Phrynoderma, meaning toad skin, is a type of follicular keratosis coined and described by Nicholls in 1933 [1]. Since, then many investigators over the past several decades proposed deficiencies of various nutrients like Vitamin A (Vitamin A), Vitamin B complex (Vitamin B), essential fatty acids (EFA) and Vitamin E as possible aetiological factors. Majority of these studies were conducted in early and middle part of last century and these observations were mainly based on therapeutic response in patients with phrynoderma. Therefore, a cohort of patients with phrynoderma was selected to study its clinical features, association with nutritional deficiencies, histopathological features and treatment. In the initial study [2], we found that the clinical and histopathological features were consistent with existing knowledge. However, the proportion of phrynoderma cases and its association with nutritional deficiency states was found to be less in comparison to studies which were conducted in last century. This decline has been attributed to improvement in nutritional status [2]. In the past, Vitamin A, Vitamin B, EFA and Vitamin E have been tried in different dosages and routes with variable therapeutic outcomes. Hence, in continuation of our previous study on phrynoderma, the present study was undertaken, as there are very few studies comparing the efficacy of these nutrients in the treatment of patients with phrynoderma.
Material and Methods
An open label randomized controlled study was conducted on cohort of patients with phrynoderma who were selected over a period of two years from patients attending out-patient department of Dermatology in a tertiary care hospital. After obtaining institutional ethics committee approval and informed consent from the parents of children, the patients were included in the study irrespective of age and gender. The patients presenting with discrete, skin to brown colored, keratotic and acuminate papules with central keratinous plug distributed over elbows, knees, extensor extremities and/or buttocks constituted study subjects. Those with underlying systemic or debilitating disease, pregnant women and who did not give consent to the study were excluded. The details of age and gender were recorded and then the patients were randomized into five therapeutic regimen groups. Simple randomization method was used in which the patients were divided into group of five and each patient in the group received one of the five regimens respectively starting from regimen 1 (e.g. first patient in every group received regimen 1, second patient received regimen 2…and fifth received regimen 5). The first four regimens consisted of nutrients along with topical keratolytic, and in the fifth regimen only topical keratolytic (salicylic acid 3% ointment) was used [Table/Fig-1]. Hence, the regimen 5 was considered as control. During the therapy no dietary modification was advised. Therefore, the therapeutic response obtained in the study can be attributed to drugs used. In the first two regimens, intra-muscular route was used as it showed better response compared to oral administration in previous studies. The primary outcome measure was therapeutic response at the end of regimen. The patients were followed-up once in 15 days to note the response and any side effects. The response was graded based on percentage of flattening and decrease in number of lesions. Less than 25% improvement was graded as poor, 26-50% improvement as moderate, 51-75% improvement as good and more than 75% improvement as excellent response. The response was documented through serial photographs at each visit. Both patients and the investigator were not blinded to the interventions as it is an open label study. In the statistical analysis, the frequency variables were presented as numbers and percentages and comparison was done by using Chi-square and Fisher’s exact test. For comparison, good and excellent responses were combined as they indicate the effectiveness of the therapy. The p value of less than 0.05 was considered as statistically significant.
Therapeutic regimens used in the study
| Regimen | Drug* | Dose | Route | Dosage schedule | Duration |
|---|
| 1 | Vitamin A | One lakh Units | IM | Alternate day | Ten injection |
| 2 | Vitamin B complex† | Two ml | IM | Alternate day | Ten injection |
| 3 | Safflower oil | Two tbs | Oral | With each meal | Four weeks |
| 4 | Vitamin E | 400mg | Oral | Once daily | Four weeks |
| 5 | Keratolytics alone | — | Topical | Twice daily | Four weeks |
Note: IM, intramuscular; tbs, table spoon. * The patients in all five regimens received topical keratolytics † one ml contains thiamin 90.9mg, riboflavin 4.5mg, pyridoxine 4.5mg, niacinamide 90.9mg and vitamin B12 90.9μg
Results
A total of 125 patients were included in the study with 25 patients each in five regimen groups. There were 79 (63.2%) males and 46 (36.8%) females with a ratio of 1.72:1. The age of the patients ranged from 3 to 26 years with mean of 10.1±4.3 years. In around 106 (84.8%) patients, the lesions were localized to elbows, knees, extensor extremities and/or buttocks and in remaining 19 (15.2%) the lesions were also present on trunk. The gender and age distribution of the patients in various regimens were comparable [Table/Fig-2]. Out of 125 patients, 105 completed the study and remaining 20 patients lost to follow-up. In regimen 1 and 2, significant number of patients showed good to excellent response whereas significant number of patients in remaining regimen group showed moderate to poor response with a p value of <0.05 [Table/Fig-3]. The comparison of therapeutic response between the regimens is presented in [Table/Fig-4,5,6,7&8]. No side effects were noted except for irritation at the site of application of topical keratolytics.
General characteristics of patients
| Regimen | Male | Female | Age (Mean) in years |
|---|
| 1 | 17 | 8 | 9.42±3.02 |
| 2 | 15 | 10 | 10.92±4.64 |
| 3 | 17 | 8 | 9.52±2.51 |
| 4 | 16 | 9 | 9.40±2.67 |
| 5 | 14 | 11 | 10.88±4.08 |
| Regimen | Grade of response n (%) | Lost to follow-up n (%) | Total n (%) |
|---|
| Poor | Moderate | Good to Excellent |
|---|
| 1 | 0 (0) | 4 (18.19) | 18 (81.81) | 3 (12) | 25 (100) |
| 2 | 0 (0) | 4 (16.67) | 20 (83.33) | 1 (4) | 25 (100) |
| 3 | 4 (20) | 13 (65) | 3 (15) | 5 (20) | 25 (100) |
| 4 | 15 (78) | 4 (21.05) | 0 (0) | 6 (24) | 25 (100) |
| 5 | 17 (85) | 3 (15) | 0 (0) | 5 (20) | 25 (100) |
Note: Refer [Table/Fig-1] for details of regimen used in the study
Comparison of therapeutic response between the regimens. Note: p value <0.05 is significant
| Regimens compared | X2 | p-value |
|---|
| 1 Vs 2 | 0.02 | 0.89 |
| 1 Vs 3 | 19.4 | <0.01 |
| 1 Vs 4 | 32.9 | <0.01 |
| 1 Vs 5 | 35.1 | <0.01 |
| 2 Vs 3 | 21.1 | <0.01 |
| 2 Vs 4 | 34.9 | <0.01 |
| 2 Vs 5 | 37.1 | <0.01 |
| 3 Vs 4 | 14.1 | <0.01 |
| 3 Vs 5 | 17.3 | <0.01 |
| 4 Vs 5 | 0.24 | 0.62 |
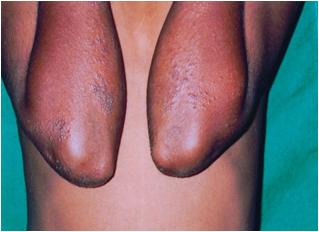
Before Vitamin A therapy (regimen 1)
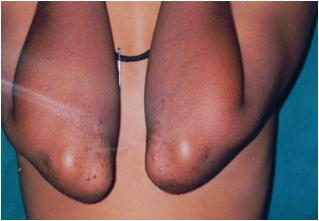
After 4 weeks of Vitamin A therapy showing excellent response with disappearance and flattening of lesions
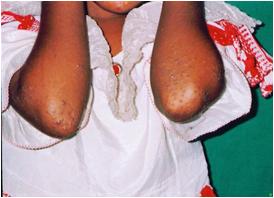
Before Vitamin B complex therapy (regimen 2)
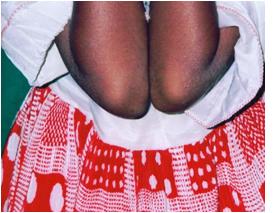
After 4 weeks of Vitamin B complex therapy showing excellent response with disappearance and flattening of lesions
Discussion
Phrynoderma has long been considered as one of the nutritional deficiency disorder commonly occurring in children between five and fifteen years of age. Recent studies have revealed a significant decrease in the proportion of phrynoderma cases owing to improvement in nutritional status of population due to implementation of various nutritional programs [2]. However, the exact aetiology of phrynoderma is yet to be established and hence, there is no single specific therapy available for the disease.
In the present study, the therapeutic response to Vitamin A and Vitamin B therapy was significantly better than other regimens. A complete response to Vitamin A therapy has been reported in patients who developed the disease after bowel surgeries [3,4]. This favours the popular assumption that Vitamin A deficiency is the cause for phrynoderma. However, the ocular manifestations which are considered as signs of Vitamin A deficiency are commonly seen in children below five years of age in whom the occurrence of phrynoderma is rare [2]. In addition, diet survey [5,6] and serum Vitamin A levels in apparently healthy phrynoderma patients and normal controls revealed no statistically significant difference between the two groups [7–9]. Moreover, the response to Vitamin A therapy has been reported to be poor [8,10,11]. Hence, the therapeutic response to Vitamin A therapy can be attributed to its effect on follicular keratinization. The response to Vitamin B therapy in various studies has been good [11–14]. When vitamins of B complex group were used alone there was no significant improvement in the disease [8,10,12]. However, when used together as Vitamin B complex significant improvement has been achieved. Though duration of therapy, mode of administration and composition of drug varied in different studies, a good response has been obtained by providing higher concentration of individual vitamins per day. It was achieved by giving oral Vitamin B tablets three times daily [13], Vitamin B forte tablets two times daily [12] or Vitamin B IM injections 2cc on alternate day for 10 to 15 injections [11,14]. Therefore, in the present study, the response to intra-muscular Vitamin B therapy was better and comparable with other studies.
The response to safflower oil, a rich source of linoleic acid (EFA), was moderate in the present study which is comparable with other study [13]. A good to excellent response has been obtained with linseed oil [6,15] and safflower oil [8,14,16]. However, in these studies longer duration of treatment up to 2-5 months was used. The response to Vitamin E therapy was poor in the present study which is lower than other studies where complete response after 10 weeks of treatment [13] and good response in 36.4% of patients [17] has been reported. The occurrence of EFA and Vitamin E deficiency is very unlikely in the absence of metabolic disorders. The dietary intake of EFA in Indian population has found to be sufficient as the poorest of Indian diet provides adequate quantity of linoleic acid [13,17]. The ratio of eicosatrienoic acid and arachidonic acid, an accurate indicator of EFA deficiency, was found to be 0.12 which is normal (<0.4) in patients with phrynoderma [13,16]. The levels of linoleic acid and phospholipids (sensitive indicators of EFA deficiency), and ratio of linoleic to arachidonic acid (indicator of efficient conversion of linoleic acid to arachidonic acid) were also normal in phrynoderma patients [13].
The combination therapy of Vitamin B-complex and EFA [8,10] or Vitamin B-complex and Vitamin E [17] have shown better results when compared to single drug therapy, and the serum levels of linoleic acid [13,16] and Vitamin E [17] also increased significantly during combination therapy, respectively. Therefore, Vitamin B appears to play a major role in the occurrence of phrynoderma. The cases of phrynoderma have been reported in apparently healthy patients in the absence of other signs of nutritional deficiencies [2,8,10,11]. This can be explained by role of individual vitamins of Vitamin B complex group in the metabolism of each other and EFA. The intersecting biochemical pathway demonstrates that riboflavin is required for pyridoxine metabolism which in turn is required for tryptophan- niacin metabolism, and niacin is necessary for fatty acid synthesis [18]. The lack of biochemical investigation is a drawback of the study. However, various studies have demonstrated normal levels of Vitamin A [7–9], Vitamin B [9,19] and EFA [13,16] in apparently healthy individuals with phrynoderma. In the presence of normal levels of nutrients, the occurrence of phrynoderma has been compared to formation of comedones in acne where a low level of linoleic acid in sebum has been attributed in the pathogenesis of follicular hyperkeratinization [20]
In conclusion, intra-muscular injections of Vitamin A or B complex are effective in the treatment of phrynoderma. Addition of topical keratolytics helps in faster clearance of keratotic lesions. However, considering the role of intersecting biochemical pathways and multifactorial nature of the disease, a combination therapy including Vitamins and EFA along with adequate nutrition will help in better management of patients with phrynoderma.
Note: IM, intramuscular; tbs, table spoon. * The patients in all five regimens received topical keratolytics † one ml contains thiamin 90.9mg, riboflavin 4.5mg, pyridoxine 4.5mg, niacinamide 90.9mg and vitamin B12 90.9μg
Note: Refer [Table/Fig-1] for details of regimen used in the study