Acute coronary syndrome (ACS) is becoming the leading cause of morbidity and mortility in developing countries like India too. The spectrum of presentation is wide from unstable angina to acute myocardial infarction [1]. Previous data suggests that only one fifth of chest pain cases require emergency care and hospitalization but in absence of segregation of these cases at the beginning, physicians over-admit such patients [2]. It burns out the precious resources of a resource poor public setups. Further it may reduce the quality of care for those who actually require it. Consensus guidelines on a universal definition of myocardial infarction have been issued recently by the International Federation of Clinical Biochemistry [3], European Society of Cardiology, the American College of Cardiology, and the American Heart Association and the World Heart Federation [4] that recommend cardiac troponin I (cTnT) and cardiac troponin T (cTnT) measurements as the preferred biochemical cardiac biomarkers for diagnosing ACS. However, the diagnostic efficiency of cardiac troponins within 2 to 4 hours of the symptom onset is limited [5]. Therefore, other laboratory biochemical tests that successfully reduce emergency department delays in provision of immediate care for cardiac pain patients are required in conjunction with established markers [6]. Platelets play a crucial role in the pathogenesis of atherosclerosis and thrombus formation after coronary plaque rupture [1]. Platelet activation leads to the formation of free arachidonic acid, which can be transformed into prostaglandins, such as thromboxane A2, one of the most potent vasoconstricting and platelet-aggregating substances, or into leukotrienes, which can amplify the acute inflammatory response. Consequently, larger and hyperactive platelets play a pivotal role in accelerating the formation and propagation of intracoronary thrombus, leading to the occurrence of acute thrombotic events [7]. These observations have led to the hypothesis that increased mean platelet volume ( MPV) may be a potentially useful predictor in cardiovascular risk stratification [8]. As MPV is an index of platelet size that correlates with platelet activation. In patients with non–ST elevation ACS, it has been shown that higher MPV indicates not only an increased risk of non-STEMI, but also ischemic complications [9]. Other investigators have demonstrated in small observational studies that MPV is higher in patients with myocardial infarction than in those with stable angina pectoris and in healthy controls, suggesting that MPV is a risk factor for the severity of coronary artery disease [10].
Materials and Methods
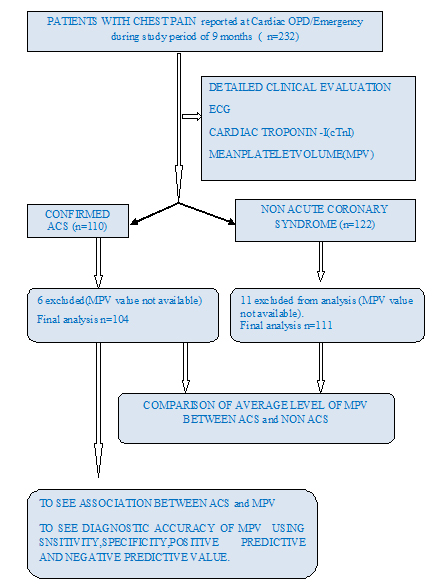
This hospital based observational, descriptive, comparative study was done during January 2013 to September 2013. There were 232 eligible patients (< 6 hours of chest pain) out of all 1160 chest pain attended the Cardiology OPD/ Emergency during the study period. The study protocol was approved by the Clinical Trial Screening and the Ethics Committee of the hospital, and written informed consent was obtained from the patients. All subjects were subjected to detailed history, physical examination and relevant investigations. Venous sample collected (within 6 hrs of chest pain) for routine hematological testing in 3 ml BD vacutainer containing K2 EDTA 5.4 mg and MPV measured within 1-2 hrs of sample collection using SYSMEX 4000 I analyzer. Sample for Trop I collected at 6 hr and at 12 hr if required and level measured using Triage pro(Biosite) analyzer (cTrop- I level>0.01ng/ml). All patients underwent 12 lead ECG and interpreted according to conventional criteria. Total 232 subjects were recruited, out of which 110 were confirmed ACS and 122 were non ACS. 6 patients from ACS group and 11 patients from non ACS group were excluded because the value of MPV was not available. Finally 104 patients in ACS group and 111 patients in non ACS group were analyzed. ACS was diagnosed based on the presence of either of the criteria: 1) detection of rise in cardiac biomarker Trop I >0.01 for acute myocardial infarction. 2) For unstable angina where Trop I is < 0.01 , with at least one of the following four a) typical symptoms of ischemia b) ECG changes indicative of new ischemia c) development of pathological Q wave in the ECG d) Echocardiographic evidence of new regional wall motion abnormality. Non ACS group subjects were diagnosed based on atypical symptoms, no ECG evidence of ischemia, negative cTropI, no Echo evidence of RWMA, negative TMT if required and normal coronary angiogram in some patients [11].
Exclusion Criteria
Patients with chest pain of > 6 hours, Critically ill patients (ACS associated with renal failure, hepatic failure, myeloproliferative disorder or malignancy), patients having any platelet disorder as thrombocytopenia or thrombocytosis, patients with any bleeding or clotting disorder and patients on antiplatelet therapy.
Statistical Analysis
The data were entered on excel sheet and analyzed using Microsoft excel. Quantitative data were summarized in the form of mean and standard deviation. The difference in average MPV value among ACS and non ACS cases were evaluated using student’s t test. There was a significant difference in MPV levels in both groups. The logistic regression was done to find out the independent predictors for ACS using multiple variables. A test of the full model was statistically significant, indicating that the predictors as a set reliably distinguished between ACS and non cardiac chest pain (Chi square = 150.654 p < .0001 with df = 10). Also Hosmer and Lemeshow Test show a good fit by high p value (0.949) and low Chi-square value (2.746). The level of significance was kept 95% for all statistical purpose. The diagnostic efficacy of MPV was evaluated and sensitivity, specificity, positive predictive value and negative predictive value were found out.
Results
Two hundred fifteen subjects comprising of 104 ACS patients and 111 non cardiac chest pain (non ACS) group were included in the present study. Demographic characteristics of patients shown in [Table/Fig-1]. As shown in [Table/Fig-2], Mean platelet volume( MPV) was found to be higher among ACS patients as compared to non ACS patients which is statistically significant (11.44±1.23 vs 9.91±1.27 fl , p-value<0.001).
Clinical profile of study population
| Profile of cases | ACS [n=104] | NON ACS [n=111] |
|---|
| Age mean±SD | 55.09 ± 11.13 Yrs | 43.04 ± 9.44 Yrs |
| Gender | M-82 F- 22 | M-88, F-23 |
| Diabetes Mellitus | 25[24.03%] | 7[6.30%] |
| Hypertension | 39[37.50%] | 24[21.61%] |
| Smoking | 81[77.88%] | 47[42.34%] |
| Dyslipidmia | 40[38.46%] | 16[14.41%] |
| Obesity | 10[9.61%] | 9[8.10%] |
| Family History | 13[12.50%] | 14[12.61%] |
| ACS= Acute coronary syndrome |
MPV in Patients with ACS vs NON ACS
| ACS | NON ACS | p-value (student’s t test) |
|---|
| Mean MPV±SD | 11.44±1.23 | 9.914±1.275 | <0.001 |
EXP(B) value in logistic regression indicates that when MPV raised by one unit the odd’s ratio is 1.22 times as large and therefore patient has 1.2 times more likely to have cardiac chest pain. The diagnostic efficacy of MPV was evaluated and sensitivity(89.42%), specificity (46.84%), positive predictive value (61.18%) and negative predictive value(82.53%) were found out [Table/Fig-3,4].
| MPV | ACS [n=104] | NON ACS [n=111] |
|---|
| Positive (>9.8) | 93 | 59 |
| Negative(<9.8) | 11 | 52 |
MPV=Mean platelet volume in fl.
Sensitivity: TP/TP+FN = 89.42%
Specificity: TN/TN+FP = 46.84%
Positive Predective Value: TP/TP+FP = 61.18%
Negative Predictive Value: TN/TN+FN = 82.53%
Logistic regression analysis
| Variables | B | S.E. | Wald | df | Sig. | Exp(B) | 95.0% C.I.for EXP(B) |
|---|
| Lower | Upper |
|---|
| Step 1a | Age | .027 | .021 | 1.617 | 1 | .203 | 1.027 | .986 | 1.070 |
| SEX(1) | .953 | .674 | 1.999 | 1 | .157 | 2.594 | .692 | 9.723 |
| HTN(1) | -.934 | .538 | 3.012 | 1 | .083 | .393 | .137 | 1.128 |
| SMO(1) | .487 | .623 | .611 | 1 | .434 | 1.627 | .480 | 5.515 |
| DM(1) | -.292 | .608 | .230 | 1 | .631 | .747 | .227 | 2.458 |
| DYSLIP(1) | .024 | .498 | .002 | 1 | .961 | 1.025 | .386 | 2.719 |
| OBE(1) | .819 | .751 | 1.192 | 1 | .275 | 2.269 | .521 | 9.880 |
| FH(1) | -.713 | .719 | .983 | 1 | .321 | .490 | .120 | 2.006 |
| ECG(1) | -5.467 | 1.078 | 25.719 | 1 | .000 | .004 | .001 | .035 |
| MPV | .201 | .090 | 4.964 | 1 | .026 | 1.222 | 1.024 | 1.458 |
| Constant | -2.993 | 1.973 | 2.301 | 1 | .129 | .050 | | |
a. Variable(s) entered on step 1: age, sex, HTN=Hypertension, SMO= Smoking, DM=Diabetes mellitus, DYSLIP= Dyslipidemia, OBE=Obesity, FH=Family history, ECG=Electrocardiography, MPV=Mean platelet volume, SE=standard error.
Discussion
Coronary artery disease is the most important cause of mortality and morbidity in industrialized as well as in developing countries. Both endogenous and exogenous risk factors such as smoking, hypercholesterolemia, DM, and hypertension increase the risk of ACS [12]. Nevertheless, these risk factors accounts for only a part of ACS cases [13].
Therefore, it is needed to identify other related risk factors so as to predict individual risk in the development of ACS. By leading to the formation and dissemination of intracoronary thrombus, larger and hyperactive platelets may accelerate the emergence of clinical picture called as acute coronary syndrome. Platelet function is strongly associated with atherogenesis and atherothrombosis — processes important in pathogenesis of cardiovascular diseases [14]. MPV is a parameter which states platelet size and indirectly proves its activity. It is known that larger platelets are more reactive due to higher concentration of active substances in microgranules (e.g. thromboxane A2 and B2, platelet factor 4, P-selectin, platelet-derived growth factor) and expression of adhesive receptors (glycoprotein IIb/IIIa) [15]. Furthermore, increased MPV values are associated with shortened bleeding time [16]. MPV is considered a useful prognostic marker of cardiovascular risk [15,17,18]. In general population, higher MPV value is associated with increased occurrence of myocardial infarction (MI) [16]. Klovaite et al. found that in general Danish population the risk of MI has increased by 38% in individuals with MPV 7.4 vs. < 7.4 fl independently of known cardiovascular risk factors [19]. Increased MPV has been discussed recently as a predictor of death in patients with ACS, but the cut-off point of MPV in relation to poor prognosis has not been estimated so far [16, 18].
In our study, we examined the platelet volume of our cases, which we think to be likely a risk factor for ACS [Table/Fig-5]. While a number of studies established an association between MPV and coronary artery disease or MI formation; some other studies suggested that there was no such association [19, 20].
Endler et al., in their study where they compared AMI patients to those with stable AP, found MPV to be increased [21]. They also suggested that increased MPV was an indicator for larger and more active platelets and an independent risk factor for MI in coronary artery disease. Likewise, Park et al., also considered increased MPV to be a risk factor for platelet activation [22]. Martin et al, in their study, suggested that increased MPV might be an independent risk factor for post-MI recurrence of coronary events and mortality [8]. In the study where they compared unstable angina pectoris and MI patients, Mathur et al. found platelet counts to be significantly lower and MPV to be higher in patients with unstable angina pectoris [23].
In the study consisting of 518 chronic hemodialysis patients with concurrent coronary artery disease, Hening et al., concluded that high MPV might be associated with coronary heart disease in hemodialysed patients [24]. In that study, they also detected that MPV was higher in unstable angina pectoris subjects who required emergent angioplasty, compared to the rest of unstable angina pectoris patients. These findings rendered them to claim that platelets of larger volume contributed to the formation of thrombus in coronary arteries.
There was no significant difference found between unstable angina pectoris and control arms in terms of MPV, in the study consisted of 54 patients with unstable angina pectoris performed by Butkiewicz and his colleagues [20]. We detected that MPV was significantly higher in ACS group vs. non ACS group, and the PPV , NPV were 61.18% & 82.53% respectively. Based on these findings, we have concluded that larger platelet volumes may constitute a high risk for acute coronary syndrome.
For this purpose, we think that MPV measurement, which is a non-invasive and easy-to-perform method, may be an important tool for the discriminating ACS patients from noncardiac chest pain. Nonetheless, conflicting results of other studies make this issue controversial, which warrants performing of more comprehensive studies in future.
Conclusion
MPV is a simple and economic laboratory measurement (costless when a complete blood count is requested), and owing to its negative predictive value of 82.53%, we suggest that it might be useful as an assisting rule-out test in conjunction with other conventional biochemical cardiac markers in the early prediction of the risk of ACS in patients admitted to the emergency department. It should be substantiated with further studies.
Study Limitation
Patients with non-cardiac chest pain (atypical symptoms) might have unstable angina in the absence of electrocardiography changes and cardiac troponin positivity, have resulted in selection bias. The absence of coronary angiographic findings might be a limitation in this study.
MPV=Mean platelet volume in fl.Sensitivity: TP/TP+FN = 89.42%Specificity: TN/TN+FP = 46.84%Positive Predective Value: TP/TP+FP = 61.18%Negative Predictive Value: TN/TN+FN = 82.53%
a. Variable(s) entered on step 1: age, sex, HTN=Hypertension, SMO= Smoking, DM=Diabetes mellitus, DYSLIP= Dyslipidemia, OBE=Obesity, FH=Family history, ECG=Electrocardiography, MPV=Mean platelet volume, SE=standard error.