 100% |
| Sidhaling reddy & Andola [12] | 94.1% | 100% |
| Hemalatha et al., [10] | 94.1% | 100% |
| Present study | 95.35% | 100% |
Various authors have evaluated the usefulness of CB study along with routine smears. In a study by Liu et al., CB’s provided additional information in 12% of the cases [6]. Wojcik and Selvaggi showed that 84.5% of their cases had identical smear and CB result, and 14.3% had superior smear results in detecting malignancy [8]. In none of their case of negative smear, CB was positive. Nathan et al., in their study showed 15.2% improvement when both smears and CB’s were studied together [7]. They described a modified CB technique using Nathan alcohol formalin fixative. Basnet et al., observed that the CB’s were superior in diagnosing neoplasms than smears with diagnostic accuracy of 95.91% and 91.8% respectively [2]. They quote the added advantage of having multiple sections of the same material from CB for special stains and immunocytochemistry. In the present study out of 42 cases in which CB were available, in 7 cases (15.55%) including 4 cases of malignancy and 3 cases of abdominal TB, the CB’s significantly added to the final cytodiagnosis. In 35 cases the results were identical to smears and reinforced the cytodiagnosis. However in both the cases with negative smear (false negative) CB’s were also negative.
Conclusion
Real time USG is a simple, cost effective imaging modality; has an advantage over CT in guiding non axial needle trajectories and allowing procedures to be accomplished more quickly than CT. USG guided FNAC is a safe and reliable method not only for cytodiagnosis, but also for differentiating neoplastic from non-neoplastic intra-abdominal lesions. However, the negative FNAC result in a patient with suspected malignancy may need further evaluation. The architectural preservation in the CB allows interpretation of aspirated material in a biopsy-like fashion and thus the CB analysis can be employed routinely to contribute to the final results of FNAC.