Spontaneous Hepatocellular Adenoma in Paediatric Age Group – Case Report
Rajalakshmi Vaithianathan1, Philipchandran2, G. Selvambigai3, P. Jayaganesh4
1 Professor & Head, Department of Pathology, Esic Mc & Pgimsr,Chennai–78, India.
2 Professor, Department of Pediatric Surgery, Ich, Madrs Medical College, Chennai, India.
3 Associate Professor, Department of Pathology, Chengleput Medical College, Chennai, India.
4 Associate Professor, Department of Pathology, Tagore Medical College & Hospital, Chennai, India.
NAME, ADDRESS, E-MAIL ID OF THE CORRESPONDING AUTHOR: Dr. V. Rajalakshmi, 22/2 Murrays Gate Road, Alwarpet, Chennai–18, India.
Phone: 09841077698,
E-mail: raji_path@rediffmail.com
Hepatocellular adenoma (Hca) is a rare, benign, liver cell tumour. Hca is most frequently seen in women with a history of oral contraceptive use. Hca is also reported in children with glycogen storage disorders, galactosaemia, Hurler’s syndrome, severe immune deficiency states, diabetes mellitus, sex hormone disturbances, Fanconis anaemia, in those who are on androgen therapy and also in seizure disorder patients who are on carbamazepine therapy. Usually, Hca arises typically in a clinical setting of hormonal or metabolic abnormalities which stimulate hepatocyte proliferation. Though it is rare, a few cases of spontaneous Hca have been reported in children and also in adults, which were not associated with any of the known risk factors which are associated with Hca. Hca has to be differentiated from focal nodular hyperplasia, hepatocellular carcinoma and hepatoblastoma. The histological features and the assessment of cell proliferation by using immuno histochemistry, help in confirming the diagnosis of Hca. In a few cases of Hca, malignant transformations have been reported. Hence, a careful search for malignant transformations is necessary. In this report, we have documented two cases of spontaneous Hca which occurred in two normal children.
Liver, Hepatocellular adenoma, Paediatric age group, Spontaneous
Case 1
A six-year-old male child presented with abdominal pain and fever of four days duration, which was consistent with a clinical diagnosis of a liver abscess. Ultrasonogram of the (Usg) abdomen revealed a large, well-defined, 9 x 6 x 5 cms sized, echogenic mass in the right lobe of liver and computed tomography (CT ) of the abdomen revealed a large, well-defined, isodense mass in the right lobe of liver, with increased vascularity within the tumour, pushing the right kidney downwards. Haematological investigations, liver function tests and serum alpha foeto protein levels were all with in normal limits. Laparotomy and segmental liver resection were done and the specimen was sent for a histopathological examination.
Case 2
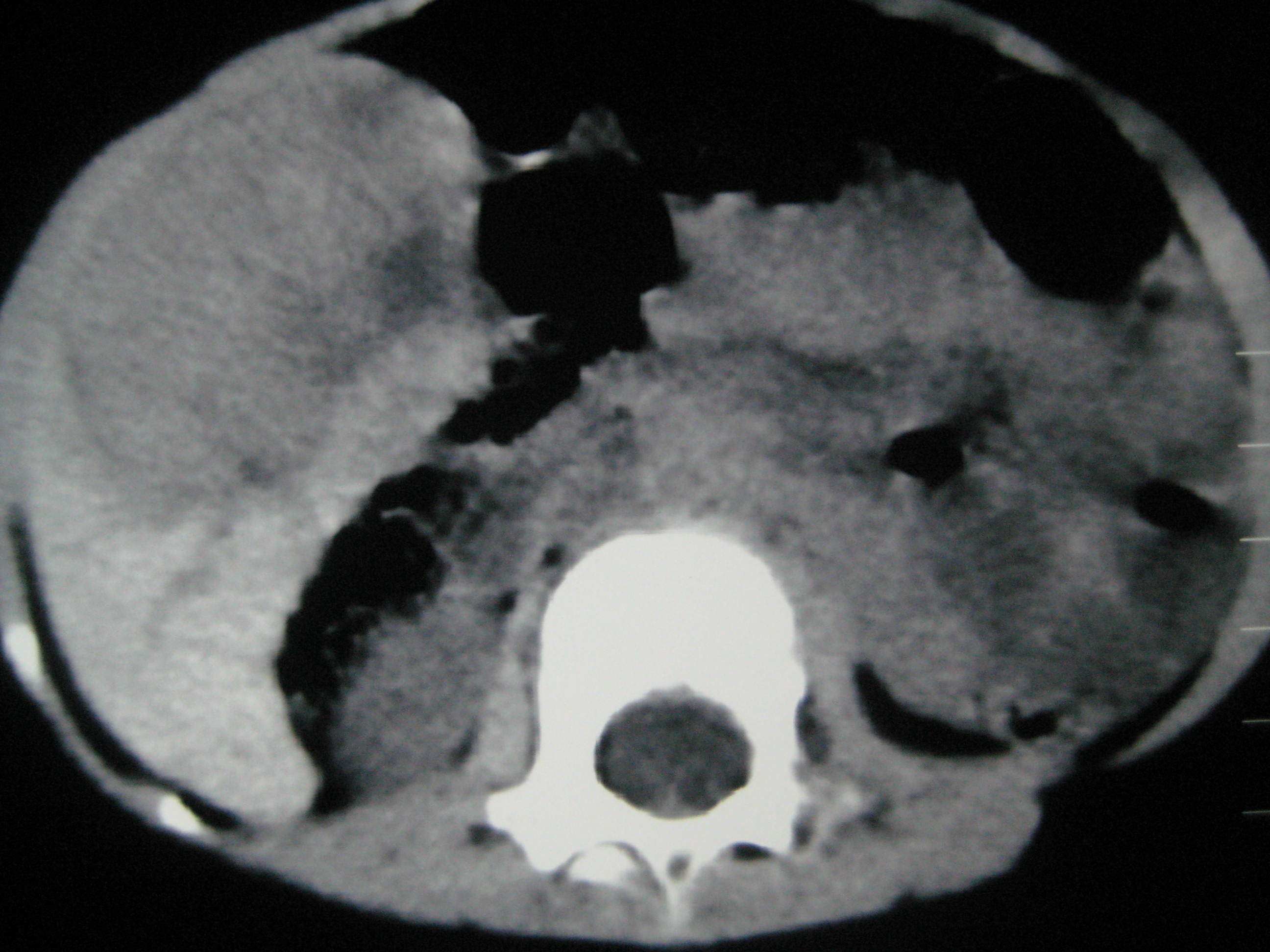
A 2–year–old female child presented with fever and incessant crying of two days duration, with a firm mass in the right hypochondrium, extending 3 cms below the costal margin. USG of the abdomen revealed a lobulated hyperechoic mass which measured 5.4x 5.5 x 5.6 cms, which had increased intra-tumoural vascularity. CT of the abdomen showed a non enhancing lesion and a radiological diagnosis of a mesenchymal hamartoma was made [Table/Fig-1]. Laparotomy and segmental liver resection were done and the specimen was sent for a histopathological examination.
Ct scan picture of case two showing a non enhancing lesion in the right lobe of the liver
Gross: Of case 1 showed resected portion of liver which measured 8 x 6x 4 cms, with a pedunculated mass of size, 6 x 4 cms. Cut section showed a circumscribed variegated mass with central yellow areas.
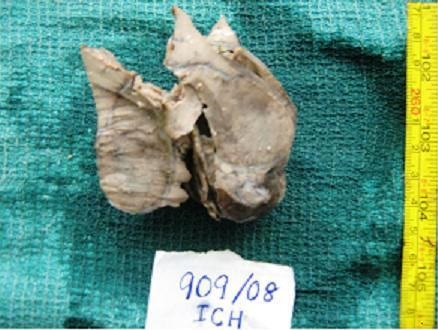
Gross: Of case 2 showed resected portion of liver which measured 6 x5 x 3cms, with a well-defined mass of size, 5x5 cms. Cut section showed a partly encapsulated, tan coloured mass with a wedge of normal liver tissue [Table/Fig-2].
Gross photograph of case no 2 showing a circumscribed mass rimmed by normal liver parenchyma at one resected margin
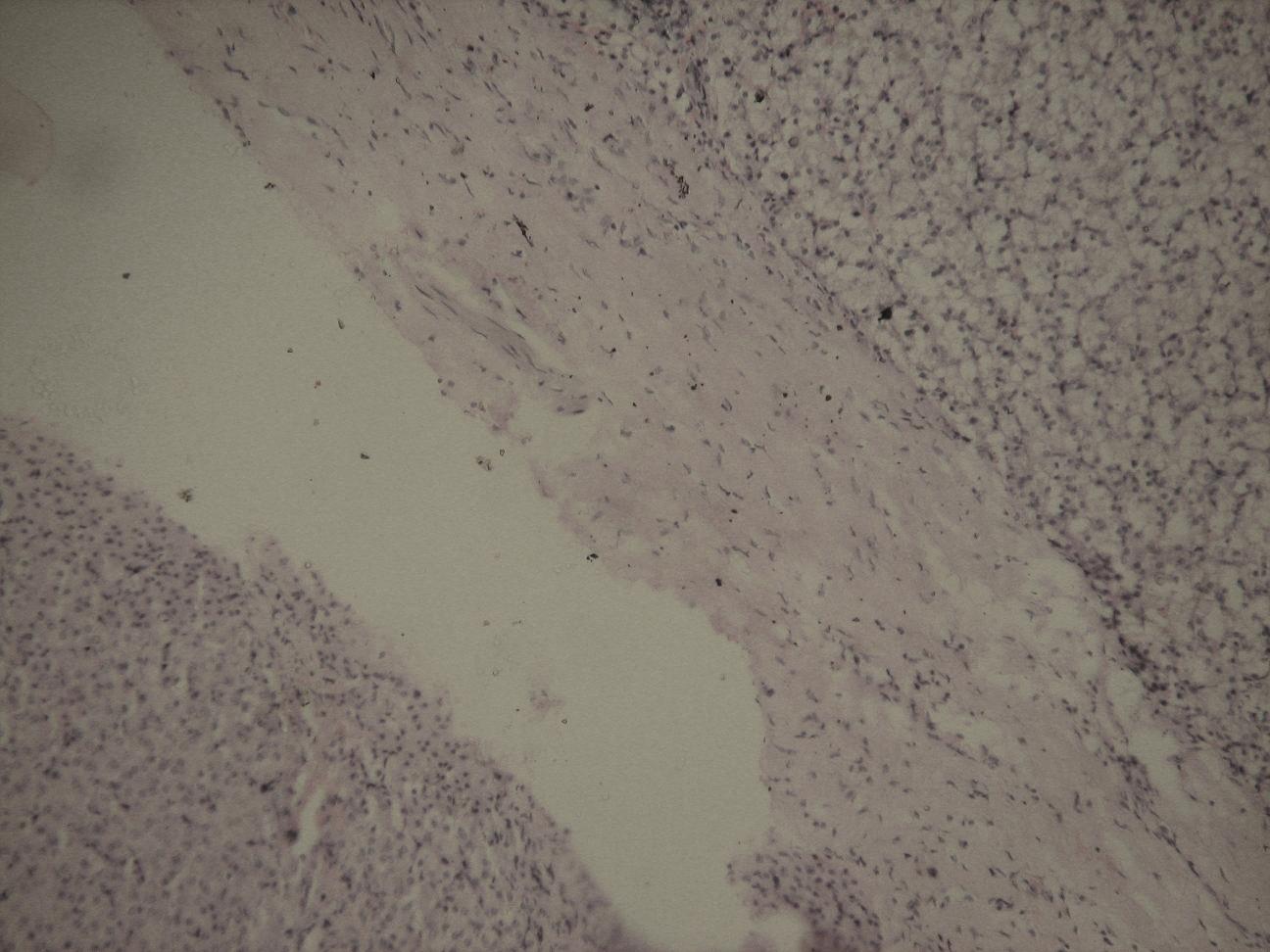
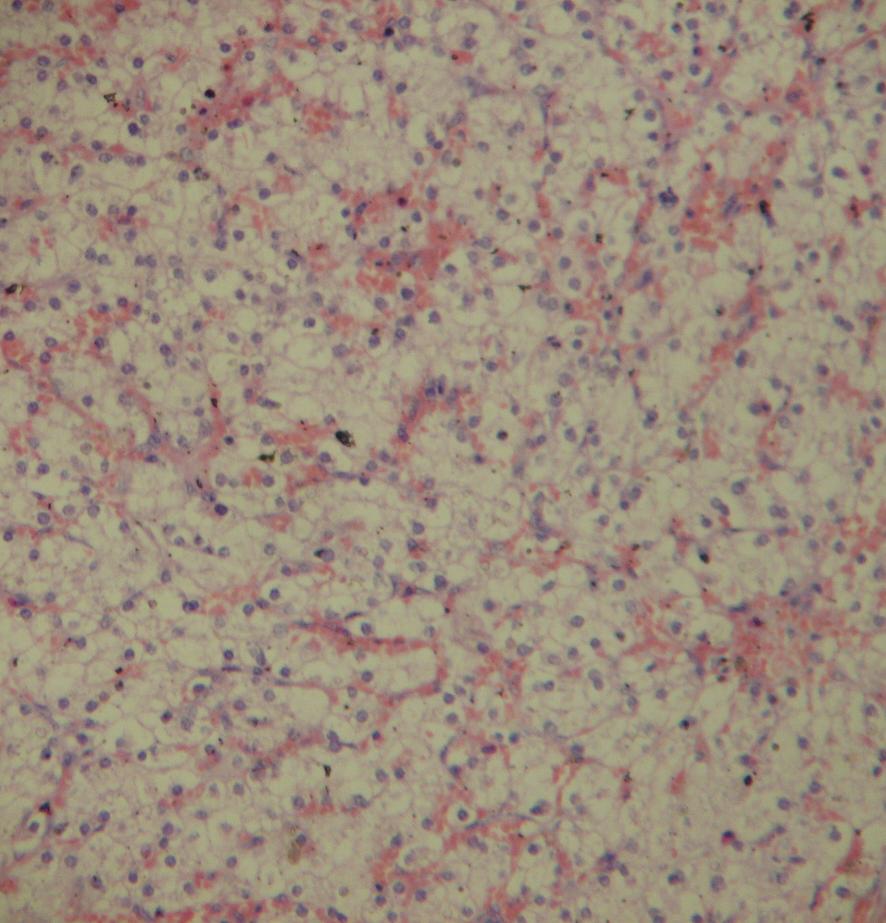
Microscopy of both the specimens showed partly capsulated neoplasms with slightly enlarged hepatocytes with pale cytoplasm, which were arranged in cords which had thicknesses of two to three cells. The nuclei were small and uniform, with a minimally increased nuclear cytoplasmic ratio. No nucleoli were seen. No dysplastic cells were seen. Minimal fibrosis was seen in occasio+nal foci. No bile ducts , no portal tracts or central veins were seen within the tumour. The surrounding liver parenchyma was normal in both the cases [Table/Fig-3,4].
(H&E,x 400) Circumscribed capsule rimming the neoplastic cells with normal liver parenchyma seen outside the capsule
(H&E,x400) Pale slightly enlarged hepatocytes arranged in cords in two cells thickness
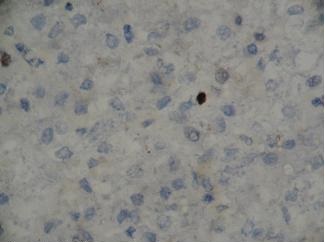
Ki - 67 labelling done by using MIB-1 monoclonal antibody showed only occasional nuclei which had taken up the stain [Table/Fig-5].
(MIB1ab x 400) Ki 67 immunostaining showing only occasional cells staining positive
Based on the above findings, a diagnosis of Hca was made in both the cases.
Discussion
Hca is a rare, benign liver cell tumour which always arises in the context of an abnormal hormonal or metabolic milieu [1–3]. Hca constitutes 2% to 4 % of all liver tumours in children. Hca is most frequently seen in women with histories of oral contraceptive use [1]. It is rarely seen in children with glycogen storage disorders, galactosaemia, Hurler’s syndrome, diabetes mellitus, in seizure disorder patients who are on carbamazepine therapy and in children who have Fanconi’s anaemia and who are on androgen therapy [1–3]. Giant Hca has been reported in a 16–year–old girl with a seizure disorder, who was on oxycarbazepine therapy [3]. A few cases of spontaneous Hca have been reported in children and in adults, which were not associated with any hormonal or metabolic abnormalities [1]. Trevor Lesse, et al., in their 12 years of surgical experience on liver cell adenomas, have reported twenty three cases, among which seven cases were of spontaneous type, with the rest of the cases having either hormonal or metabolic abnormalities [4]. Patients can be asymptomatic or they can present with episodic abdominal pain. They may also present with acute abdominal pain caused by a haemorrhage into the tumour or into the peritoneal cavity. As has been seen in the literature, in our two cases, haematological investigations, liver function tests and serum alpha foetoprotein levels were within normal limits.
Usually,Hca is an encapsulated or a partly capsulated, circumscribed, single lesion that can be differentiated from liver cell adenomatosis, which shows multiple nodules. Its size may vary from few cms to 15 cms. Areas of necrosis can be seen. Encapsulation, absence of central veins, absence of bile ducts and absence of portal tracts differentiate Hca from normal liver parenchyma. Hca is differentiated from Fnh by the absence of a central fibrous scar. Hca is differentiated from well–differentiated hepatocellular carcinoma by normal levels of serum alpha foetoprotein, microscopically by the absence of a trabecular pattern of tumour cells with thickness of more than three cell layers, non pleomorphic nuclei with a nuclear cytoplasmic ratio which is similar to that of non neoplastic hepatocytes and by the absence of vascular invasion by the tumour cells. As malignant transformationa have been reported in few cases of Hca, a thorough sampling is essential, to rule out a malignant transformation. Histologically, Hca is differentiated from foetal type of Hbl in that the cells show a normal nuclear cytoplasmic ratio, unlike in Hbl, in which the cells show a high nuclear-cytoplasmic ratio [3]. Serum levels of alpha foetoprotein are of value, as they are elevated in Hbl.
The assessment of cell proliferation activity is very useful in differentiating Hca from Hcc and Hbl .Resnick M .B. et al., [2], in their study, found PCNA-Labelling Index (LI) to range from 0.3% to 1.7% in Hca , from 21.8% to 44.3% in Hbl and from 9.6% to 23.8 % in Hcc.
Girigioni et al., [5], in their study, measured the proliferation index by using antibody to Ki-67 antigen .They found that Ki-67 –LI in their two cases of Hca was less than 5%, in contrast to the LI of more than 15% which was seen in all the 14 cases of Hcc. In our two cases, Ki 67, which was assessed by using a monoclonal antibody to MIB-1, showed that only occasional nuclei took up the stain. The Ki-67 LI was less than 5 %, which confirmed the diagnosis of Hca [2].
A complete surgical resection of Hca is recommended whenever it is technically feasible, to prevent a risk of rupture, haemorrhage and a malignant transformation.
In this communication, we have highlighted two rare cases of spontaneous Hca in children, which were not associated with abnormal hormonal or metabolic milieu.
[1]. Resnick M.B, Kozakewich HPW, Perez–Atayde AR, Hepatic adenoma in the pediatric age groupAm J Surg Pathol 1995 19:1181-90. [Google Scholar]
[2]. Wheeler DA, Edmondson HA, Reynolds JB, Spontaneous liver cell adenoma in childrenAm J Clin Pathol 1986 85:6-12. [Google Scholar]
[3]. Lautz TB, Finegold MJ, Chin AC, Superina RA, Giant hepatic adenoma with atypical features in a patient on oxcarbazepine therapyJ Ped Surg 2008 43:751-4. [Google Scholar]
[4]. Trevor Lesse, Olivier Farges, Henry Bismuth, Liver Cell Adenomas: A12-years Surgical Experience From a Specialist Hepatpto-Biliary UnitAnn Surg 1988 208:558-64. [Google Scholar]
[5]. Girigioni WF D, Erico A, Bacci F, Primary liver cell neoplasms: evaluation of proliferative index using MoAb Ki67J Pathol 1989 158:23-29. [Google Scholar]