Granulocytic sarcoma is a rare variant of a myeloid malignancy, which shows an extra-medullary tumour mass which is composed of myeloblasts and myeloid precursors with varying degrees of differentiation. It occurs most commonly in bone, periosteum, soft tissue, lymph nodes, and skin; although it can occur anywhere throughout the body. Here, we are reporting two cases of orbital granulocytic sarcoma in children, which presented clinically with proptosis and periorbital swellings, which were first diagnosed by Fine Needle Aspiration Cytology (FNAC). Later, peripheral blood and bone marrow aspirate examinations revealed the evidence of Acute Myeloid Leukaemia (AML). These cases are being documented to demonstrate the utility and diagnostic accuracy of FNAC in evaluation of this entity, in cases of unsuspected AML. Recognition of this rare entity is important, because giving an early aggressive chemotherapy can cause regression of the tumour and thus improve the patient survival.
AML, Auer rod, FNAC, Granulocytic sarcoma, Orbit
Case Report
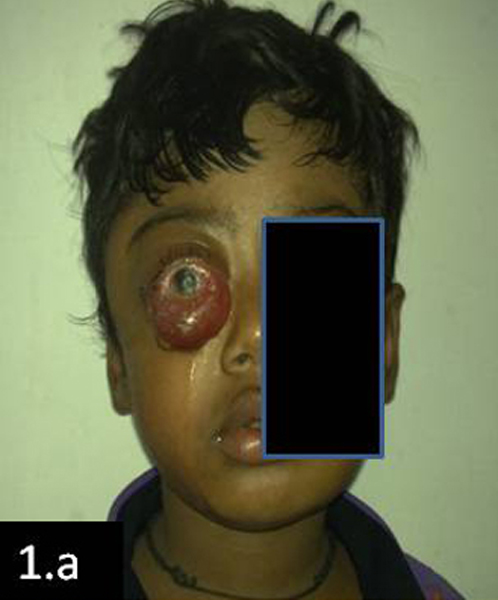
Case 1: A 7 years old male presented with swellings in left periorbital and right temporal regions, which were there since ten days. He also complained of fever and weakness since one month. On examination, it was found that there was severe proptosis and chemosis in left eye [Table/Fig-1a]. The swelling in temporal region was 3 x 2 cm., firm, non–tender and fixed. A clinical diagnosis of retinoblastoma was made.
Child presenting with an orbital mass (case 1)
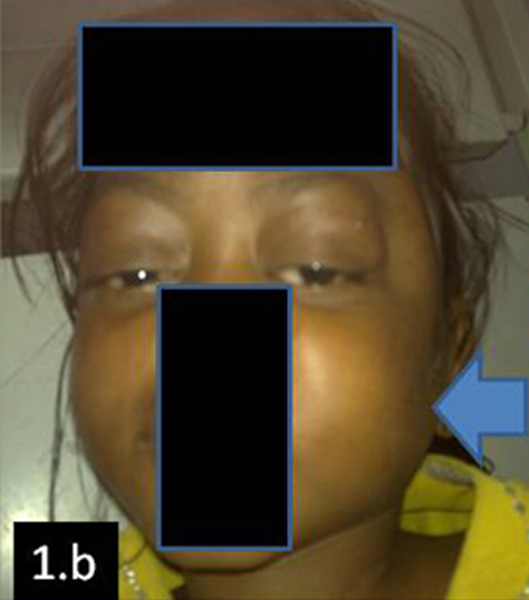
Case 2: A 10 years old female [Table/Fig-1b] presented to us with complaints of proptosis, a left retromandibular swelling, fever and weight loss, which were there since the past two months. On examination, the swelling was found to be 2 x 2 cm in size, firm to hard, mobile and nontender.
Child presenting with proptosis and left retromadibular swelling (case 2)
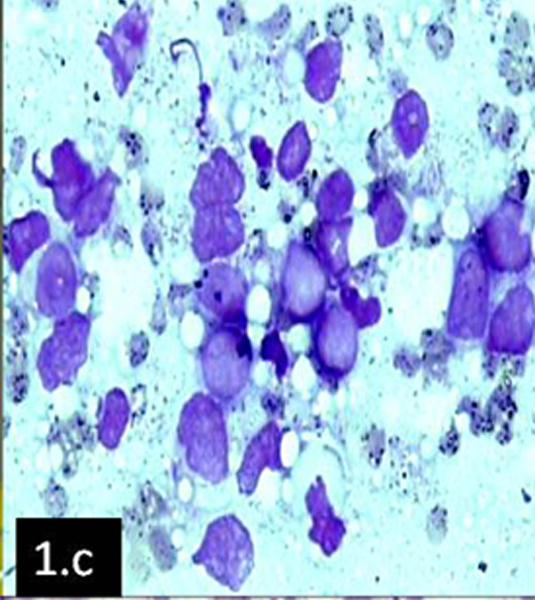
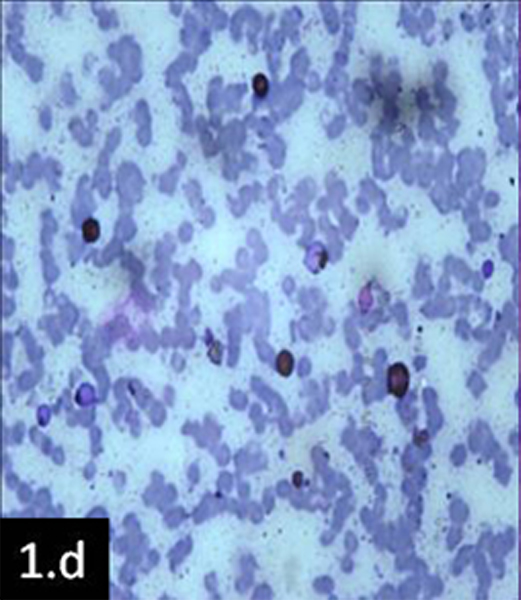
FNAC was advised as the first diagnostic procedure in both cases. No other documentation of previous investigations was available. Fine needle aspiration (not image guided) was done after getting proper informed consents and smears were prepared from the swellings of both patients. They were stained with Giemsa and Papanicolaou stains, which revealed similar pictures. Smears showed clusters of atypical cells [Table/Fig-1c] with a high nuclear cytoplasmic ratio, open chromatin, one to two prominent nucleoli, a deep blue rim of cytoplasm and fine azurophilic granules (? blast cells). In case 2, the presence of occasional atypical cells suggested the presence of Auer rods. Special staining for Sudan Black B, which was done, showed positivity [Table/Fig-1d] in both cases and diagnosis of Granulocytic Sarcoma were considered.
FNA smear showing numerous blast cells in clusters (Giemsa x400)
FNA smear showing Sudan Black B positivity in blast cells (x100)
Later on, peripheral blood and bone marrow aspiration smears were prepared from both patients.
Case 1: Peripheral blood showed a total leucocyte count which was within normal limits (9,500cells/mm3). His differential leucocyte count showed 12% neutrophils, 56% lymphocytes and 32% blasts. The bone marrow aspirate showed hypercellular marrow smears with a blast percentage of 63%.
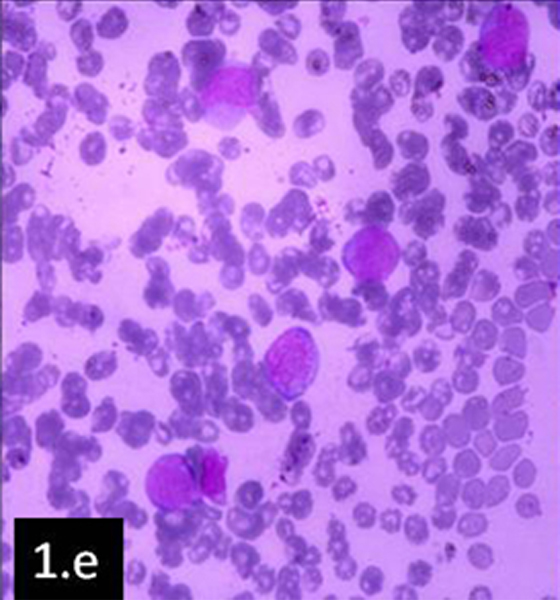
Case 2: Peripheral blood showed an increased total leucocyte count (16,000cells/mm3). Her differential leucocyte count revealed 5% neutrophils, 23% lymphocytes, 3% eosinophils, 1% monocytes, 56% myeloblasts (some of them showed Auer rods) and immature myeloid precursors which included promyelocytes, myelocytes and metamyelocytes. The bone marrow aspirate showed hypercellular marrow smears with replacement of haemopoietic cells by blasts [Table/Fig-1e].
Peripheral blood smear showing blast cells (Leishman x400)
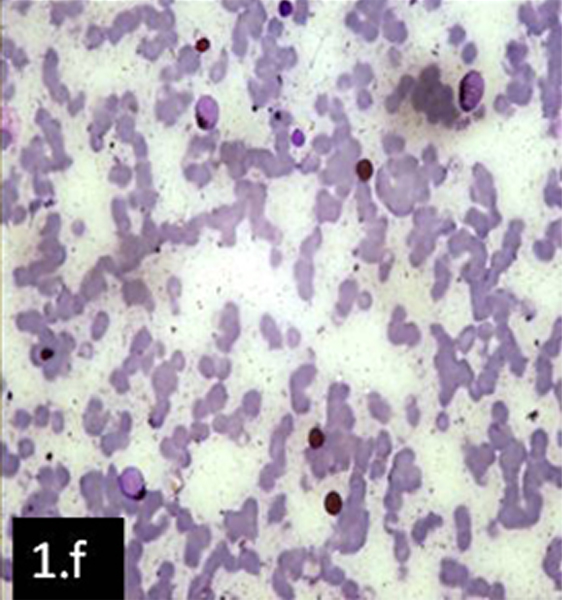
Blasts were Sudan Black B positive in both cases [Table/Fig-1f].The findings in both cases were suggestive of AML. Flow cytometric studies were advised. Patients were lost for follow up.
Peripheral blood smear showing Sudan Black B positivity in blast cells (x100)
Discussion
Granulocytic sarcoma (GS) is a tumour mass which consists of myeloid blasts with or without maturation, which occurs at an anatomical sites other than the bone marrow [1]. Granulocytic sarcomas (also called chloromas) were first described by the British physician, A. Burns in 1811.However, because upto 30% of these tumours can be white, gray or brown rather than green, the more correct term, ‘granulocytic sarcoma’ was proposed by Rappaport in 1967 [2].
Granulocytic sarcomas are associated with AML (FAB sub Types M2, M4 and M5); they may also be associated with myelodysplastic syndrome (MDS) and myeloproliferative disorders (MPS). Very rarely can granulocytic sarcomas can arise de novo without a known pre-existing or concomitant diagnosis of acute leukaemia or MDS/MPS [2].The WHO has classified granulocytic sarcomas into 3 main types, depending on the degree of maturation (i) Blastic – which is composed mainly of myeloblasts (ii) Immature- which is composed of myeloblasts and promyelocytes and (iii) Differentiated- which is composed of promyelocytes and more mature myeloid cells [1, 2].
Granulocytic sarcoma develops preferentially in bones or periosteum (especially in the skull) and it may lead to proptosis [3]. The proptosis in these cases is mainly caused by leukaemic infiltrates, a retrobulbar haemorrhage, an orbital muscle infiltration or a venous blockage. Other presentations which are caused by an orbital involvement include ptosis, lacrimal gland involvement, conjunctival masses and an iridic and a diffuse uveal involvement. Most of the reported cases had decreased visual acuity and restricted extra-ocular movements [4].
It presents a diagnostic dilemma when it precedes the prior diagnosis or development of other haematological or systemic manifestations. Patients with isolated GS have good prognosis if they are treated with current AML therapy. For those patients who have GS in addition to a marrow involvement, the patients with disease which is limited to the skin do worse than those without GS; those with AML that involves sites other than skin (e.g., orbit, head, and neck), have similar prognosis as those of patients who have bone marrow leukaemia alone [5].
An extra–medullary leukaemic infiltration is a bad prognostic sign, because it might act as a reservoir for proliferation of leukaemic cells and an eventual relapse. In such cases, fine needle aspiration cytology may be a useful diagnostic tool for making an early diagnosis. Cytological preparations give us better morphological details of blasts; the identification of Auer rods and cytochemistry can help in clinching the diagnosis of an undetected AML [6]. As in our two cases, FNAC along with special cytochemistry can help in deciding on the diagnosis.
The diagnosis of a granulocytic sarcoma can be difficult and sometimes it may be misdiagnosed. These tumors can occur anywhere in the body and they have to be differentiated from lymphomas, carcinomas or infectious process [4]. In the first case, the clinical diagnosis of retinoblastoma was contemplated and FNAC was advised as the primary investigation. Recognition of this rare entity is important, because giving an early aggressive chemotherapy can bring about regression of the tumour and improve survival.
In our cases, based on the locations of the tumours and cytological examinations of the fine needle aspiration smears, small round cell tumour or granulocytic sarcoma were considered the most probable diagnosis. Further investigations- general blood pictures, bone marrow morphologies and special staining (Sudan Black B) helped in making the diagnosis. Leukaemic proptosis, however, may not always be associated with leukocytosis or immature cells in the peripheral smear. Studying peripheral smears along with bone marrow aspirates and biopsies in all patients of AML who manifest with proptosis, who are in the paediatric age group, is therefore justified [7].
Conclusion
Our case report demonstrates the clinical utility and accuracy of FNAC in making the diagnosis of GS from an extramedullary sites. The work-up of these masses can be aided by the use of cytochemistry (MPO, Sudan Black and nonspecific esterase) and immunophenotyping. The correct recognition of this entity and its differentiation from other mimics, are crucial to make an accurate diagnosis, for a proper patient management and for an appropriate follow-up.
[1]. Jaffe ES, Harris NL, Stein H, World Health Organisation Classification of TumoursTumours of Haematopoietic and Lymphoid Tissues 2001 :105 [Google Scholar]
[2]. Kundu S, Chatterjee S, Mondol D, Dastidar AG, Roy A, Extramedullary granulocytic sarcomaIndian J Med Paediatr Oncol 2008 29:28-30. [Google Scholar]
[3]. Alkatan H, Chaudhry I, Myeloid sarcoma of the orbitAnn Saudi Med 2008 28:461-65. [Google Scholar]
[4]. Bangerter M, Hildebrand A, Waidmann O, Griesshammer M, Diagnosis of Granulocytic Sarcoma by Fine-Needle Aspiration Cytology Acta Haematol 2000 103:102-08. [Google Scholar]
[5]. Dusenbery KE, Howells WB, Arthur DC, Extramedullary leukaemia in children with newly diagnosed acute myeloid leukemia: a report from the Children’s Cancer GroupJ Pediatr Hematol Oncol 2003 25:760-68. [Google Scholar]
[6]. Tangri R, Prakash N, Khurana A, Myeloid sarcoma of the orbitJ Cytol 2008 25:111-12. [Google Scholar]
[7]. Rajput D, Naval R, Yadav K, Tungaria A, Behari S, Bilateral proptosis and bitemporal swelling: A rare manifestation of acute myeloid leukemiaJ Pediatr Neurosci 2010 5:68-71. [Google Scholar]