Case Report
A 28-year-old man reported to the Department of Periodontics, Mahatma Gandhi Missions Dental College and Hospital, Navi Mumbai, with a chief complaint of swollen gums involving the entire upper and lower jaws. History revealed appearance of gingival enlargement at 10 years of age. With slow progression, the condition became increasingly severe and generalized, causing unaesthetic appearance together with difficulties in speaking and mastication [Table/Fig-1a] besides these, no other complaints were present, such as pain, bleeding or halitosis. The patient reported a history of prior surgical excision seven years ago which was followed by orthodontic intervention during which the gingival overgrowth recurred. Medical history was not contributory as the patient was not on any medication that could be associated with gingival hyperplasia, such as phenytoin, cyclosporine, or calcium channel blockers.
Extra oral and intra oral views of gingival enlargement in 28 year old man (A1 and A2), his sister (B1 and B2), and his niece (C1 and C2)
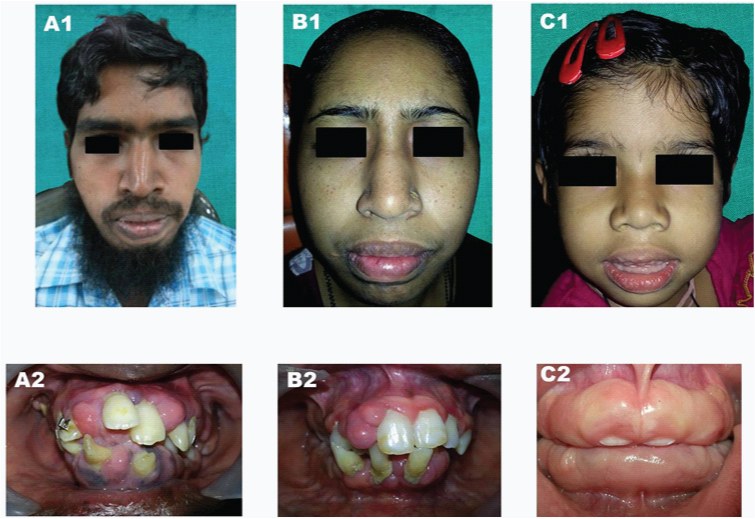
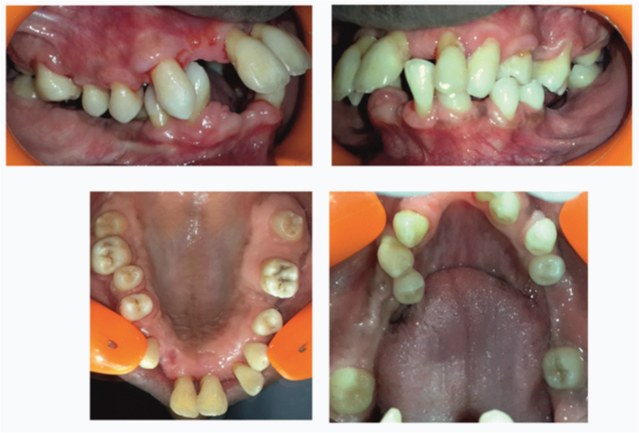
Family history revealed, the sister (33-year-old) [Table/Fig-1b], niece (7-year-old) [Table/Fig-1c], and mother (60-year-old) [Table/Fig-2a-c] all exhibited signs of gingival enlargement. The sister gave a similar history as the patient with appearance of enlargement at 10 years of age, with gradual increase in size and extent of severity. In the niece, gingival enlargement was noticed early at the age of four years, and by the age of seven had gradually increased in size to cover the crowns of all the teeth. The mother reported development of isolated areas of gingival enlargement at the age of 16 which gradually increased in severity involving all the teeth, resulting in aesthetic disfigurement and deranged occlusion. At the age of 24, she underwent full mouth extraction in an attempt to eliminate the enlargement, and has been wearing complete denture prosthesis since then.
Extra oral (a) and intra oral views of residual alveolar ridges with bulbous flabby tissue (b, c) inpatient’s mother
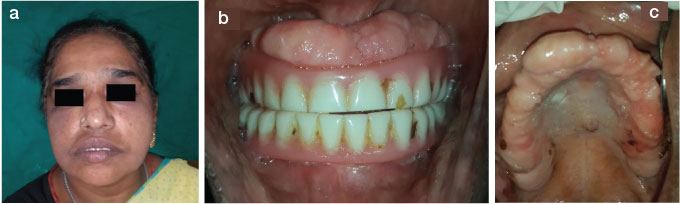
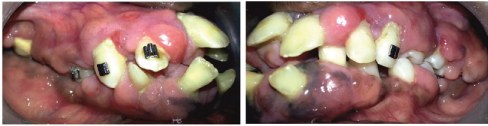
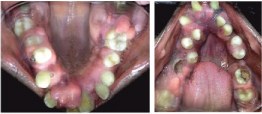
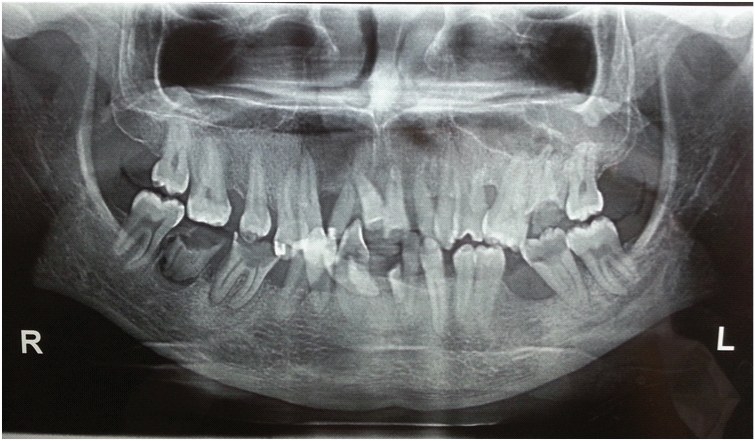
Extra oral examination revealed a dysmorphic face with evident protrusion of the lips. The patient was unable to close his lips completely because of the gingival overgrowth. Other features noticed were macrocephaly, hypertelorism, bushy eyebrows with synophrys (unibrow), hypoplastic nares and cupid`s bow mouth [Table/Fig-1]. Intra-orally, the examination found generalized, severe gingival overgrowth involving both buccal and lingual regions in the maxillary and mandibular arches [Table/Fig-3,4]. The enlarged gingiva was pink, firm, and smooth. No acute inflammatory signs were present. The patient’s dentition suffered from serious malocclusion, involving displacement, rotation, and diastemas. Other features noticed were a highly arched palate, and v-shaped maxillary and mandibular arches. The extra and intra-oral features were remarkably similar in the patient’s mother, sister (underwent cosmetic correction of synophrys), and niece. Intra-oral examination of the mother revealed severely resorbed residual alveolar ridges. The ridges were covered by significant amounts of loose flabby tissue, which resulted in a poorly fitting prosthesis. Panoramic radiographs [Table/Fig-5] of the patient indicated moderate to severe alveolar bone resorption around multiple teeth indicating presence of chronic generalized periodontitis.
Intra oral view of severe gingival enlargement – Buccal view
Intra oral view of gingival enlargement – Palatal and Lingual view
Based on the symptomatology and family history, a diagnosis of Gingival fibromatosis with distinctive facies was arrived at (see discussion below), which was confirmed by a preoperative biopsy.
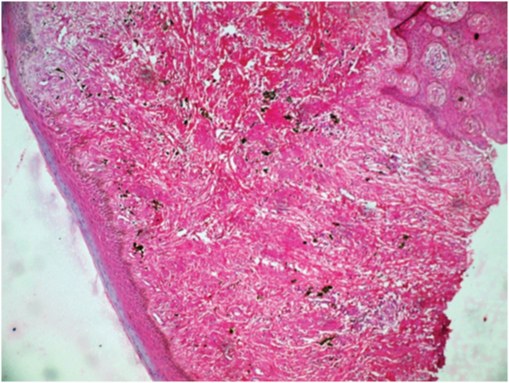
The treatment envisaged of an external bevel gingivectomy at all quadrants, the treatment plan was explained to the patient and informed consent obtained. The surgical intervention was carried out under general anaesthesia, using a combination of electrosurgery and scalpel.The decision to operate under general anaesthesia was taken based on the following considerations; reduction of treatment time from 4 sittings to 1 sitting, secondly in the maxillary tuberosity region, the gingival enlargement was in close proximity to the greater palatine foramen, as such there was an anticipated risk of accidental perforation of the greater palatine vessels, and the management of this would have been easier under general anaesthesia. Lastly, the sheer bulk of the gingiva needing removal would have resulted in significant patient discomfort and difficulty in feeding. To counter this it was decided to monitor the patient on an in-patient basis with ryles tube feeding if required. Taking into consideration the amount of heat generated while using electrosurgery, an 8 to 15 seconds cooling period was advocated between successive incisions or recontouring of the gingiva. Teeth no. 12,27, 37,36, 31,41,42,45,46,47 were extracted during the procedure due to their poor periodontal prognosis. Excised tissue was dispatched for histopathological examination. The postoperative course was uneventful, and the patient returned as scheduled for observation. The healing was good. Two small fragments of bone necrosis were seen in the 2nd and 4th sextant at the 2nd week follow up. They were easily removed, leaving the surface bleeding. Histological examination showed no unusual histologic features, the epithelium was normally structured with elongated rete pegs. Further, the connective tissue was characterized by an abundance of interwoven fascicles of dense fibrous tissue, with few fibroblasts, few blood vessels, and mild inflammatory cell infiltration [Table/Fig-6]. The patient was kept under observation for 10 months [Table/Fig-7] to check for signs of recurrence prior to orthodontic intervention. The patient is currently under follow up, and undergoing orthodontic therapy. Efforts are on to motivate the patient’s sister to seek treatment for herself and her daughter.
10 months post-surgical view
Discussion
One of the rare causes of gingival overgrowth is hereditary gingival fibromatosis (HGF), also known as familial or idiopathic gingival fibromatosis. HGF is a characterized clinically by gradual and progressive enlargement of keratinized gingiva, which is of normal colour, firm in consistency, non-haemorrhagic, and asymptomatic [1]. The first case was described by Gross in 1856 and the prevalence has been estimated at 1:750000 [2]. Oral manifestations of HGF are highly heterogeneous and can vary from isolated areas of gingival enlargement typically seen around the maxillary tuberosities and the labial gingiva around the lower molars to generalized involvement [3]. The degree of enlargement may vary from mild to severe. The onset of gingival enlargement is usually at the time of eruption of the permanent dentition but can develop with the eruption of the deciduous dentition and rarely can be seen at birth [4,5]. HGF occurs only in dentulous areas and has been known to reduce in size or regress completely with the loss of the teeth, suggesting an influence of dental plaque on its pathogenesis [2,6]. The gingival enlargement results in significant aesthetic and functional impairment in affected individuals. The most common effects are diastemas, malpositioning of teeth, prolonged retention of primary dentition, delayed eruption, cross and open bites, prominent lips, and open lip posture [7,8].
The gingiva in HGF displays normal colour, exaggerated stippling, and firm consistency. The tissue is densely fibrotic (bony hard on palpation) and displays a nodular or pebbled surface. Both attached and free gingiva is involved but does not extend beyond mucogingival junction [9,10]. The histologic appearance of HGF is non-specific and is characterized by a well-structured epithelium with elongated and thin papillae inserted in fibrous connective tissue that is rich in collagen fibers [11]. Due to this, the definitive diagnosis of HGF should be based on family history and clinical findings.
HGF can develop as an isolated disorder or more rarely it is associated with other manifestations as part of a syndrome or some chromosomal abnormality [Table/Fig-8]. Accordingly, HGF has been divided into two forms: non-syndromic and syndromic.The syndromic characteristics seen in association with HGF range from hypertrichosis [12], mental retardation [13], epilepsy [14], progressive hearing loss [15], and abnormalities of fingers and toes [16]. Of these, hypertrichosis is the most commonly associated feature. HGF has predominantly an autosomal dominant mode of inheritance; however, autosomal recessive modes of inheritance has also been reported in the syndromic variant [17].
Syndromes associated with HGF (Adapted from Coletta RD and Graner E) [17]
| Syndrome | Mode of Inheritance | Features in addition to gingival overgrowth |
| Gingival fibromatosis with distinctive facies | Autosomal recessive | Macrocephaly, hypertelorism, bushy eyebrows with synophrys, downslanted palpebral fissures, flat nasal bridge, hypoplastic nares, Cupid’s bow mouth, and highly arched palate |
| Gingival fibromatosis with hypertrichosis | Autosomal dominant | Hypertrichosis, mental retardation |
| Rutherfurd | Autosomal dominant | Corneal opacities and retarded tooth eruption |
| Jones | Autosomal dominant | Progressive deafness |
| Prune-belly | Unknown | Absence of abdominal muscles, abnormalities of urinary tract, cryptorchidism, and facial dimorphism |
This paper documents a family spanning 3 generations with individuals manifesting the syndromic form of HGF – Gingival fibromatosis with distinctive facies in which the transmission occurs through an autosomal recessive trait.This syndrome was described by Goldblatt and Singer [7]. Although familial aggregation of HGF has been widely reported in the literature [1,6,7,18], to the best of the authors knowledge, this is the first 3 generational case report of gingival fibromatosis with distinctive facies.
The exact pathogenesis of HGF currently remains unknown, however a review of the literature reveals the following pertinent mechanisms; genetic analysis of HGF has revealed several chromosomal mutations, the most studied of which are 3 different gene loci associated with chromosome 2 and 5 (GINGF on2p21-22, GINGF2 on 5q13-q22 and GINGF3 on 2p22.3-p23.3). Among these loci, the SOS1 (son of seven less one) gene has been recognized as being an essential part of the GINGF locus [19]. The SOS1 protein is a guanine nucleotide exchange factor which plays an essential role in signal transduction during cell growth and differentiation. The SOS1 mutation linked to HGF causes a single base insertion, which as a result of the ensuing frameshift, generates a premature stop codon. This truncated SOS1 protein production leads to enhanced activity of the SOS1–Ras–MAPK pathway which enhances the production of type I collagen, transforming growth factor (TGF)-β, and tissue inhibiting factor of matrix metalloproteinases (TIMPs), whereas it decreases the expression of matrix metalloproteinases (MMPs). The net result of which is increased gingival connective tissue synthesis coupled with inhibition of connective tissue breakdown, leading to excessive connective tissue accumulation [17]. It has also been demonstrated that fibroblasts from HGF patients produce 30% to 50% more collagen than normal gingival fibroblasts [20]. This is thought to be due to the excessive local production of TGF-β, which also brings about myofibroblast differentiation, which has been demonstrated in HGF patients [21]. Myofibroblasts are seen in abundance in fibrotic lesions, and their presence in HGF lesions points to a possible role in the pathogenesis of HGF. However, it should be mentioned, the pathogenic cascade as described above is based on piecemeal evidence, and the true pathogenesis remains to be elucidated.
HGF cannot be definitively treated but may be managed with varying and unpredictable degrees of success. The sheer bulk of excessive gingival tissue necessitates surgical intervention in most cases. The choice of technique used is dictated by the clinician’s acumen and experience as well as the extent and degree of gingival enlargement. Techniques that have been used for the excision of the enlarged gingival tissues, including external or internal bevel gingivectomy in association with gingivoplasty, an apically positioned flap, electrocautery, and CO2 laser [12,15,17,18]. The patient though still has to deal with the risk of recurrence. The risk of recurrence though, should be weighed against the benefit of psychological well-being, improved function, and improved plaque control. Recent reports also attest to the fact that regular follow up and meticulous plaque control can prevent or minimize recurrence for up to two years following treatment [22–24]. Recently, a novel non surgical therapy for HGF has been proposed wherein antibodies that target the C-terminal telopeptide of the α2 chain of collagen I, are used to inhibit extracellular collagen fibril formation [25]. This research is still in the in vitro phase, but may be of potential future benefit.
Conclusion
A family with three generations affected with HGF in which the disease is transmitted by an autosomal recessive mode of inheritance is described in this study.Efforts are underway to elucidate the pathogenic mechanism of HGF. This can further illustrate the correlation between gene mutations, molecular changes, histology, and the clinical situation. Further information has the potential of providing not only novel methods for disease prognosis and differential diagnosis, but also targets for disease prevention and treatment.
[1]. Bozzo L, de Almedia OP, Scully C, Aldred MJ, Hereditary gingival fibromatosis. Report of an extensive four-generation pedigreeOral Surg Oral Med Oral Pathol 1994 78:452-54. [Google Scholar]
[2]. Fletcher J, Gingival abnormalities of genetic origin: a preliminary communication with special reference to hereditary generalized gingival fibromatosisJ Dent Res 1966 45:597-612. [Google Scholar]
[3]. Bittencourt LP, Campos V, Moliterno LF, Ribeiro DP, Sampaio RK, Hereditary gingival fibromatosis: Review of the literature and a case reportQuintessence Int 2000 31:415-18. [Google Scholar]
[4]. Bozzo L, Machado MA, de Almeida OP, Lopes MA, Coletta RD, Hereditary gingival fibromatosis: Report of three casesJ Clin Pediatr Dent 2000 25:41-6. [Google Scholar]
[5]. Singer SL, Goldblatt J, Hallam LA, Winters JC, Hereditary gingival fibromatosis with a recessive mode of inheritance: Case reportsAust Dent J 1993 38:427-32. [Google Scholar]
[6]. Cuestas-Carnero R, Bornancini CA, Hereditary generalized gingival fibromatosis associated with hypertrichosis: Report of five cases in one familyJ Oral Maxillofac Surg 1988 46:415-20. [Google Scholar]
[7]. Goldblatt J, Singer SL, Autosomal recessive gingival fibromatosis with distinctive faciesClin Genet 1992 42:306-08. [Google Scholar]
[8]. Clocheret K, Dekeyser C, Carels C, Willems G, Idiopathic gingival hyperplasia and orthodontic treatment: A case reportJ Orthod 2003 30:13-9. [Google Scholar]
[9]. Baptista IP, Hereditary gingival fibromatosis: a case reportJ Clin Periodontol 2002 29(9):871-74. [Google Scholar]
[10]. Newman MG, Takei HH, Carranza FA, Carranza’s clinical periodontology 2002 9. edPhiladelphiaWB Saunders Company:285 [Google Scholar]
[11]. Martelli-Junior H, Graner E, Bozzo L, Coletta RD, Microscopic and proliferative comparison of gingival fibroblasts from patients with normal gingiva and with hereditary gingival fibromatosisPesqui Odontol Bras 2000 14(2):123-29. [Google Scholar]
[12]. Horning GM, Fisher JG, Barker BF, Killoy WJ, Lowe JW, Gingival fibromatosis with hypertrichosis. A case reportJ Periodontol 1985 56:344-47. [Google Scholar]
[13]. Araiche M, Brode H, A case of fibromatosis gingivaeOral Surg Oral Med Oral Pathol 1959 12:1307-10. [Google Scholar]
[14]. Ramon Y, Berman W, Bubis JJ, Gingival fibromatosis combined with cherubismOral Surg Oral Med Oral Pathol 1967 24:435-48. [Google Scholar]
[15]. Hartsfield JK, Bixler D, Hazen RH, Gingival fibromatosis with sensorineural hearing loss: An autosomal dominant traitAm J Med Genet 1985 22:623-27. [Google Scholar]
[16]. Skrinjaric I, Bacic M, Hereditary gingival fibromatosis: Report on three families and dermatoglyphic analysisJ Periodontal Res 1989 24:303-09. [Google Scholar]
[17]. Coletta RD, Graner E, Hereditary gingival fibromatosis: a systematic reviewJ Periodontol 2006 77(5):753-64. [Google Scholar]
[18]. Prasad SSR, Radharani C, Sinha S, Kumar SVK, Hereditary gingival fibromatosis with distinctive faciesJ Contemp Dent Pract 2012 13(6):892-6. [Google Scholar]
[19]. Hart TC, Zhang Y, Gorry MC, Hart PS, Cooper M, Marazita ML, A mutation in the SOS1 gene causes hereditary gingival fibromatosis type 1Am J Hum Genet 2002 70:943-54. [Google Scholar]
[20]. Martelli-Junior H, Cotrim P, Graner E, Sauk JJ, Coletta RD, Effect of transforming growth factorbeta1, interleukin-6, and interferon-gamma on the expression of type I collagen, heat shock protein 47, matrix metalloproteinase (MMP)-1 and MMP-2 by fibroblasts from normal gingiva and hereditary gingival fibromatosisJ Periodontol 2003 74:296-306. [Google Scholar]
[21]. Sobral LM, Montan PF, Martelli-Junior H, Graner E, Coletta RD, Opposite effects of TGF-beta1 and IFN gamma on transdifferentiation of myofibroblast in human gingival cell culturesJ Clin Periodontol 2007 34:397-406. [Google Scholar]
[22]. Bansal A, Narang S, Sowmya K, Sehgal N, Treatment and two-year follow-up of a patient with hereditary gingival fibromatosisJ Indian Soc Periodontol 2011 15(4):406-9.doi: 10.4103/0972-124X.92581 [Google Scholar]
[23]. Gita B, Chandrasekaran S, Manoharan P, Dembla G, Idiopathic gingival fibromatosis associated with progressive hearing loss: A nonfamilial variant of Jones syndromeContemp Clin Dent 2014 5(2):260-3.doi: 10.4103/0976-237X.132387 [Google Scholar]
[24]. Zandinejad A, Atarodi M, Abdel-Azim T, Leonhart RM, Morton D, Complete oral rehabilitation of a patient with hereditary gingival fibromatosis: A clinical reportJ Prosthet Dent 2014 Sep 29 doi: 10.1016/j.prosdent.2014.07.013 [Google Scholar]
[25]. Gawron K1, Lazarz-Bartyzel K, Lazarz M, Steplewska K, Pyrc K, Potempa J, In vitro testing the potential of a novel chimeric IgG variant for inhibiting collagen fibrils formation in recurrent hereditary gingival fibromatosis: chimeric antibody in a gingival modelJ Physiol Pharmacol 2014 65(4):585-91. [Google Scholar]