The World Health Organization estimates that a third of the world’s population is latently infected with Mycobacterium tuberculosis (MTB), and approximately 10% of this one third will develop active disease during lifetime [1]. Identifying the cases of latent TB infection (LTBI) is a part of the global plan of TB control. Active TB is predominantly pulmonary in nature. Clinical criteria are relied upon for diagnosis along with the acid-fast bacilli sputum smear microscopy and mycobacterial culture. Patients with LTBI have no clinical manifestations and are difficult to diagnose by the usual clinical and laboratory tests. In clinical practice, LTBI is defined by the presence of a specific immune response directed towards mycobacterial antigens in the absence of clinical disease [2]. Assays for the diagnosis of LTBI comprise the tuberculin skin test (TST) or methods to detect a specific cellular immune response based on the induction of cytokines after stimulation of lymphocytes with mycobacterial antigens [3]. The interferon-gamma (IFN-γ) is most commonly used as read out and commonly referred to as IFN-γ release assay (IGRA).
A number of antibody detection tests have been developed. Over 40 serologic TB tests that use various antigenic compositions are currently commercially available. All commercial tests which have been evaluated in active pulmonary TB patients present a high variability in terms of sensitivity (0-90%) and specificity (31-100%) [4]. WHO’s policy recommendation issued that all commercial blood tests for diagnosing active TB disease are bad practice. However, blood tests for LTBI are not currently under review by WHO. Data on patients with LTBI and children are limited. Pediatric TB diagnosis is difficult because young children are unable to expectorate sputum samples. There are many challenges in diagnosis of pediatric TB as compared to adults: lower bacterial load and lack of a sputum sample to demonstrate tuberculosis bacilli for confirmation of active TB. In this study, the performance of in-house strip test was compared with the most commonly-used IGRA (Quanti FERON®-TB Gold In-Tube test) and TST. Strip test based on a combination of four recombinant protein antigens was examined to establish whether it was more associated with low TB exposure than reference standard and could be the test of choice for pediatric TB screening.
Materials and Methods
Patients
A total of 91 out-patient subjects were enrolled at the time of their first visit to the Infectious Disease Unit at Phramongkutklao Hospital in Bangkok, Thailand. Of these, 12 were diagnosed as having active TB and 32 were as LTBI. The remaining 14 were household TB contacts without clinical evidence of TB and 33 were other non-TB diseases, such as non-tuberculosis mycobacteria (NTM) lymphadenitis, bronchitis, cancer and pneumonia. The latter two groups were selected as controls. The majority (64%) of patients were children aged under 15 y. The median age was 9 y (range, 1-58 y), with 40 male and 51 female [Table/Fig-1].
Characteristics of study subjects
| Patient group | No. of cases | Gender(m/f) | Median age (yr) |
|---|
| Active TB | 12 | 7/5 | 16 |
| Latent TB | 32 | 11/21 | 12 |
| Healthy TB contacts | 14 | 4/10 | 7.5 |
| Non-TB diseases | 33 | 18/15 | 10 |
Blood sample handling
A blood sample was collected followed by the TST. Blood was collected in specifically designed tubes for IGRA, EDTA-coated tubes for general laboratory test parameters, and a tube without anticoagulant for separation of serum. Sera were frozen to -20°C on the day of collection and were later tested with the test strip at the Queen Saovabha Memorial Institute, Bangkok. Both TST screening and IGRA were carried out at Phramongkutklao Hospital.
Tuberculin skin test (TST)
Tuberculin PPD RT23 (Statens Serum Institute, Copenhagen, Denmark) was injected intradermally on the ventral surface of the forearm. After 48-72 h, the transverse diameter of the skin induration was measured with the ballpoint-pen method using a plastic ruler graduated in millimeters. A positive TST result was defined as an induration of ≥5 mm [5].
IFN-γ release assay (IGRA)
Whole blood specimens were tested with QuantiFERON®-TB Gold In-Tube test (Cellestis, Carnegie, Australia) to measure IFN-γ according to the manufacturer’s protocol. A positive IGRA result was defined as a difference in the IFN-γ levels between the test sample and negative control equal to or greater than 0.35 IU/ml.
Strip test
The test strip was prepared in-house and only used for research purposes. Proteins of MTB were selected either because they were known to elicit antibody responses, e.g., ESAT-6 [6], MPT-64 [7]and Rv3615c [8],or because it was expected to be preferentially expressed in non-replicating tuberculosis bacilli. For example, Rv2031c is encoded by gene found in the so-called dormancy (dosR) regulon [9]. MTB proteins were expressed as recombinant products in Escherichia coli and purified by column chromatography [10]. The strip test utilizes the principle of immunochromatography and is a unique two-site immunoassay on a membrane. A combination of four recombinant protein antigens was conjugated with the colloidal gold as the detector reagent, and Staphylococcal protein A (SPA) was immobilized on the nitrocellulose membrane as the capture reagent. As the test serum sample (50 μl) flows through the membrane assembly of the device, the antigen-colloidal gold conjugate complexes with anti-MTB antibodies in the sample. This complex moves through the membrane to the test region, where it is immobilized by the SPA coating of the membrane, leading to formation of a clear red-purple colored band which confirms a positive test result. The absence of this colored band in the test region indicates a negative result. The unreacted conjugate and unbound complex move further on the membrane and are subsequently immobilized by rabbit anti-MTB antibodies on the membrane at the control region, forming a red-purple band. This control band serves to validate the test results. Interpretation of the test results was completed in15 min after sample application.
Statistical Analysis
For each subject, TST status, IGRA and strip test were recorded as positive or negative. Sensitivity and specificity were assessed for all three diagnostic tests. The detection accuracy among the tests was calculated by using a receiver operating characteristic (ROC) analysis [11].
Ethical consideration
The protocol of this study was approved by the Institutional Review Board of the Royal Thai Army Medical Department. All of the subjects were provided with all relevant information about the study before they gave written consent.
Results
Performance of TST, IGRA, and strip test
A total of 12 patients were diagnosed as having active pulmonary TB based on culture and smear positive data. The median age for study subjects was 16 y (range, 1 to 51 y). All of the patients (100%) were positive both in the TST and IGRA, while 7 (58.3%) had a positive strip test result [Table/Fig-2].
TST, IGRA, and strip test results on TB patients and control subjects
| Patient group | Positive rate |
|---|
| TST | IGRA | Strip test |
|---|
| No. | (%) | No. | (%) | No. | (%) |
|---|
| Active TB | 12/12 | 100 | 12/12 | 100 | 7/12 | 58.3 |
| Latent TB | 28/31 | 90.3 | 12/32 | 37.5 | 12/32 | 37.5 |
| Healthy TB contacts | 0/14 | 0 | 0/14 | 0 | 0/14 | 0 |
| Non-TBdiseases | 15/33 | 45.5 | 2/33 | 6.5 | 4/33 | 12.1 |
In the latent TB patient group, the median age for study subjects was 12 y (range, 1 to 58 y). The latent TB category was defined by a positive response to the TST and/or IGRA, the absence of clinical, bacteriological, or radiographic evidence of current disease. Out of 32 patients, one patient did not return for TST reading. Twenty eight of the 31 patients (90.3%) had a positive TST result whereas of the 32 patients, 12 (37.5%) gave positive results for each of IGRA and strip test.
In the control groups, none of the 14 healthy TB contacts had a positive result in all three diagnostic tests. Of the 33 patients with non-TB diseases, 15 (45.5%) had a positive TST result whereas 2 (6.1%) and 4 (12.1%) gave a positive result in IGRA and strip test, respectively. Based on these results, the overall specificities of the TST, IGRA and strip test were 68.1 (32 of 47), 95.7(45 of 47) and 91.5% (43 of 47), respectively.
Evaluation of results from multiple tests for diagnosis of LTBI
The positive rates were calculated using the number of patients who were positive in any combination of tests divided by the number of patients in each group. Of the 32 patients with LTBI, 2 were strip and IGRA positive, 10 were strip positive but IGRA negative, and vice versa, and the remaining 10 were negative according to both tests. IGRA and strip test in conjunction increased the number of detectable cases by 10 or 31.3%,in total 68.8% (22 of 32) of LTBI cases. By using combined results from the three diagnostic tests (IGRA and strip test plus TST), the overall sensitivity increased to 93.5% (29 of 31).
Detection accuracy among test results
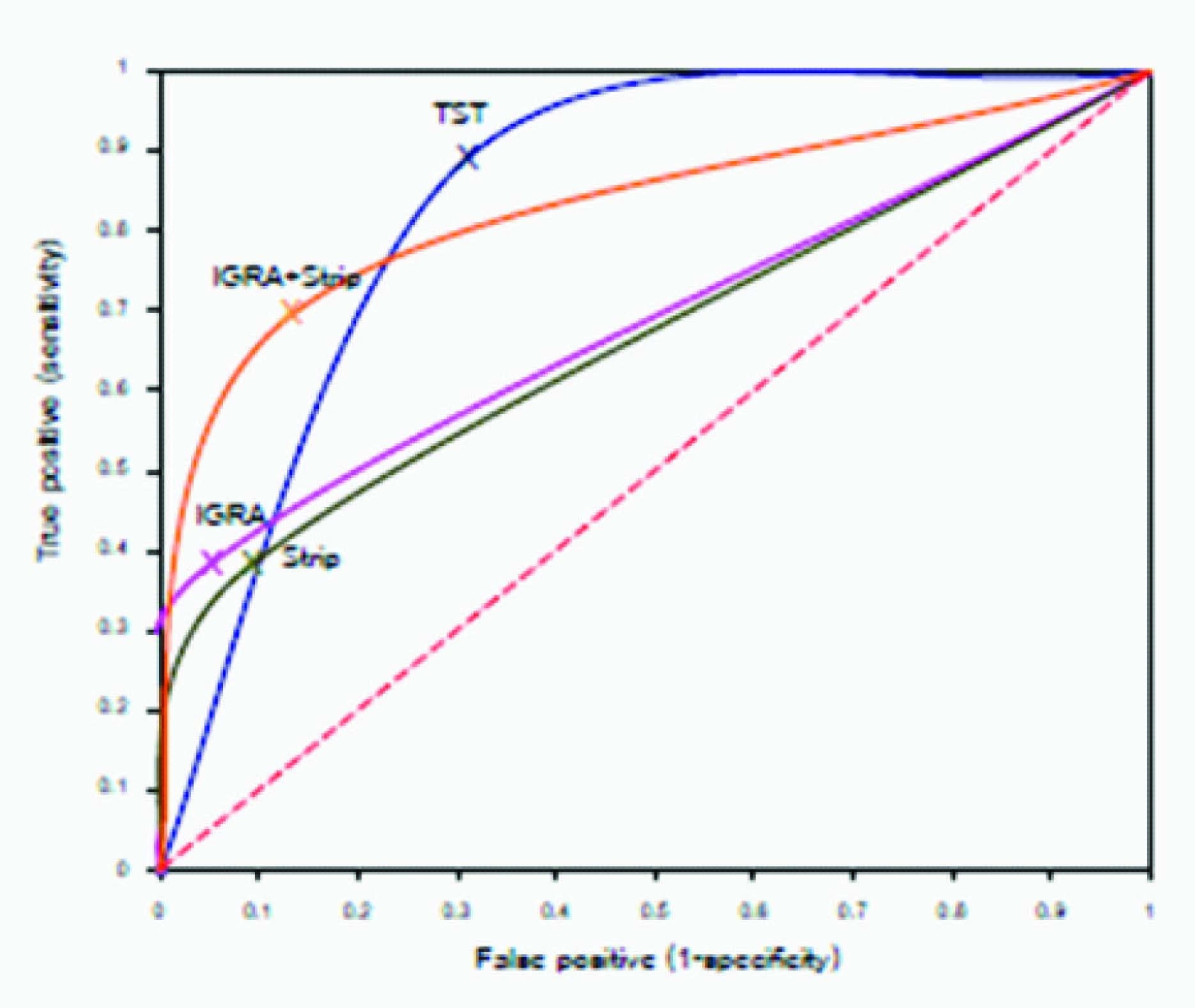
Comparisons of detection accuracy among the three diagnostic tests were conducted by using ROC curves. LTBI was arbitrarily taken as the reference state for this analysis [Table/Fig-3].The curves represent the accuracy in terms of sensitivity and specificity of the tests. Both IGRA and strip test showed low sensitivity and high specificity, while TST showed high sensitivity and low specificity. The greater area under the curve in the two-test combination (IGRA plus strip test), corresponding to overall diagnostic accuracy, illustrates that the combined use of IGRA and strip test resulted in increased diagnostic sensitivity and specificity for LTBI as compared with the single test. Comparison of the strip test plus IGRA with the TST showed the same overall accuracy (0.82 versus 0.83, respectively), however, the curve for the combined blood tests was shifted to the left indicating a higher specificity as compared with the TST.
ROC curve analysis for three diagnostic tests in subjects with LTBI
Notes: A=The area under the curve
Discussion
Thailand is a country with high TB burden and routine BCG vaccination at birth. The TST has been used as an initial screening test to identify TB disease for a century. It is an in vivo assay that elicits a delayed type hypersensitivity reaction towards tuberculin. Tuberculin is a mixture of more than 200 nonspecific antigens shared with NTM and with the strains developed from M. bovis used for BCG vaccination [12,13]. A major drawback of the TST is its low specificity, as a tuberculin-specific skin reaction may not only originate from a previous encounter with MTB but also from prior BCG vaccination or infection with NTM. Although BCG-vaccinated individuals are more likely to have positive TST results [14,15], some authors found that BCG vaccination at birth has limited impact on TST results. TST positivity associated to BCG in the neonatal age decreases rapidly in a few years [16]. Beginning in the 1990s, IGRAs have been developed that overcome the relatively low specificity associated with TST. An increase in specificity is conferred by the use of ESAT-6 and CFP-10 antigens encoded in the region of difference 1 (RD1) of the MTB genome [3]. One of the recent advances and emerging technologies has been the identification of a panel of MTB peptide biomarkers in human serum exosomes for proteomic diagnosis of TB [17].This bioassay is more technically complicated to do than conventional diagnostic tests and has yet to be fully established. During the last three decades, an antibody-based serological test for TB has long been sought. Testing for active TB disease through antibodies found in the blood is difficult. Patients can have different antibody responses [18]. The diverse antibody response to MTB may be governed by HLA types [19]. In addition, active TB and LTBI were associated with serological reactivity to different antigen sets. The targets of the immune response during LTBI differed from those occurring during active disease [20,21]. The present study involved the development of a strip test using a new set of antigen compositions, including ESAT-6, MPT-64, Rv2031c and Rv3615c, that differed from those used in commercial kits. The selected antigens are found only in MTB and few other mycobacterial species. ESAT-6 is secreted in the early phase of infection and antibody response to ESAT-6 was found in 96% of TB patients [22]. MPT-64 and Rv3615c are secreted during active infection [8,23]. Antibodies to the proteins encoded from the dosR regulon and ESAT-6 were associated with inactive TB [20]. A group of about 50 genes belonging to the dosR regulon are known as latency antigens [24]. Rv2031c antigen is encoded by gene that is a member of the dosR regulon [9].
In our study, TST screening and IGRA could diagnose the active TB cases correctly (100%). However, neither TST nor IGRA should be used for the diagnosis of active TB disease, as recommended by WHO [25], Because the quality of evidence for use of IGRA and the TST in diagnosis of active TB is low. In addition, IFN-γ alone may not be sufficient as a prognostic biomarker [26]. The sensitivity of strip test in patients with LTBI was 37.5%. Interestingly, the vast majority (89%) of TB patients with a positive strip test result were children. The combined use of IGRA and strip test would maximize sensitivity and specificity in the diagnosis of LTBI, which was superior to TST in terms of the detection accuracy [Table/Fig-3].
TST will continue to be a simple, low-cost tool in developing countries and may be a useful initial screening test. The lower specificity of TST is associated with a high number-needed-to treat value. Although treatment of TB in Thailand is free of charge, non-medical direct and indirect costs may be a financial burden for patients. Factors associated with TST false-positive reactions may include age, history of previous BCG vaccination and infection with other mycobacteria [27]. There is a possibility of generating false TST positivity at a cut-off of >5 mm as the result of BCG vaccine administered at infancy, but it appears unlikely to affect the diagnosis of LTBI at larger indurations (≥10 mm) [28]. An improved skin test that uses specific TB antigens rather than PPD will be more feasible and avoid the consequences of unnecessary treatment giving high false-positives of TST [29,30]. IGRA is more costly and technically complex to do than the TST. The cost for IGRA (QuantiFERON®-TB Gold In-Tube test) was US$ 94, representing an approximately 1,075% increase over skin test (US$8). At this time, the role for IGRA in low-income, high burden settings is rather limited. Simplification of the test format and reduction of costs might enhance applicability in such settings. Strip test is more convenient and less expensive as compared to the IGRA and more specific than TST. It can help overcome barriers to accessing LTBI diagnosis, as it is cost saving (US$ 9-10 per test strip) for patients. The authors believe that strip test may be a valuable test to rule-in clinical suspicion of LTBI under various conditions. IGRA and strip test evaluate different responses. Blood specimens for IGRA need to be delivered to the laboratory within a relatively short time after being obtained because IGRA requires viable T lymphocytes and delays in incubation IGRA blood samples cause false-negative results [31], whereas strip test uses a few volumes of serum specimens that can be frozen for a long period before testing. However, the antigens used in both IGRA and strip test do not represent the whole spectrum of MTB antigenicity. They represented a small portion of numerous antigens, and more reliable results could be achieved by adding new antigens to the methods. Incorporation of multiple antigens in a diagnostic test to cover the broad spectrum of antigen recognition by different individuals may increase the sensitivity of tests. A drawback of the present study is the relatively small number of patients and the current analysis was limited to only one latency antigen. Some MTB specific proteins may be promising biomarkers for differentiating LTBI from TB and may have diagnostic utility [32,33]. The role of adding more latency-associated antigens to try to improve diagnostic sensitivity needs to be assessed in proper sample sizes. Validation studies should be conducted to evaluate the eventual benefit of strip test for the detection of antibodies against potential latency candidate antigens in latent TB patients. Antibody-based method for LTBI is still experimental and should only be used for research purposes until validation and authorization for use has been obtained from the WHO. If its superior diagnostic capability is found to hold up in routine clinical practice, it could confer several advantages on TB control.
Conclusion
Although WHO warns against the use of strip test for active TB, but the assay may be particularly significant in situations where TB is clinically difficult to diagnose like LTBI. Additional research will be needed to determine whether strip test can be further improved its sensitivity to bring better, more rapid and less expensive test that is suitable for use in children in countries with high TB infection rate. Currently we are working on a set of latency antigen candidates to ascertain their antibody profiles in latent TB state.