Intraocular pressure (IOP) is one of the vital factors, whose maintenance within normal limits is essential for the eye to serve its function as a light gathering and a transducer organ. It is the pressure within the eye which occurs as a result of an intricate balance between constant formation and drainage of the aqueous humor [1]. Longstanding elevations of IOP, above the normal level, may lead to glaucomatous visual field defects. Although raised IOP may not necessarily be the only risk factor for a glaucomatous optic nerve damage, the development of a glaucomatous optic nerve damage, as is observed by visual field loss and/or optic disc cupping, is more likely to be associated with an abnormally high intraocular pressure [2].
A number of physiological factors may influence IOP in normal subjects and these effects may sometimes be marked and relatively sustained [3]. Few of these physiological variables include factors like age, sex, body position, Valsalva maneouver, exercise, diurnal variations, pregnancy and sex hormones [4].
Previous studies have shown that females recorded consistently higher values than age matched males. A routine assessment of IOP is usually confined to the clinical population which is aged over 40 years because these individuals are considered to be most at a risk of developing glaucoma [5]. It is a well known fact that there are variations in female sex hormones during the reproductive years and after menopause, which may influence the IOP in these populations. Determining IOP distribution in different ethnic populations and factors associated with it can also be helpful in understanding the differences in glaucoma prevalence and its associated factors in different areas of the world. However, scarcity of data in south Indian populations regarding this issue and the fact that observations in other populations cannot be construed as “gospel truth”, necessitated the present study, whose twin objectives were to observe the effect of menopause on IOP and also to explore its relationship with Body Mass Index (BMI).
Material and Methods
The present study was carried out at Kasturba Medical College Hospital, Mangalore, India, after obtaining the appropriate institutional ethical clearance. A total of 120 females who were in the age group of 40 – 55 years were recruited for this study. They were divided into two groups of 60 each: premenopausal and postmenopausal.
Sixty healthy females who were aged below 55 years, who had amenorrhoea for more than one year, were considered as qualifying to be the postmenopausal group, whereas 60 healthy females who were still menstruating were considered as premenopausal group. Individuals beyond the age range (i.e. 40 – 55 years), with a history of premature menopause/surgical menopause, with diseases like diabetes mellitus, hypertension, hypothyroidism and liver damage, who were on medications (such as anti – inflammatory, ocular medications and steroid hormones), with a history of ocular diseases (refractive errors, glaucoma, etc), and those who underwent eye surgeries within the past three months were excluded.
A detailed history was taken and a thorough clinical examination was done after obtaining informed written consents from all the participants. Height was measured to the nearest of 0.1 centimetre without footwear, by using a vertically movable scale. Weight was measured to the nearest 100 grams by using a digital scale and Body Mass Index (BMI) was calculated. Resting blood pressure (systolic and diastolic) was measured by using a mercury sphygmomanometer, with the subject in a supine posture and after a rest of 15 minutes in this posture. IOP was recorded by using a Schiotz tonometer and following the standard protocol, with the subject in supine position. The scale of Schiotz tonometer was calibrated in such a fashion that each scale unit represented 0.05 mm protrusion of the plunger. The recording of the IOP was always started with 5.5 gms weight. However, if the scale reading was less than one, additional weight was added to the plunger to make it 7.5 gm or 10 gm, as indicated. A conversion table or a Friedenwald nomogram was then used to derive the IOP in mm Hg from the scale reading and the plunger weight [6].
Statistical Analysis
Obtained data was analysed by using independent ‘t’ test for comparison of BMI, BP and IOP between the groups. Pearson’s correlation was used to find the correlation between BMI and IOP. A p value of less than 0.05 was considered to be statistically significant. All the statistical analysis was done by using SPSS, 11.
Results
The postmenopausal women had significantly higher BMIs as compared to the women with normal menstrual cycles. However, the blood pressure (systolic and diastolic) was similar in both the groups [Table/Fig- 1].
Showing Age, BMI and Blood Pressure recordings in both groups
| Premenopausal | Postmenopausal | p value |
|---|
| Age | 42.67 + 1.77 | 50.17 + 2.29 | <0.0001* |
| BMI | 22.9 + 3.84 | 27.39 + 4.27 | <0.0001* |
| Systolic Blood Pressure
(mm of Hg) | 117.53 + 7.69 | 120.03 + 8.24 | 0.0594 |
| Diastolic Blood pressure (mm of Hg) | 74.93+ 6.99 | 76.37+ 6.84 | 0.2564 |
Values are expressed as Mean + Standard deviation, *significant
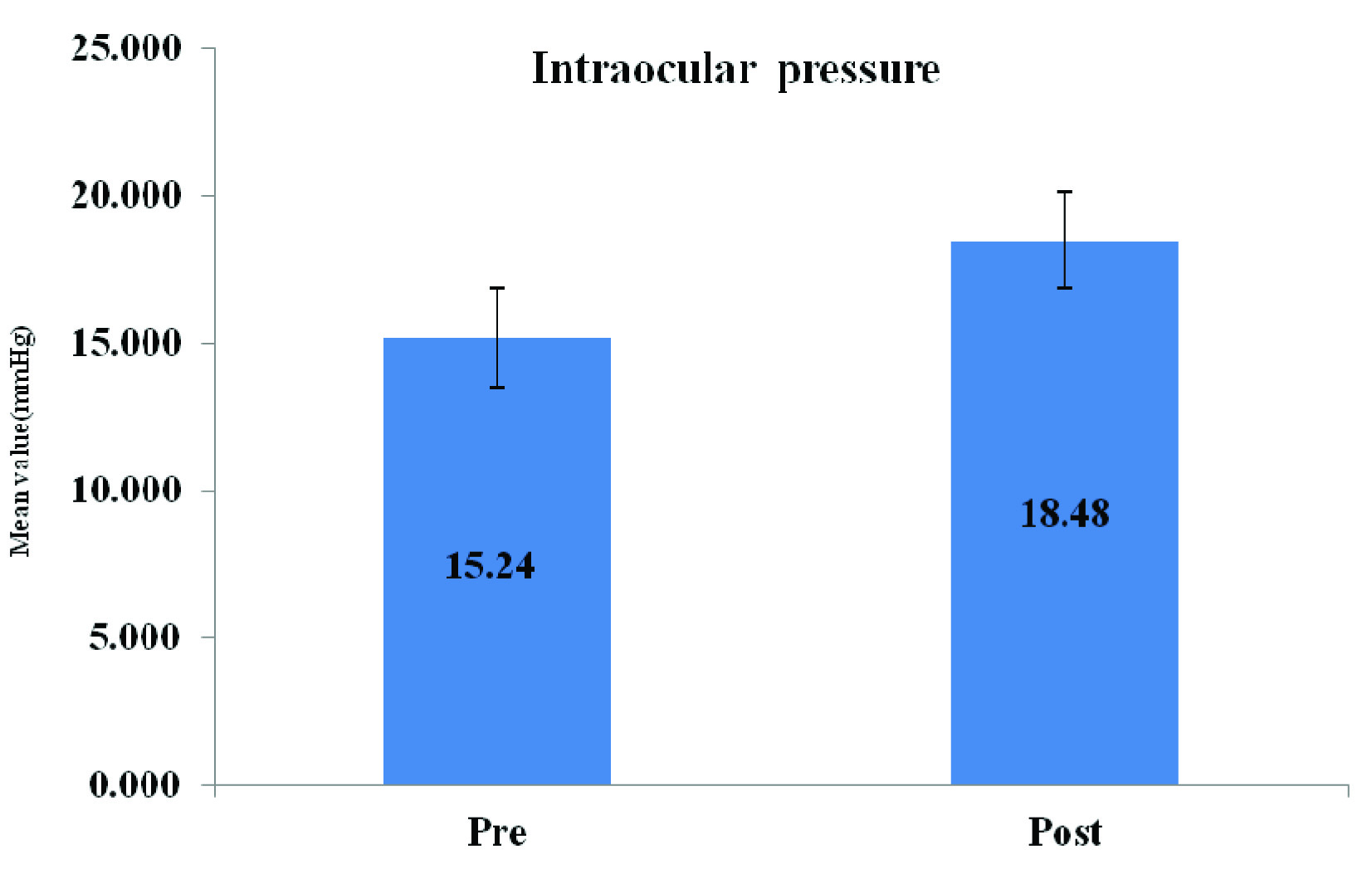
Mean values of IOP were significantly higher in postmenopausal women as compared to those in premenopausal females, but they were still were within normal limits [Table/Fig- 2].
Showing IOP Values in both groups
The intraocular pressure exhibited a positive correlation with BMI, but this was significant enough only in postmenopausal women [Table/Fig- 3].
Showing Correlation of IOP with BMI
| r value | p value |
|---|
| Premenopausal | 0.128 | 0.16 |
| Postmenopausal | 0.381 | 0.002* |
*Significant
Discussion
The intraocular pressure is maintained within the normal range of 10-20 mmHg, by maintaining a balance between various mechanical parameters. Values between 21-24 mm Hg are regarded as suspicious, whereas pressures which exceed 24 mm Hg are considered to be abnormal [7]. In the present study, we observed significantly higher IOP in postmenopausal group as compared to those in premenopausal women. One of the important differences between the menstruating and postmenopausal women is the hormonal levels, with postmenopausal period being characterised by very low oestrogen and progesterone levels. It is probable that the decrease in oestrogen and progesterone levels after menopause may play a key role, by altering any/several components of the IOP regulating mechanisms. It is a well known fact that oestradiol increases endothelial nitric oxide levels by enhancing the activity of the enzyme nitric oxide synthase III [8]. Many studies have also reported that nitric oxide produces a decrease in intraocular pressure by causing relaxation of the trabecular meshwork [9, 10]. Thus, oestradiol in particular, increases the nitric oxide and prostacyclin vasodialative action, as well as reduces the response capacity of unstraited musculature to endothelin 1 [11].
Additionally, there is ample evidence that progesterone has the properties which are akin to those of a glucocorticoid antagonist [12]. Glucocorticoids are known to elevate intraocular pressure [12,13]. Hence, normal levels of progesterone may thwart the ocular hypertensive effect of endogenous glucocorticoids by competing for the receptor binding site. Such receptors have indeed been localized in human trabecular meshwork cells [14], which are capable of binding both glucocorticoids and progesterone. Post menopausally, with low levels of progesterone, this IOP lowering effect may be lost, thereby resulting in higher values of IOP. Furthermore, hormone replacement therapy has been found to lower intraocular pressure [15].
Apart from sex differences, IOP is also influenced by parameters like age, height and weight. Qureshi et al., reported that IOP positively correlated with age in a Pakistani population and more so, in females [16]. Mori K et al., observed that in Japanese females, IOP was increased in those who were aged upto 60 years, but that this longitudinal change in IOP was more strongly influenced by changes in weight and variations in BP than actual aging [2]. We also noticed higher BMIs in postmenopausal women, which were suggestive of increases in weight due to fat deposition. So, our findings are in consonance with those of Mori K et al. As there was no significant difference in the BP in present study, it is possible that changes in IOP were attributable to an increase in BMI.
A study which was done in Korean population observed that the mean IOP increased proportionally with degree of obesity in both males and females [17]. Similar findings were also reported by Barbados and the Beaver Dam Eye studies [18,19], where a large body size, as measured by BMI, was associated with an increasing IOP. Danish Zafar et al, also observed a positive correlation between BMI and IOP in both males and females among a Pakistani population [20]. Thus, influence of BMI on IOP transcends all ethnic and regional barriers.
On correlation, BMI was found to be correlated positively with IOP in both groups. However, this correlation was found to be significant only in case of postmenopausal women. This may be because of the dual effect of BMI, as well as reduced sex hormonal levels in post menopausal women. The probable reason that has been suggested is that the orbital pressure from excess fat may cause a rise in episcleral venous pressure and a decreased outflow facility [19]. Obesity also tends to increase blood viscosity via increased red cell counts, haemoglobin, and hematocrit, thus increasing outflow resistance of episcleral vein [21, 22].
Conclusion
This study was an attempt which was made to decipher the role of menopause and BMI on IOP in south Indian females. It showed that oestrogen and progesterone offered protection from raised IOPs and that their withdrawal postmenopausally, removed this protective veil. Further, IOP exhibited a positive correlation with BMI, especially in a postmenopausal, sex hormone starved milieu. This knowledge could be extrapolated and it could be used as a tool for screening disorders which are related to IOP, such as glaucoma. Thus, oestrogen replacement and BMI regulation in the postmenopausal period would offer protection against raised IOPs.
Limitations
The major limitation of the present study was that the relative contribution of BMI and/or reduced sex hormones for the observed higher IOPs postmenopausally, could not be apportioned. Hence, future studies which explore the relative contributions of these factors are warranted.
Values are expressed as Mean + Standard deviation, *significant
*Significant