Diabetes Mellitus (DM) is a group of metabolic disorders and it is characterized by hyperglycemia. The current status of DM in India- around 51 million people are suffering from this disease in 2010 [1]. Its incidence is expected to rise by 170% in the next 20 years [2]. By 2030, patient load of Type 2 DM alone will be 79.4 million [3]. Even today, India has the largest population of DM patients in the world and studies say that India will become the global diabetes capital by 2050 [2,4].
DM is a chronic disease and has many chronic complications. Diabetic Nephropathy (DN) is one of its chronic micro-vascular complications. It develops in around 30% cases of DM and its incidence is increasing in the developing world especially in the South East Asia region which is most severely affected by this disease [5]. The prevalence of DN is increasing parallel to the increase in the prevalence of DM [6]. These statistics show picture that current status of diabetic nephropathy has reached the proportion of epidemic in developing countries including India. This condition of epidemic is extremely worrying because DN is the leading cause of End Stage Renal Disease (ESRD) worldwide as well in India accounting for approximately 30% of cases [7]. It is also the leading cause of DM related morbidity and mortality [8].
Chronic hyperglycemia is an important aetiological factor in the pathogenesis of DN [9–11]. Long duration of DM, smoking ,obesity and Family history are other risk factors which predispose to the development of DN and further progression of nephropathy in DM patients [11,12].
Micro-albuminuria is the earliest clinical manifestation of diabetic nephropathy. It is defined as appearance of low amounts of protein (albumin) in urine (30-300mg/day in 24 hours collection or 30-300 μg/mg creatinine in a spot collection). It progresses to overt proteinuria in 20-40% cases within 10 years period and further progresses to ESRD in 20% of cases [8,13]. Thus, Micro-albuminuria assessment is done for early diagnosis and screening of DN [8]. Micro-albuminuria may be present even before the diagnosis of DM (especially in Type 2 DM). At this juncture, it is a potentially reversible form of kidney injury. Therefore effective screening measures are required for early diagnosis [14].
For quantification of albuminuria, 24 hours urinary albumin concentration (24 hours UAC) in 24 hours urine sample (timed collection) is considered as gold standard. But it has major limitations of time consumption, sample collection errors, poor patient compliance and is expansive. To avoid this time consuming and cumbersome procedure, random urine sample can be used for quantification of albuminuria. Patient compliance is better with random urine sample collection. Urinary albumin concentration in random urine sample is variably attributable to circadian rhythm, variation in hydration status, diuresis, exercise and diet [15]. This variation can be eliminated by calculating random urinary albumin: creatinine ratio (RUA: C) in random urine sample. As it is the ratio of two stable rates, it eliminates the effect of time. Moreover, it is a comparison of the spot urinary albumin excretion to spot urinary creatinine excretion; hence if there is any change in glomerular filtration rate secondary to hydration status and other factors or any underlying pathology, the ratio remains unaffected [16]. Thus, RUA: C in random urine sample correlates well with 24 hours UAC in 24 hours urine sample [17].
This study aims at assessing the predictive value of random urine A:C ratio as a screening method for Micro-albuminuria assessment in DM patients as compared to 24 hours urine albumin.
Material and Methods
Study design: A cross sectional observational study was conducted.
Ethical considerations: Prior to the study, ethical clearance was taken from Institutional Ethical Committee of Veer Chandra Singh Garhwali Govt. Medical Sciences and Research Institute (L No: MC/STS/2012-13/07) on 17th April 2012.
The study was conducted from 20th April 2012 to 19th June 2012.
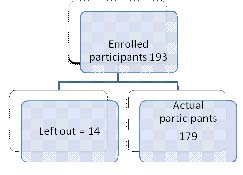
Sample size: 193 patients diagnosed with DM were enrolled in the study but 14 participants didn’t turn up with 24 hours urine sample. Thus, 179 people actually participated in the study.
Study population: All DM patients who attended Out Patient Departments (OPDs) and In Patient Departments (IPDs) of Medicine, Surgery and Orthopaedics of HNB Base Hospital and Teaching Institute, Srinagar (Garhwal) Uttarakhand, India during the study period participated in the study.
Informed and voluntary consent was taken from all the participants under study.
Inclusion criteria: Patients diagnosed with DM (according to WHO criteria) [8] who attended OPD and IPD, during the study period were included in the study.
Exclusion criteria: Patients who were suffering from hypertension, haematuria, fever and urinary tract infection were excluded on basis of relevant history, examination and investigation. Pregnant women were also excluded from study.
History was taken in the form of questionnaire: Information regarding gender, age, duration since diagnosis of DM, smoking, alcohol intake, medical history, dietary history, family history of DM and Diabetic Nephropathy, history of hypertension and any cardiovascular event and history of recent infection were taken and recorded.
Examination of patients: Height and weight measurement to know Body Mass Index (BMI), waist and hip circumference to know the waist: hip ratio (W: H ratio) and blood pressure measurements were taken.
Investigations: Fasting blood glucose measurement to know the current glycaemic status of patients. Fasting glucose was measured by ‘Oxidase-peroxidase method’. Urine analysis was done for urinary albumin and urinary creatinine. Two samples were collected from each participant. One, 24 hours sample and other random sample. Sterile urine container of 5L capacity, containing 5ml of 10% thymol in iso-propanol as preservative, was provided to the each participant for 24 hours urine sample collection and appropriate instructions were given to the participants for sample collection to minimise sample collection errors. Random samples were collected in sterile urine container (10 ml/capacity) on the day patient visited OPD. Twenty four hours urine samples were used to measure urinary albumin concentration while urinary albumin to creatinine ratio was measured from random urine sample. Urinary albumin was measured by ‘Pyrogallol Red method’ while urinary creatinine was measured by ‘Jaffe’s Spectrophotometric method’. The results of the tests were recorded on case record sheets.
Instructions given to the participants: Participants were instructed to begin urine collection immediately after discarding first morning urine and collect all urine in same container including final voided urine next morning till completion of 24 hours. Participants were instructed to collect mid-stream urine for random urine sample. Participants were also instructed not to indulge in heavy muscular work on the day of 24 hours collection and day of random sample collection.
Examination and investigations were recorded on case record sheet.
Those detected to have Micro-albuminuria or overt albuminuria; were appropriately prescribed by the physician.
Data Collection
Data was collected in the form of questionnaire including some relevant history points.
Instruments used during study were weighing machine, non-stretchable measuring tape, stethoscope, sphygmomanometer, containers for urine sample collection and kits for urine analysis, fasting blood glucose measurement.
All data were kept confidential and subject’s identity was not disclosed at any time during or after the research.
Results
A total of 193 patients suffering from DM.Out of 193 patients, 14 were lost to follow up. Thus, 179 patients with DM participated in the study. Out of 179 participant’s, 103 (57.54%) were male and 76 (42.46%) were female. Participants’ age ranged from 23 to 88 years, with an average of 56.42 ± 12.2 years [Table/Fig-1].
Characteristics of the study population are given in the [Table/Fig-2].
Characteristics of participants
| Particulars | Normo albuminuric | Micro albuminuric | Macro albuminuric |
|---|
| Duration since diagnosis of DM (years) | 4.82 ± 3.21 | 6.90 ± 4.29 | 11.29 ± 2.69 |
| BMI (Kg/m2) | 23.5 ± 3.5 | 24.03 ± 3.91 | 29.73 ± 1.43 |
| W:H ratio (Male) | 0.95 ± 0.04 | 0.96 ± 0.06 | 1.02 ± 0.03 |
| W:H ratio (Female) | 0.82 ± 0.03 | 0.86 ± 0.06 | 0.92 ± 0.03 |
| FBS | 190.92 ± 42.13 | 196.06 ± 56.73 | 253.67 ± 47.25 |
| 24 hour UAC | | 134.02 ± 80.55 | 764.57 ± 213.31 |
| RUA:C | | 127 ± 76.91 | 775.14 ± 246.82 |
Albuminuria assessment by using 24 hours urinary albumin concentration (24 hours UAC):
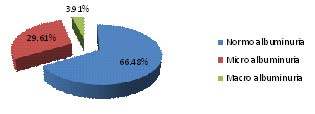
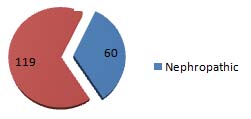
Out of 179 participants, 7(3.91%) participants were macro albuminuric and 53 (29.61%) participants were having urinary albumin concentration in Micro-albuminuria range and 119 (66.48%) participants were normal [Table/Fig-3]. Sixty (33.52%) participants were detected to have Diabetic nephropathy by using 24 hours UAC for albuminuria assessment [Table/Fig-4].
Albuminuria assessment by 24 hours urinary albumin concentration
Prevalence of Diabetic nephropathy by using 24 hours UAC
Albuminuria assessment by using random urinary albumin: creatinine ratio (RUA:C):
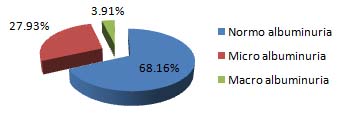
Out of 179 participants, the same 7 (3.91%) participants were having urinary albumin concentration in macro albuminuria range and 50 (27.93%) participants were in Micro-albuminuria range and 122 (68.16%) were found to be in normal range according to RUA:C ratio method [Table/Fig-5].
Albuminuria assessment by random urinary albumin: creatinine ratio
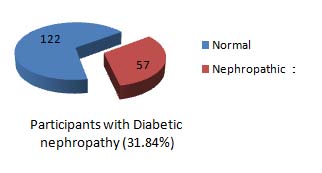
Out of 179 participants, 57 participants (31.84%) were found to have nephropathy by using RUA:C [Table/Fig-6].
Prevelance of Diabetic nephropathy by using RUA:C
Comparison of random urinary albumin: creatinine ratio (RUA:C) with 24 hours urinary albumin concentration (24hours UAC) for Micro-albuminuria assessment:
As shown in [Table/Fig-7], only 45 participants were found to be micro albuminuric by both methods viz. 24 hours UAC method and RUA:C ratio method. Sensitivity and specificity of RUA:C ratio method is 84.9% and 95.8% respectively, considering 24 hours UAC method as gold standard. Negative and positive predictive values of RUA:C ratio method are 0.93 and .090 respectively .Percentage of detecting false negative microalbuminuria was 15.1%.
Comparison of random urinary albumin: creatinine ratio (RUA:C) with 24 hours urinary albumin concentration(24hr UAC) for Micro-albuminuria assessment
| Micro-albuminuria by RUA:C | Micro-albuminuria by 24 hours UAC |
|---|
| POSITIVE | NEGATIVE |
|---|
| POSITIVE | 45 | 5 |
| NEGATIVE | 8 | 114 |
| Sensitivity of RUA:C = 84.9%, Specificity of RUA:C = 95.8%, Negative predictive value = 0.93, Positive predictive value = 0.90, False negative % = 15.1%, False positive % = 4.2% |
[Table/Fig-8] shows that there is no significant statistical difference between the results of 24 hours UAC method and RUA:C ratio method for Micro-albuminuria assessment with X2= 0.1247 and p-value= 0.724.
Comparison of number of participants for random urinary albumin: creatinine ratio (RUA:C) with 24 hours urinary albumin concentration(24hr UAC) for Micro-albuminuria assessment
| Micro-albuminuria Normo albuminuria | 24 hours UAC | RUA:C | X2= 0.1247 p value=0.724 |
|---|
| 53 | 50 |
| 119 | 122 |
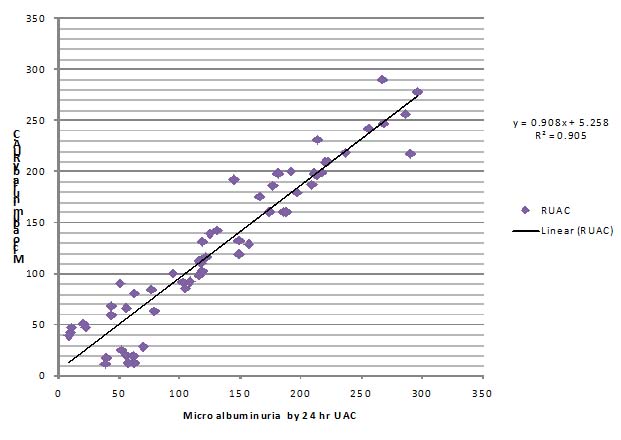
[Table/Fig-9] shows positive correlation between 24 hours UAC and RUA:C. (R2= 0.905: coefficient of determination, r = Square root of R2= 0.951: Pearson’s correlation co efficient) The area under the ROC curve for RUA:C at various cut-off was 0.849 (95% confidence interval,0.753-0.945; p= 0.01) [Table/Fig-10]. Sensitivity of 84.9% and specificity of 80% were achieved to detect proteinuria at the RUA:C ratio ≥33.5
Correlation between 24-hour UAC and RUA:C in Type 2 diabetes mellitus subjects
ROC curve at various cutoff points for RUA:C
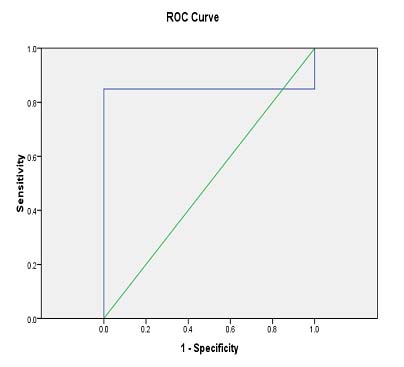
[Table/Fig-11] shows sensitivity and specificity of RUA:C at different cutoff levels.
Shows sensitivity and specificity of RUA:C at different cut off levels.
| Cut off value of RUA: C | Sensitivity | Specificity |
|---|
| 10.00 | 100% | 0% |
| 33.50 | 84.9% | 80% |
| 49.00 | 84.9% | 90% |
| 55.00 | 84.9% | 100% |
| 99.00 | 64.2% | 100% |
| 199.50 | 20.8% | 100% |
| 291.00 | 0% | 100% |
Discussion
In the light of high prevalence of DM and its chronic complication, Diabetic nephropathy; it is important to detect renal involvement promptly as renal involvement is reversible at the initial stage, and progression can be controlled. Early detection reduces both mortality and treatment cost in those affected. This can be done through screening of DM patients for Micro-albuminuria as its the earliest clinical manifestation of renal disease.
Twenty four hours UAC, although being gold standard for screening of Micro-albuminuria, has greater time constraints and its collection is difficult in OPD setup and in female patients especially in hilly regions like garhwal which is facing infrastructural problems in health sector and transportation. Considering this, random urine sample testing can be an easy alternative for screening of Micro-albuminuria. RUA: C in random urine sample correlates well with 24 hours UAC in 24 hours urine sample theoretically.
In the present study, Prevalence of Micro-albuminuria at tertiary health center is 29.61% by using 24 hours UAC method and 27.93% by using RUA: C method. This prevalence is comparable with other clinic based studies. John et al., [18], Gupta et al., and Yajnik et al., showed Micro-albuminuria prevalence of 19.7%, 26.6% and 23.0% in clinic based studies at Vellore, New Delhi and Pune respectively [19]. Another clinic based study by Varghese et al., at Chennai showed Micro-albuminuria prevalence of 36.3% [5]. The prevalence of overt nephropathy i.e., macro albuminuria in our study is 3.91%. Varghese et al., showed overt nephropathy prevalence of 2.2 % [5].
The present study shows the validity of RUA: C as screening method for diabetic nephropathy. Our results show positive correlation between 24 hours UAC and RUA: C with coefficient of determination R2 = 0.905 and Pearson’s correlation coefficient; r= 0.951. Biradar SB et al., [20] and Yadav BK et al., have also shown the similar results [13]. This suggest that RUA:C is appropriate for quantitative assessment of Micro-albuminuria and can be used instead of using 24 hours UAC. Moreover, the results showed high sensitivity (84.9%) and specificity (95.8%) with positive predictive value of 90% and negative predictive value of 93%.
The ROC curve analysis showed that the area under the curve is 0.849 indicating that RUA: C is sufficiently acceptable for estimation of Micro-albuminuria. Biradar SB et al.,) [20] and Yadav B K et al., [13] in their respective studies showed similar results (area under the ROC curve 0.947 and 0.88 respectively). Price et al., [15] reviewed 16 studies and the area under the curve was in the range of 0.70 to 0.90. Valizadeh M et al., [21] also found similar results with area under curve 0.764, suggesting that the RUA: C can assess the amount of albumin excreted in the urine. At the optimal cut-off point of 30μg albumin/mg creatinine, the sensitivity was 86% and specificity was 20 %. At higher cut-off point of 55μg albumin/mg creatinine; sensitivity and specificity were 84% and 100% respectively and at still higher cut-off point of 290 μg albumin/mg creatinine; sensitivity and specificity were 0% and 100% respectively.The area under the ROC curve and high sensitivity and specificity of RUA: C suggests that it can be used as screening method for detection of Diabetic nephropathy. But, it is important for each laboratory to determine the cut-off value of RUA: C which has maximum sensitivity and specificity to be used in those settings. This will aid in early diagnosis of DN as well as good compliance of the patients in areas of difficult terrain where the patients are lost to follow ups due to infrastructural issues.
The limitations of the present study were that the prevalence which is calculated in this study is the prevalence among those who attended tertiary health center which can’t be applied to general population. Further study is required to estimate the prevalence of Diabetic nephropathy in general population. This triggers the need for studies with bigger sample size to assess various mass screening programs that are patient friendly and cost effective.