Herpes simplex virus (HSV) is the most common identifiable cause of serious or life threatening sporadic, endemic encephalitis. Typical HSV encephalitis in patients outside neonatal age is caused by HSV-1. A 23-month-old girl was referred to our hospital with a three-day history of fever, listlessness, slurred speech, and suspicious oesophageal foreign body impaction. Laboratory evaluations showed white blood cell count of 10900 /mm3 with 65% neutrophils. Upper endoscopy revealed diffuse severe ulceration in middle to distal third of oesophagus and no foreign body was found in oesophagus or stomach. Parenteral acyclovir was prescribed for herpes encephalitis in addition to antibiotics for central nervous system infection. Chest X-ray and brain MRI was unremarkable. Lumbar puncture revealed normal protein and glucose with 10 white cell count. She developed a raising liver enzyme tests. Total and direct bilirubin was 1.2 mg/dc and 0.2 mg/dc respectively. Because of neurological symptoms, acyclovir was adopted for our patient from the beginning. The girl did not respond to medication and died after 28 days. Progression of her disease prior to referral appears to contribute to the administered treatment inefficacy. Severe rapid progression of disease prior to referral and potential resistance to acyclovir could cause treatment failure.
Acyclovir, Encephalopathy, Herpes simplex
Case Report
A previously well, 23-month-old girl was referred to our hospital with a three-day history of fever, listlessness, slurred speech, and suspicious oesophageal foreign body impaction. She had difficulty swallowing liquids and solids as well as drooling. There was no significant medical history. Vital signs: heart rate: 120/min, temperature: 39.1°C and other physical findings were normal. In physical examinations, there was no lesion, ulceration, or erythema of the gingiva, buccal mucosa, and tongue. The height was 87 cm (50-75th) and the weight was 11 kg (10-25th).
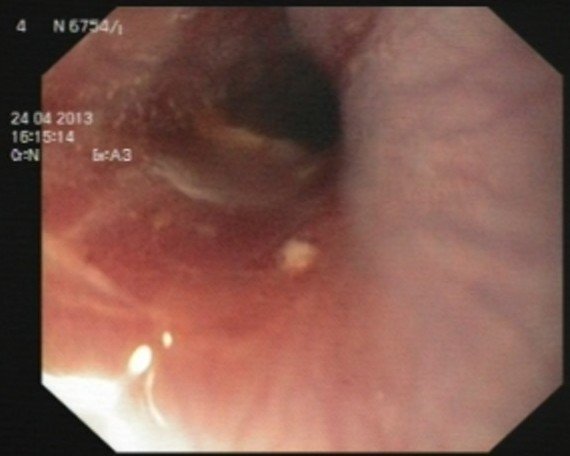
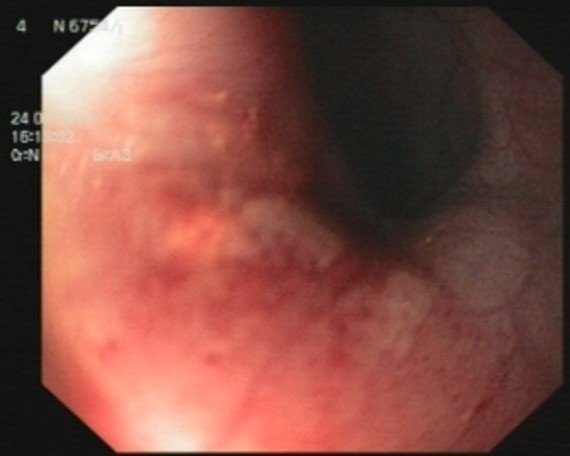
Laboratory evaluations showed white blood cell count of 10900 /mm3 with 65% neutrophils. Liver function was normal. HIV test was negative. Upper endoscopy revealed diffuse severe ulceration in lower to middle third of oesophagus and no foreign body was found in oesophagus or stomach [Table/Fig-1,2]. Since endoscopy was done to find foreign body, no biopsy was taken. Then, as cerebral symptoms progressed, HSV was raised.
On the night of admission, she became lethargic and experienced status epilepticus and her seizures were resistant to drugs. As a result, she was intubated and thiopental started. After observation of neurologic signs, parenteral acyclovir was prescribed for herpes encephalitis in addition to antibiotics for CNS infection.
Other investigations including chest X-ray and brain MRI, which was run prior to intubation, was unremarkable. Lumbar puncture revealed normal protein and glucose with 10 white cell count (PMN=75%). Cerebrospinal fluid (CSF) viral polymerase chain thirdreaction (PCR) confirmed herpes simplex virus type 1 (HSV-1). In follow-up she developed a raising liver enzyme tests (AST=2700, ALT=2300). Total and direct bilirubin was 1.2 mg/dc and 0.2 mg/dc respectively. A repeated lumbar puncture on day 14 showed negative result for HSV.
Discussion
Primary HSV infection is commonly seen in children. The maximum incidence of the disease is at two years of life with gingivostomatitis [1] . Herpes oesophagitis in the immunocompetent host is a self-limited condition and usually a primary infection, but it may be reactivated occasionally [2] ; it has been associated with gastric involvement in a few cases [3] . The trauma to the oesophageal tissue could predispose to herpes oesophagitis in an immunocompetent host [4] . Other important observations have been bloody vomiting and ooesophageal ulceration [5] .
HSV-1 may lead to chronic ulcerative infection in immunosuppressed children. It is also a common reason for fatal sporadic encephalitis [6] . Central nervous system (CNS) infected with HSV has various manifestations. Recently, a great deal of knowledge has been gathered about the pathogenicity, diagnosis, and treatment of HSV [7] . As with other reports, dysphagia and fever were noted in this report. In the young child refusal of oral intake may be due to both odynophagia and oral discomfort [8]. Although mucosal lesions and ulcers in the mouth and tongue were not noted in physical examinations of our patient, severe lesions caused by inflammation of the oesophagus were seen by endoscopy, which is consistent with other reported HSV-infected cases [8].
HSV is also a common cause of ulcerative oesophagitis in the immunocompromised or debilitated host [9]. The presence of HSV-1 was confirmed in our patient’s CSF. In the studies where the patients had symptoms similar to our patient, the isolated HSV was mostly HSV-1 [8,10], which is in agreement with the present report.
Antiviral therapy with acyclovir has decreased mortality. However, morbidity is still high. The clinicians must be highly suspicious because HSV could mimic other CNS diseases and speedy initiation of treatment is essential [7] .
The management of hospitalized patient includes intravenous (IV) acyclovir, IV therapy and supportive care for nutrition and acid suppression [8].
In a study, the most interesting finding in 21 children with confirmed herpes simplex encephalitis was negative HSV CSF PCR test in 29% of the patients and extratemporal lobe involvement in 59% of the patients [10]. In a study on Nigerian children, children of 9-16 weeks age were more susceptible to HSV-1 infection and HSV-1 infection was significantly associated with age. A higher prevalence was obtained in female children (61.5%) than male (54.5%), as well [6] . In a study in Australia from 1999 to 2011, totally 29 mortalities due to HSV occurred in children under 15 years, of whom nine were female and 15 died within the first month of life. 69% of the children died prior to the age of 12-month-old [11].
Al-Hussaini and Fagih reported three cases of oesophagus inflammation due to HSV referred with fever, dysphagia, and odynophagia. One of them was diagnosed as suspicious herpes oesophagitis. Another child, a 3-year-old girl, was administered with IV acyclovir, and the third case was treated as self-restricted. All three were healthy in follow-up [8]. Lack of similar research could be due to rarity of the patients similar to our reported case. According to previous works, early onset of acyclovir, particularly in patients with odynophagia, may assist in recovery [8].
Ali et al., confirmed HSV-1 infection in an 11-month-old infant’s CSF and administered parenteral acyclovir. The infant responded to treatment and was discharged after 16 days without neurological complications [12]. Wang et al., study indicated that the prevalence rate of acyclovir-resistant HSV-1 in children was higher than predicted. Moreover, various mechanisms resulting in the resistance were identified. These results suggest that new anti-herpetics with various working mechanisms should be seriously considered [13].
HSV infections are prevalent. Children are infected with HSV, which may cause asymptomatic acquisition of life-threatening disease. HSV treatment in children can be addressed per severity and time of acquisition [14].
MRI findings are more likely to be abnormal at initial evaluation for HSV encephalitis because of its high sensitivity to changes in brain water content. Findings in MRI include hyperintensity of temporal and brain stem areas on T2 weighted images, and imaging results may be normal if performed in the early stages of disease [15]. In the present patient, MRI was performed in early stage of the disease and normal MRI result, which has also been reported in the setting of the disease, does not rule out herpes meningoencephalitis [16].
Because of neurological symptoms, acyclovir was adopted for our patient from the beginning. Progression of her disease prior to referral appears to contribute to the administered treatment inefficacy. It seems that severe rapid progression of disease prior to referral and potential resistance to acyclovir caused treatment failure.
Middle oesophageal one-third
Lower oesophageal one-third
Conclusion
The most obvious finding from this case is herpes oesophagitis, which must be considered in the patients with drooling and dysphagia.
Acknowledgment
Hereby, we gratefully thank Research and Technology Deputy of Shahrekord University of Medical Sciences.
[1]. S Krugman, SL Kats, Herpes virus infection. In: Krugman S, Katz SL, editors. Infectious diseases of children 1981 St. LouisThe CV Mosby Company:130 [Google Scholar]
[2]. WT Depew, RS Prentice, IT Beck, JM Blakeman, LR DaCosta, Herpes simplex ulcerative oesophagitis in a healthy subjectAm J Gastroenterol 1977 68(4):381-85. [Google Scholar]
[3]. M al-Samman, MJ Zuckerman, A Verghese, D Boman, Gastric ulcers associated with herpes simplex oesophagitis in a nonimmunocompromised patientJ Clin Gastroenterol 1994 18(2):160 [Google Scholar]
[4]. JA DiPalma, CE Bradyn, Herpes simplex oesophagitis in a nonimmunosuppressed host with gastrooesophageal reflux.Gastrointest Endosc 1984 30(1):24-25. [Google Scholar]
[5]. MJ Chusid, HW Oechler, SL Werlin, Herpetic oesophagitis in an immunocompetent boyWis Med J 1992 91(2):71-72. [Google Scholar]
[6]. AM Shaibu, M Aminu, BO Musa, MA Bugaje, Seroprevalence of IgG antibodies to herpes simplex virus type-1 in Nigerian childrenNiger J Med 2014 23(1):40-45. [Google Scholar]
[7]. RW Widener, RJ Whitley, Herpes simplex virusHandb Clin Neurol 2014 123:251-63. [Google Scholar]
[8]. AA Al-Hussaini, MA Fagih, Herpes simplex ulcerative oesophagitis in healthy children Saudi J Gastroenterol 2011 17(5):353-56. [Google Scholar]
[9]. DH Buss, M Scharyj, Herpesvirus infection of the oesophagus and other visceral organs in adults. Incidence and clinical significanceAm J Med 1979 66(3):457-62. [Google Scholar]
[10]. JC Galbraith, SD Shafran, Herpes simplex oesophagitis in the immunocompetent patient: report of four cases and reviewClin Infect Dis 1992 14(4):894-901. [Google Scholar]
[11]. G Khandaker, C Raynes-Greenow, H Smithers-Sheedy, R Booy, R Menzies, C Jones, Mortality from Herpes Simplex Virus (HSV) Infection in Australian Children, 1999-2011 Using National Datasets.Infect Disord Drug Targets 2014 29(6):364-71. [Google Scholar]
[12]. J Ali, H Walsh, S Sanapala, N Syed, Concurrent meningococcal and herpes simplex infection in a non-immunocompromised childBMJ Case Rep 2014 :2014 [Google Scholar]
[13]. Y Wang, Q Wang, Q Zhu, R Zhou, J Liu, T Peng, Identification and characterization of acyclovir-resistant clinical HSV-1 isolates from children J Clin Virol 2011 52(2):107-12. [Google Scholar]
[14]. RJ Whitley, Herpes Simplex Virus in ChildrenCurr Treat Options Neurol 2002 4(3):231-37. [Google Scholar]
[15]. K Gutierrez, B Pinsky, Herpes Simplex Viruses. In: Feigin and Cherry’s Textbook of Pediatric Infectious Diseases, 7th, Cherry JD, Harrison GJ, Kaplan SL, et al. (Eds),TetracyclineTeratology 2014 PhiladelphiaElsevier Saunders:1947 [Google Scholar]
[16]. HG Hatipoglu, B Sakman, E Yuksel, Magnetic resonance and diffusion-weighted imaging findings of herpes simplex encephalitisHerpes 2008 15(1):13-17. [Google Scholar]