Acute Exacerbation of Chronic Rhinosinusitis (AECRS) with Orbital Complications in an Atrophic Rhinitis Patient: A Mere Co-incidence?
Mainak Dutta1, Soumya Ghatak2
1 RMO-cum-Clinical Tutor, Department of Otorhinolaryngology and Head-Neck Surgery, Medical College and Hospital, Kolkata, India.
2 RMO-cum-Clinical Tutor, Department of Otorhinolaryngology and Head-Neck Surgery, R. G. Kar Medical College and Hospital, Kolkata, India.
NAME, ADDRESS, E-MAIL ID OF THE CORRESPONDING AUTHOR: Dr. Mainak Dutta, Department of Otorhinolaryngology and Head-Neck Surgery and Medical College and Hospital, Kolkata, India. 88, College Street, Kolkata – 700073, West Bengal, India.
Phone: +91-9830737587, Email: duttamainak@yahoo.com
Recurrent Acute Exacerbations of Chronic Rhinosinusitis (AECRS) in a setting of atrophic rhinitis are perplexing clinical situations. While either may act as the primary factor, repeated inflammation in a vessel-poor atrophied tissue is unusual. Progressive mucosal degeneration, vascularity and the effect of bacterial colonization are the factors suggested to play an important role for the pathobiogenesis of the co-existence of the two diseases. Through presentation of a case, a brief attempt has been made here to explore the temporal relationship between rhinosinusitis and atrophic rhinitis; in the process, other noteworthy features of the patient have also been highlighted.
Atrophic rhinitis, Rhinosinusitis, Orbital cellulitis
Case Report
An 18-year-old woman attended the Otorhinolaryngology clinic with a four-day history of huge, painful, bilateral erythematous periorbital swellings [Table/Fig-1]. She had associated nasal obstruction and foul-smelling purulent nasal discharge. Fever and headache were present for seven days. Detailed history taking revealed that she had been suffering from persistent nasal obstruction, mucopurulent nasal discharge and headaches since four years with repeated acute febrile episodes and had developed anosmia, sensation of nasal blockage and occasional expulsion of dry greenish crusts from the nose for the last one year. Others also complained of her fetid breath. She had continued to suffer from recurrent attacks of acute rhinosinusitis (two episodes) during the past year that were associated with fever, headache, facial pressure and nasal purulence.
This 18-year-old girl presented with bilateral cellulitis and abscess involving both upper and lower eyelids coexistent with an acute attack of rhinosinusitis
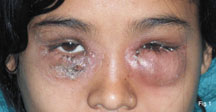
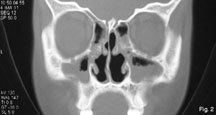
On examination, the periorbital swellings were tender, mostly with areas of cellulitis but studded with some diffuse pus points. The left side was affected more and she could barely open the eye. Chemosis, painful restricted eye movements, proptosis and visual impairment were absent. Anterior rhinoscopy revealed roomy nasal cavities with thick greenish crusts and mucopus. In subsequent diagnostic naso-endoscopy, additional findings included hypoplastic turbinates on either side. The uncinate processes were not seen and we could not examine the ostiomeatal units. The natural maxillary ostia were found blocked with dry, tenacious discharge and crusts. There was also post-nasal drip and mucopus around the Eustachian tube openings, more on the left. Some of the dried discharge and crusts from the nasal cavity were removed manually and subsequently in few sittings in the operation theatre applying 10% lignocaine spray. When asked during this period, she could not give any history of prior nasal surgery that could explain her clinical conditions like the small turbinates. CT scan of paranasal sinuses [Table/Fig-2] revealed pansinusitis, a relatively smaller left maxillary sinus, along with roomy nasal cavities and atrophied turbinates, suggestive of Atrophic Rhinitis (AR) with superimposed acute sinusitis. Diagnostic aspiration from the left lower eyelid revealed small collections of pus which was sent for staining, culture and antibiotic sensitivity along with a specimen of nasal swab taken concurrently. Haematology report showed that her haemoglobin was 9 gm% and serum iron level 45 μg/dl (normal, in adult female: 50-170 μg/dl) with normal vitamin B12 and folate levels. Her VDRL test was negative. She was put on empirical parenteral antibiotics (co-amoxiclav, 1.2 gm. thrice/day) and analgesics. The aspirate from the eyelid and the sample of nasal scraping both yielded Streptococcus pneumoniae after 72 hours of incubation, and was found sensitive to co-amoxiclav. The antibiotic was continued for seven days parenteral, and was switched over to oral co-amoxiclav (625 mg thrice/day for two weeks). The patient recovered gradually without any sequela. After the signs of periorbital inflammation and the acute episode of rhinosinusitis subsided completely, she was treated for her AR with nasal douching with sodium bicarbonate, sodium diborate and common salt (1:1:2) in lukewarm water, along with nasal drops containing 25% anhydrous glucose in glycerine. Oral medications containing haematinics, antioxidants and vitamin supplementations were also advised. She had been followed up for more than one year with satisfactory results.
CT scan of the paranasal sinuses (coronal section) shows pansinusitis and atrophied turbinates, with roomy nasal cavities. The left maxillary antrum is hypoplastic
Discussion
AR is a chronic, refractory and debilitating disease of the nasal cavity mucosa of unknown aetiology, characterized by progressive mucosal atrophy, atrophy of the underlying turbinate bones, abnormal widening/patency of the nasal cavities, with paradoxical nasal congestion, and formation of viscid secretions and dried crusts leading to a characteristic fetor (ozaena) [1–3]. It is taken to be a form as well as sequela of Chronic RhinoSinusitis (CRS) which is a known comorbidity of this disease [4–6]. However, it is unusual to have recurrent AECRS with complications in a young female suffering from AR.
Complications of acute rhinosinusitis have decreased in the antibiotic era [7], and the commonest of them are the orbital complications [8,9]. These are mostly seen in children [8] and in males [7], due to anatomical factors like abundance of diploeic veins and proximity with the paranasal sinuses. In contrast, our patient was an 18-year-old female who developed bilateral pre-septal cellulitis/inflammatory oedema (group 1; classification of orbital complications by Chandler et al.,) [10] along with lid abscess as complications of AECRS. Unlike the present case, an associated lid abscess, as documented in the recent reports, is only unilateral, involving either the upper or lower eyelid [11]. Apart from being the harbinger of other orbital complications, preseptal inflammation has the potential to cause intracranial complications if not addressed promptly.
The most intriguing feature in our case is the occurrence of recurrent episodes of AECRS in a setting of clinically and radiologically established AR. [Table/Fig-3] describes the probable clinical inter-relation between CRS and AR. Is this a mere correlation or is there any cause-effect relationship between AECRS and AR? It seems that this is a case of secondary AR occurring in a young female (where primary AR is expected to be more common); it is because the natural progression of the disease pathophysiology suggests that her four-year history of CRS with recurrent acute episodes has been followed by symptoms consistent with AR, the acute exacerbations still continuing. Secondary AR is seldom encountered in the tropics, the developing nations of eastern Asia, the Mediterraneans and eastern Europe – where the primary form is more common [12]. There is evident metaplasia of the respiratory mucosa into stratified squamous epithelium with or without keratinization along with atrophy of the nasal mucosa and the turbinates, with paucity of the mucosal glands, goblet cells, with periarteritis and endarteritis, leading to marked decrease in tissue vascularity [1,13]. So, it is quite unusual for an acute inflammatory process to recur so often in an atrophic tissue. But we could see in our patient that at least in this episode of recurrence, the inflammation had even proceeded to produce orbital complications [Table/Fig-3]. This may point out to the less common variant of AR characterized by vasodilatation, having bone eroding property as evident from the increased concentration of alkaline phosphatase in the capillary endothelium [12]; it seems quite possible that understanding the pathogenesis of this variety of AR (type II) probably holds the link that could help us associate the co-existence of AECRS (including the episodes of recurrence) and AR [Table/Fig-3]. Conventionally, type II AR is thought to constitute about 25-30% of all cases, in contrast to type I (about 50-80%) that has the characteristic “endarteritis obliterans” and consequently are hypovascular [12,13]. However, a recent study puts forward a contradictory view by stating that 80% of patients with clinical AR have capillary vasodilatation as the predominant vascular event [14]. This strengthens a probable cause-effect relationship between the two disease processes rather than speculating on coincidence. It, therefore, significantly lowers the threshold of suspicion of having one disease in presence of the other, irrespective of whichever being the primary.
The natural pathogenesis of the disease process – the inter-relation between chronic rhinosinusitis and atrophic rhinitis, and the concept of atrophic rhinosinusitis as a single disease spectrum
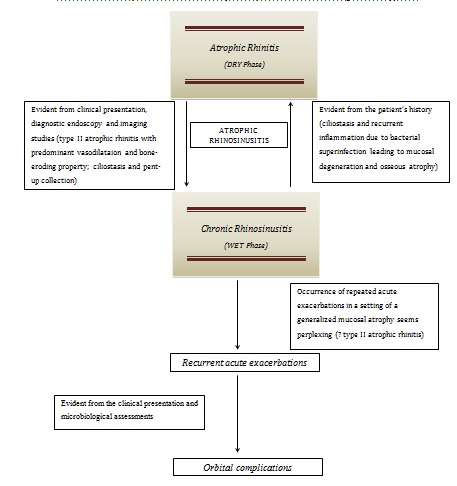
There have been documentations where CRS has been found to be associated with AR [4,6]. In a recent study by Bist et al.,[6], almost 87.7% of their study population suffering from AR had some grades of sinusitis, with more than 34% having grade III sinusitis according to the scale proposed by van der Veken et al., [15]. The pathological basis of such co-existence was not known. The probable explanation suggested was ciliostasis due to bacterial superinfection in a setting of atrophied, degenerated sinonasal mucosa. CT scan findings in the series revealed atrophy of bone and mucosa, widened nasal cavity, maxillary sinus hypoplasia, thickened medial wall of the maxillary sinus and outward bowing of the lateral wall of nasal cavity as the predominant radiological features [6], quite similar to what we had seen in our patient. Aptly therefore, the term atrophic rhinitis is increasingly being referred to as atrophic rhinosinusitis [3,5] – a single disease complex within a spectrum of a “wet” phase (acute and chronic sinusitis) and a “dry” phase (subsequent mucosal sclerosis with crusts) [3] [Table/Fig-3]. Ly et al., have also described the diagnostic criteria for secondary atrophic rhinosinusitis [5] – patients with CRS demonstrating for six months or more, any of the two features, namely, patient-reported recurrent epistaxis, episodic anosmia, physician-documented nasal purulence, nasal crusting, chronic inflammatory disease of the upper airway or two or more sinus surgeries. Though there was no prior nasal surgery or complaint of epistaxis in our patient, the typical history of CRS with recurrent exacerbations, anosmia, nasal purulence and crusting since one year, with the characteristic radiological features, put the present clinical condition in favour of the so-called atrophic rhinosinusitis [Table/Fig-3].
Conclusion
This case depicts an interesting clinical presentation where recurrent AECRS has occurred in a setting of clinico-radiologically established secondary AR in an 18-year-old woman, with one of the episodes being severe enough to result in bilateral orbital complications. These highlight the clinical intricacies in terms of temporal relationship between rhinosinusitis and AR, apart from stressing for proper vigilance in similar cases keeping a low threshold of suspicion, so that timely actions can be initiated to prevent impending complications.
[1]. Bahadur S, Thakar A, Specific chronic infections. Part 13: The Nose and Paranasal SinusesScott-Brown’s Otorhinolaryngology, Head and Neck Surgery 2008 7th. Ed.LondonHodder Arnold:1465Gleeson M. et al (Eds.) [Google Scholar]
[2]. Mishra A, Kawatra R, Gola M, Interventions for atrophic rhinitisCochrane Database Syst Rev 2012 2:CD008280(doi: 10.1002/14651858. CD008280 pub2) [Google Scholar]
[3]. deShazo RD, Stringer SP, Atrophic rhinosinusitis: progress toward explanation of an unsolved medical mysteryCurrent Opin Allergy Clin Immunol 2011 11(1):1-7. [Google Scholar]
[4]. Suzuki J, Oshima T, Watanabe K, Suzuki H, Kobayashi T, Hashimoto S, Chronic rhinosinusitis in ex-lepromatous leprosy patients with atrophic rhinitisJ Laryngol Otol 2013 127(3):265-70. [Google Scholar]
[5]. Ly TH, deShazo RD, Olivier J, Stringer SP, Daley W, Stodard CM, Diagnostic criteria for atrophic rhinosinusitisAm J Med 2009 122(8):747-53. [Google Scholar]
[6]. Bist SS, Bisht M, Purohit JP, Primary atrophic rhinitis: a clinical profile, microbiological and radiological studyISRN Otolaryngol 2012 (Article id: 404075). doi: 10.5402/2012/404075 [Google Scholar]
[7]. Bleier BS, Thaler ER, Complications of rhinosinusitisRhinosinusitis: A Guide for Diagnosis and Management 2008 New YorkSpringer:251Thaler ER, Kennedy DW (Eds.) [Google Scholar]
[8]. Oxford LE, McClay J, Complications of Acute Sinusitis in ChildrenOtolaryngol Head Neck Surg 2005 133(1):32-37. [Google Scholar]
[9]. Schramm VL, Myers EN, Kennerdell JS, Orbital complication of acute sinusitis. Evaluation, management and outcomeORL J Otolaryngol Relat Spec 1978 86:221-30. [Google Scholar]
[10]. Chandler JR, Langenbruner DJ, Stevens ER, The pathogenesis of orbital complications in acute sinusitisLaryngoscope 1970 80:141-28. [Google Scholar]
[11]. Remulla HD, Rubin PAD, Shore JW, Cunningham MJ, Pseudodacryocystitis arising from anterior ethmoiditisOphthal Plast Reconstr Surg 1995 11:165-8. [Google Scholar]
[12]. Dutt SN, Kameswaran M, The aetiology and management of atrophic rhinitisJ Laryngol Otol 2005 119:843-52. [Google Scholar]
[13]. Bist SS, Bisht M, Purohit JP, Saxena R, Study of histopathological changes in primary atrophic rhinitisISRN Otolaryngol 2011 Article ID: 269479). doi: 10.5402/2011/269479 [Google Scholar]
[14]. Bunnag C, Jareoncharsri P, Tangsuriyawong P, Bhothisuwan W, Chantarakul N, Characteristics of atrophic rhinitis in Thai patients at the Siriraj hospitalRhinology 1999 37:125-30. [Google Scholar]
[15]. van der Veken PJV, Clement PAR, Buisseret T, Desprechins B, Kaufman L, Derde MP, CT-scan study of the incidence of sinus involvement and nasal anatomic variations in 196 childrenRhinology 1990 28(3):177-84. [Google Scholar]