Giant Aggressive Mesenteric Fibromatosis- A Case Report
Anandaravi BN1, Jagadish Kumar CD2, Sreejith PS3, Mayur M4, Roopa URS5
1Associate Professor, Department of Surgery,Mysore Medical College and Research Institute, Karnataka, India.
2Postgraduate Trainee, Department of Surgery,Mysore Medical College and Research Institute, Karnataka, India.
3Postgraduate Trainee, Department of Surgery,Mysore Medical College and Research Institute, Karnataka, India.
4Postgraduate Trainee, Department of Surgery,Mysore Medical College and Research Institute, Karnataka, India.
5Postgraduate Trainee, Department of Pathology,JSSMC, India.
NAME, ADDRESS, E-MAIL ID OF THE CORRESPONDING AUTHOR: Dr. Srijith, PG Mens Hostel, Mysore Medical College, Mysore 570001, India.
E-mail: drsreejithps@gmail.com
Fibromatosis are rare, accounting for 0.03% of all tumours. Mesenteric fibromatosis is a very rare (8% of all desmoid neoplasm). Aggressive fibromatosis of mesentery is a rare surgical problem affecting 2-4 per million people. Females are more commonly affected than males (Estrogen acts as a growth factor). It is locally invasive and tends to recur but never metastasize. Here, we are discussing about 24-year-old male presented with progressive abdomen distension associated with pain since one month. Abdominal examination showed a firm non-tender intra-abdominal mass, measuring around 15x14 cm size, with intrinsic mobility, which was perpendicular to mesenteric line, all borders were well-made out. CECT abdomen showed features suggestive of GIST . Elective Laparotomy was done and a giant mass arising from mesentery without any infiltration to the surrounding structures was noted. The entire mass was excised and mesentery repaired. Histopathology showed uniform band of spindle shaped cells arranged in fascicles admixed with blood vessels in a collagenous stroma. Immunohistochemistry showed Beta Catenin +ve, CD 117-ve, CD 34 –ve and SMA-ve, which is confirmative of Fibromatosis. Postoperative period was uneventful.
Beta catenin, Desmoid tumour, Estrogen, Gist
Case Report
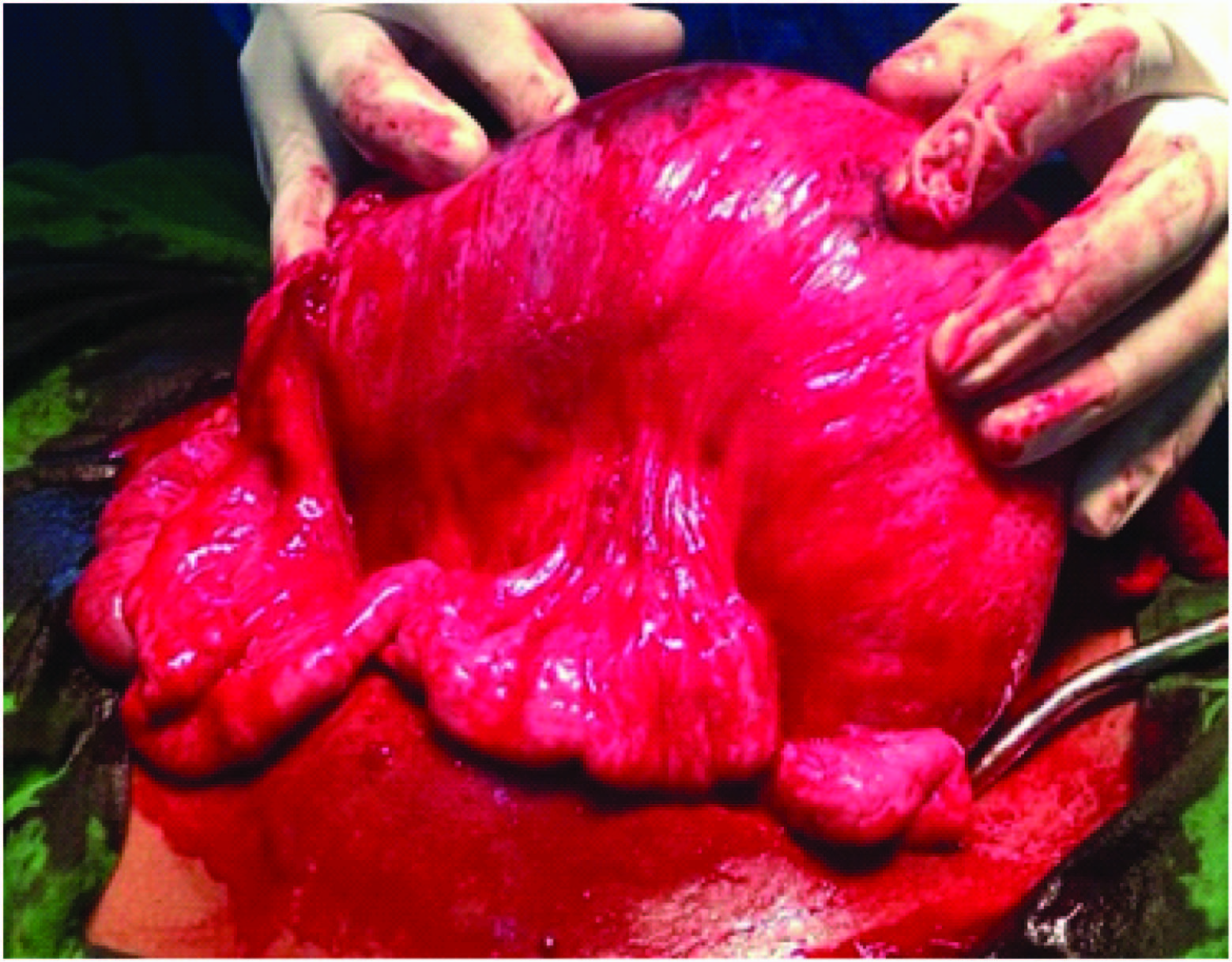
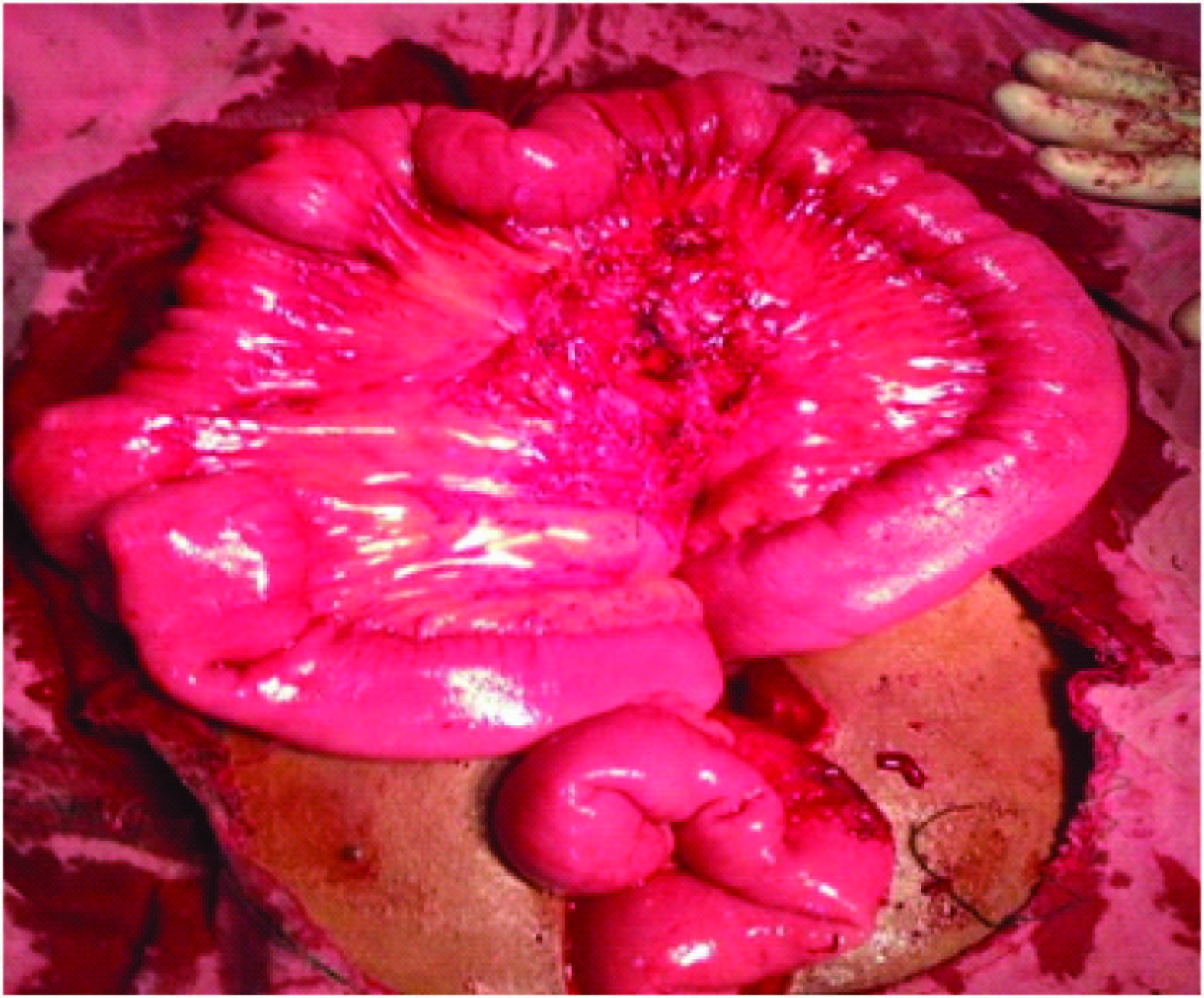
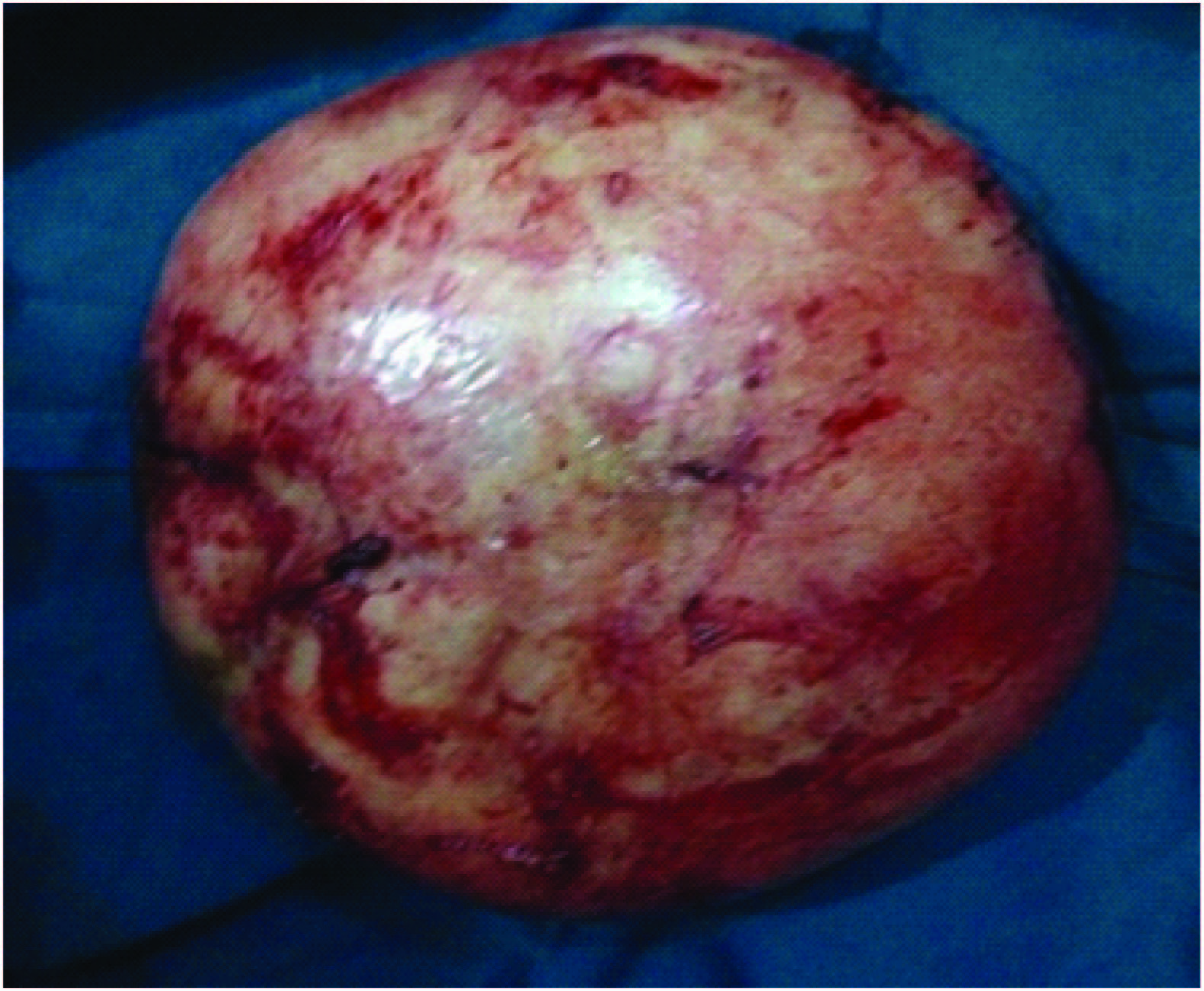
A 24-year-old male presented with progressive abdomen distention associated with pain since one month. No history of previous abdominal surgery, intestinal polyposis. Abdominal examination showed a firm nontender intra-abdominal mass, measuring around 15×14 cm size, with intrinsic mobility, mass mobile perpendicular to the mesenteric line, all borders were well made out. A clinical diagnosis of mass arising from mesentery was made. Contrast enhanced CT scan (CECT) abdomen [Table/Fig-1a,b] showed features suggestive of Gastrointestinal stromal tumour (GIST ). A large well-defined intra-abdominal mass measuring 16.4×15.2×10.6 cm extending from L2-L5 to posterior part of abdominal wall was noted. Post contrast enhancement was noted with central nonenhancing necrotic areas without any calcification. Differential diagnosis is GIST and Mesentery tumour. ESR, complete hemogram, Liver function test and Renal function test are normal. Elective laparotomy was done and a giant mass [Table/Fig-2a,Table/Fig-2b] arising from the mesentery without any infiltration to the surrounding structures was noted. The Entire mass was excised and mesentery repaired [Table/Fig-2c]. Histopathology showed uniform band of spindle shaped cells arranged in fascicles admixed with blood vessels in a collagenous stroma. Cells are infiltrating skeletal muscle without necrosis and mitosis [Table/Fig-3a,b]. All features suggestive of Fibromatosis. Immunohistochemistry showed Beta Catenin +ve, CD 117-ve, CD 34 –ve and SMA-ve, which is confirmative of Fibromatosis (Desmoid tumour) [Table/Fig-4]. Postoperative period was uneventful.
Discussion
Desmoid tumours (Fibromatosis) derived from the Greek word desmos which means ‘band of tendons’. They arise due to fibroblast cell mutations (APC and Beta Catenin). Fibromatosis are rare, accounting for 0.03% of all tumours [1] with incidence rate of 2-4 per million. Females are more commonly affected than males (Estrogen acts as a growth factor). Fibromatosis affect both superficial and deep parts of the body. Superficial fibromatosis involves the face and neck (fibromatosis coli), palms (Duputyren’s contracture), feet (Ledderhose’s disease), penis (Peyronie’s disease), shoulder, thigh, buttock and trunk. Deep fibromatosis are divided into abdominal, extra-abdominal (desmoids outside the abdominal wall) and intraabdominal types. Mesenteric fibromatosis is very rare (8% of all desmoid neoplasm) intra abdominall benign fibrous lesion which usually involves the small bowel mesentery, locally invasive, tends to recur but never metastasize. Its biological behavior is intermediate between benign fibrous lesions and fibrosarcoma. Koh et al., classified mesenteric fibromatosis into five categories based on clinical course: (1) spontaneous regression; (2) stable; (3) variable growth; (4) progressive growth; and (5) aggressive growth [2]. The exact etiopathogenesis of mesenteric fibromatosis is unknown; but various associated factors are (a) Trauma, (b) Estrogen as a growth factor (c) Garden Syndrome (d) Crohns disease. Most are asymptomatic, but few present with abdominal mass, abdominal pain, fatigue, vomiting. Mesenteric desmoid infiltrates adjacent organs and cause important complications like intestinal obstruction, ischemia and perforation, hydronephrosis, ureteric fistula and even aortic rupture [3,4]. Microscopically, mesenteric fibromatosis is characterized by a spatially homogenous proliferation of wavy spindle cells without atypia, associated with collagen among dilated vessels. The mitotic count is relatively low with no evidence of necrosis and nuclear dedifferention [5]. Important differential diagnoses for mesenteric fibromatosis are GIST and Sclerosing Fibromatosis. Immunohistochemistry (IHC) showes diffuse nuclear positivity for Beta-catenin in Mesenteric fibromatosis but not in GIST and Sclerosing mesenteritis [6]. USG and CECT scan are imaging modalities commonly used to diagnose mesenteric fibromatosis.
Treatment of choice is complete excision. Radiotherapy can be used in recurrences and to shrink the inoperable tumour. In cases where surgery and radiotherapy are unsuccessful pharmacotherapy may be used. Pharmacotherapy in desmoid tumour involves 1. Non cytotoxic drugs NSAIDS (Sulindac, Indomethacin) 2. Endocrine therapy (estrogen receptor antagonist tamoxifen, Aromatase inactivators) 3. Cytotoxic chemotherapy (dactinomycin, vincristine and cyclophosphamide) [7]. TeikYin and co-researchers [8] demonstrated the resolution of hydronephrosis due to massive mesenteric fibromatosis by using cyclo-oxygenase 2 inhibitors. Tonelli et al., [9] reported that the daily use of raloxofene decreased mesenteric fibromatosis size without significant side effects. Chemotherapy may be used in : (i) aggressive surgical resections which could result in severe disfigurement especially in young patients (eg: limb-sacrificing surgical procedures, hemimandibulectomies, hemipelvectomies, or chest wall resections). (ii) Large tumors (mesenteric or retroperitoneal) encasing vital structures such as vessels, nerves or ureters. (iii) Tumors which recur after use of other non-cytotoxic treatment such as hormonal agents or NSAIDs. It has 25-50% chances of recurrence despite of above treatments [10].
A Large well-defined lesion noted in abdominal cavity from L2-L5 level just posterior to anterior abdominal wall. Lesion measures 16.4×15.2 ×10.6 cms. Post contrast, Enhancement of the lesion was noted with central non-enhancing necrotic areas. No evidence of calcification's within the lesion. Differential Diagnosis GIST / Mesenteric Tumour

Giant tumour arising from Mesentery of small intestine
Mesentery of small bowel after complete excision of tumour
Gross Specimen of Mesenteric Fibromatosis
(Histopathology) Uniform band of spindle shaped cells arranged in fascicles admixed with blood vessels in a collagenous stroma. Cells are infiltrating skeletal muscle without necrosis and mitosis

Immunohistochemistry positive for Beta Catenin

Conclusion
Mesenteric Fibromatosis is rare problem. It can grow aggressively and attain large size as in this case within short span of time. Many times it will be confused with GIST. Different treatments available for mesenteric fibromatosis but surgery is the best.
[1]. G Guglielmi, A Cifaratti, G Scalzo, N Magarelli, Imaging of superficial and deep fibromatosisRadiol Med 2009 114:1292-307. [Google Scholar]
[2]. PK Koh, C Loi, X Cao, PY Cheah, KS Ho, BS Ooi, Mesenteric desmoid tumours in Singapore familial adenomatous polyposis patients: clinical course and geneticprofile in a predominantly Chinese populationDis Colon Rectum 2007 50:75-82. [Google Scholar]
[3]. S Holubar, AJ Dwivedi, J O'C onnor, Giant mesenteric fibromatosis presenting as small bowel obstructionAm Surg 2006 72:427-29. [Google Scholar]
[4]. D Collins, E Myers, D Kavanagh, G Lennon, E McDermott, Mesenteric desmoid tumour causing ureteric obstructionInternational J Urol 2008 15:261-62. [Google Scholar]
[5]. JA Rodriguez, LA Guarda, J Rosai, Mesenteric fibromatosiswith involvement of the gastrointestinal tract. A GIST simulator: a study of 25 casesAm J Clin Pathol 2004 121:93-98. [Google Scholar]
[6]. E Montgomery, MS Torbenson, M Kaushal, Beta-catenin Immunohistochemistry separates mesenteric fibromatosis from gastrointestinal stromal tumour and Sclerosing mesenteritisAm J Surg Pathol 2002 26(10):1296-301. [Google Scholar]
[7]. Janinis J, Patriki M, The pharmacological treatment of aggressive fibromatosis: a systematic reviewAnnals of Oncology 2003 14:181-90. [Google Scholar]
[8]. TY Ng, MD Yang, YF Chen, CH Chang, Resolution of hydronephrosisdue to massive mesenteric fibromatosis usingcyclo-oxygenase 2 inhibitorsUrology 2007 70:591 [Google Scholar]
[9]. F Tonelli, F Ficari, R Valanzano, ML Brandi, Treatment ofdesmoids and mesenteric fibromatosis in familial adenomatous polyposis with raloxofeneTumour 2003 89:391-39. [Google Scholar]
[10]. CR Baliski, WJ Temple, K Arthur, NS Schachar, Desmoid tumours: A Novel approach for local controlJ Surg Oncology 2002 80:96-99. [Google Scholar]