Micronutrients are nutrients which are required by humans and other living beings throughout life in small quantities, to orchestrate a whole range of physiological functions, which the organism itself cannot produce. It includes dietary minerals in amounts which are generally less than 100 milligrams/day as opposed to the macro minerals which are required in larger quantities [1]. The trace elements include at least iron, cobalt, chromium, copper, iodine, manganese, selenium, zinc and molybdenum. The micronutrients also include vitamins, which are organic compounds which are required as nutrients in tiny amounts by an organism [2].
Copper is an essential nutrient in humans and it acts as a critical cofactor when it is incorporated into specific cupro-enzymes that catalyze the electron transfer reactions which are required for cellular respiration, iron oxidation, pigment formation, neurotransmitter biosynthesis, antioxidant defense, peptide amidation, and connective tissue formation [3]. Both the deficiency and the excess of copper are associated with specific clinical manifestations [4]. Copper deficiency causes an accumulation of the arterial lipid peroxides; however, with oxidative damage to the membranes or the macromolecules [5]. The copper intake in invivo studies has shown both pro-oxidant and antioxidant effects.
Increased concentrations of plasma copper were observed in diabetic patients with chronic complications or macro vascular diseases [6]. On the other hand, copper supplementation exerted beneficial effects in diabetic rats by reducing the glucose levels [7]. In contrast, epidemiological studies showed that higher serum copper concentrations may promote coronary artery diseases [8,9].
The lipid profile includes cholesterol, triglycerides, High – Density Lipoprotein (HDL), Very Low – Density Lipoprotein (VLDL), and Low – Density Lipoprotein (LDL). Though cholesterol is necessary for life, it is also associated with atherosclerosis. The triglycerides (TG) may be elevated in hypothyroidism, diabetes, chronic liver and kidney diseases, and pancreatitis. LDL (Low-Density Lipoprotein) cholesterol binds to the liver receptors and it tends to control the formation of cholesterol. Very Low Density Lipoprotein (VLDL) is a type of lipoprotein that helps in carrying the triglycerides to the liver and to other parts of the body. The lipid profile is assessed to identify hyperlipidaemia, many forms of which are recognised as the risk factors for cardiovascular disease and rarely, for pancreatitis. A total cholesterol reading is used to assess an individual’s risk for heart disease, however, it should not be relied upon as the only indicator because,the individual components that make up the total cholesterol reading (LDL, HDL, and VLDL) are also important for measuring the risk.While a high total cholesterol level may give an indication that that there is a problem with the cholesterol levels, the components that make up total cholesterol should also be measured.
Oxidative damage to the unsaturated lipids is a well established general mechanism for the oxidative stressm ediated cellular injuries. The occurrence of a free radical induced lipid peroxidation causes considerable changes in the cell membrane. Also, the deficiency of the micronutrients leads to an increase in the production of free radicals. Thus, the definitive role of copper in the oxidative processes is still a matter of debate. Furthermore, experimental and epidemiological data regarding the possible role of copper on the metabolic abnormalities and atherogenesis are conflicting. Hence, the present study was undertaken to evaluate the status of the micronutrients, oxidative stress, and the lipid profile in healthy adults.
Material and Methods
The present study was conducted in the Central Research Laboratory of the Nitte University after getting the institutional ethical clearance. Informed written consents were obtained from all the participants. This study includeda total of 200 subjects who were in the age group of 20-60 years, who reported to the outpatients department. Healthy, non obese individuals who were not suffering from any organic disorders, who belonged to both the sexes were included in the study. The subjects with any systemic disorders, diseases which required specific dietary recommendations like cardiovascular diseases and diabetes, the subjects with dyslipidaemia, chronic liver/bowel, or kidney diseases and the subjects who were on vitamin/micronutrient supplementations or drugs were excluded from the study.
The subjects were grouped as males who were in the age group of 20-40 years (Group-I), females who were in the age group of 20-40 years (Group-II),males who were in the age group of 40-60 years (Group-III) and females who were in the age group of 40-60 years (Group-IV). About 5ml of venous blood was withdrawn from all the subjects and it was transferred to a plain bottle. After 30 mins, the serum was collected and it was used for the biochemical analysis.
Estimation of copper and zinc by using an atomic absorption spectrophotometer
serum was taken, nitric acid was added in the ratio of 1:2 and it was kept over a heating plate till colourless fumes appeared.Then, the volume was made upto 10ml by adding mili Q water or deionised water. Copper and zinc were estimated by using an atomic absorption spectrophotometer. The radiation fluxwithout a sample and with a sample in the atomiser were measured by using a detector, and the ratio between the two values (the absorbance) was converted to analyse the concentration or the mass by using Beer-Lambert’s Law.
Estimation of the total antioxidant status by the phosphomolybdenum method
Exactly 100μl of the sample (serum or saliva) was pipetted out into a clean test tube and 100μl of 5%TCA was added to it to precipitate out the proteins in the sample. The mixture was then allowed to stand for about 5 min. and it was centrifuged. 100μl of the clear supernatant was transferred into a clean test tube,1 ml of the TAC reagent was added to it and the mixture was then incubated in a water bath at 90°C for 90 minutes. A blank was also maintained simultaneously by substituting 100μl of water instead of the sample, in the reaction mixture. Following the incubation, the reaction mixture was cooled and the optical density of the greenish to bluish colour which was formed was read at 695 nm against the blank.
Estimation of MDA by the Malondialdehyde Method
To the diluted sample, 1ml of the TCA-TBA-HCL reagent was added. The sample was kept in a boiling waterbath for 15 min.The reaction mixture was cooled and centrifuged. The supernatant was taken and the optical density of the pink colour which was formed was read at 535nm.
Estimation of total cholesterol by the CHOD-POD Method
The total cholesterol was determined by an enzymatic hydrolysis and oxidation method. The colourimetric indicator was quinoneimine which was generated from 4 – aminoantipyrine and phenol by hydrogen peroxide under the catalytic action of peroxidase (Trinder’s reaction). 3 test tubes, blank (B), test (T) and standard (S) were taken.10μl of the serum was added to the test tube.10μl of standard was added to the standard tube.1000μl ofreagent was added to each test tube, the tubes were mixed well and they were incubated for 10 min at 370 C.The absorbance was taken at 505nm against the reagent blank spectrophotometrically.
Estimation of HDL-cholesterol by the precipitation end point Method
Sample of 200μl and the precipitating reagent (R – 2) were pipetted into a centrifuge tube and they were mixed well and centrifuged for 10 min. The supernatant was separated and cholesterol was determined. 3 test tubes, blank (B), test (T) and standard (S) were taken. 50μl of the sample (serum) was added to ‘T’ and 50ul of the standard was added to ‘S’.1000μl of reagent-1 was added to each test tube, the tubes were mixed well and they were incubated for 10 min. at 370 C.The absorbance was taken at 505nm against the green filter.
Calculation
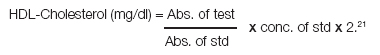
Where, Factor 2 was applied to account for the dilution of the sample in the precipitation procedure.
Estimation of total triglycerides by the GPO-POD method
The total triglycerides were determined after an enzymatic splitting with lipoprotein lipase. The indicator was quinoneimine,which was generated from 4 – chlorophenol by hydrogen peroxide under the catalytic action of peroxidase. Three test tubes, Blank (B), Test (T) and Standard (S) were taken.10μl of the sample (serum) was added to T and 10ul of the standard was added to S.1000μl of the reagent was added to each test tube, the tubes were mixed well and they were incubated for 10 min at 370 C.The absorbance was read at 505nm against the reagent blank.
Calculation
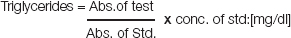
LDL was calculated by the formula, LDL=TC-(HDL+VLDL)
VLDL was calculated by the formula, VLDL=TG/5.
Statistical Analysis
The data was represented as mean ± Standard Deviation. The data was analysed by one way ANOVA, followed by Pearson’s correlation.
Results
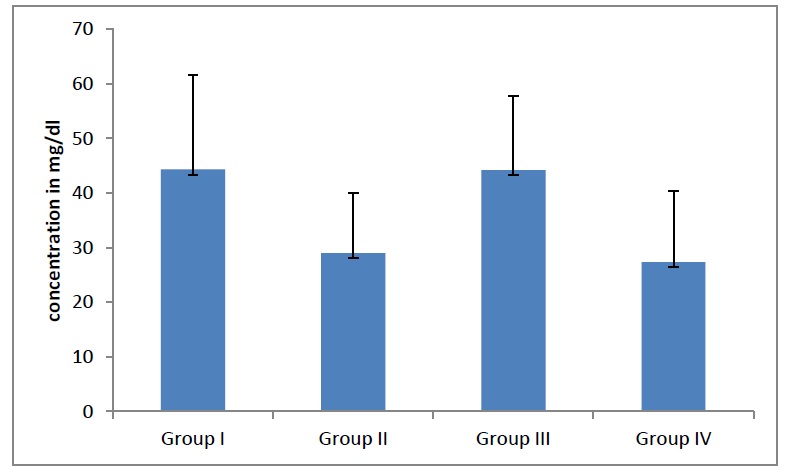
In the present study, when the serum copper levels in the adults were increased significantly (p< 0.0001), [Table/Fig- 1]. When a further correlation within the groups was done, the copper levels were found to be statistically significant (p<0.05).The serum zinc levels in males and females were increased significantly (p= 0.01), [Table/Fig- 2]. When a further correlation within the groups was done, the zinc levels were found to be statistically significant (p <0.05). When the MDA and the TAS levels of the adults were compared, it showed that in all groups of adults, they had declined significantly (p <0.0001), [Table/Fig- 3,Table/Fig- 4] as age advances.
Average concentration of copper in different groups. The serum copper levels in different groups of adults were increased significantly (p< 0.0001)
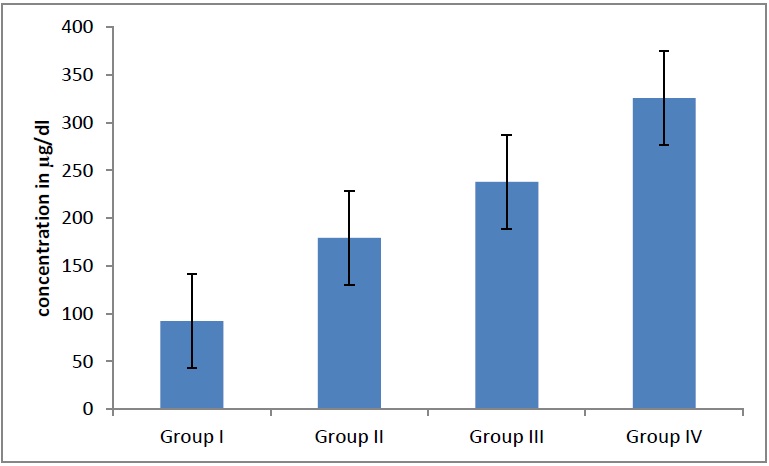
Average concentration of Zinc in different groups. The serum zinc levels in males and females were increased significantly (p = 0.01)
Average Total antioxidant status in different groups. The TAS levels in different groups of adults were declined significantly (p<0.0001) as age advances
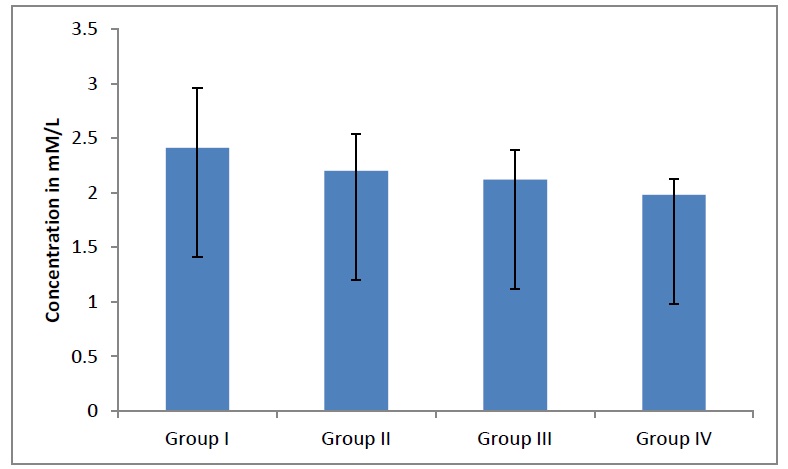
Average MDA level in different groups. The MDA levels decreases significantly (p<0.0001) in adult males as age advanced whereas, it is increased significantly (p <0.0001) in adult females as age advanced
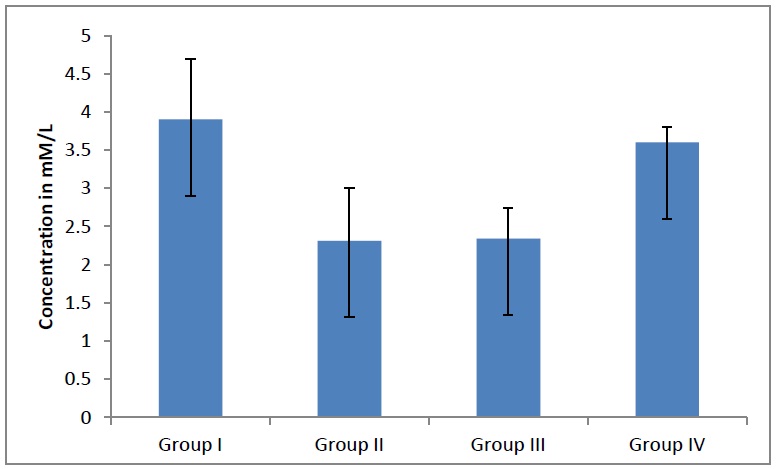
The total cholesterol levels decreased significantly (p<0.0001), [Table/Fig- 5] in the adult females as the age advanced, whereas theyincreased significantly (p <0.001) in the adult males as the age advanced. When a further correlation within the groups was done, the total cholesterol levels were found to be statistically significant. When the total triglyceride levels in the adults were compared, they showed a significant decline (p<0.04), [Table/Fig- 6] in the adult males and females as the age advanced. When a further correlation within the groups was done, the TG levels were found to be statistically significant (p= 0.04). When the HDL levels in the adults were compared, they showed a statistically significant decline (p<0.05), [Table/Fig- 7]. When a further correlation within the groups was done, the HDL levels were found to be statistically significant.
Average total cholesterol levels in different groups. The total cholesterol levels decreases significantly (p<0.0001) in adult females as age advanced whereas, it is increased significantly (p<0.001) pin adult males as age advanced
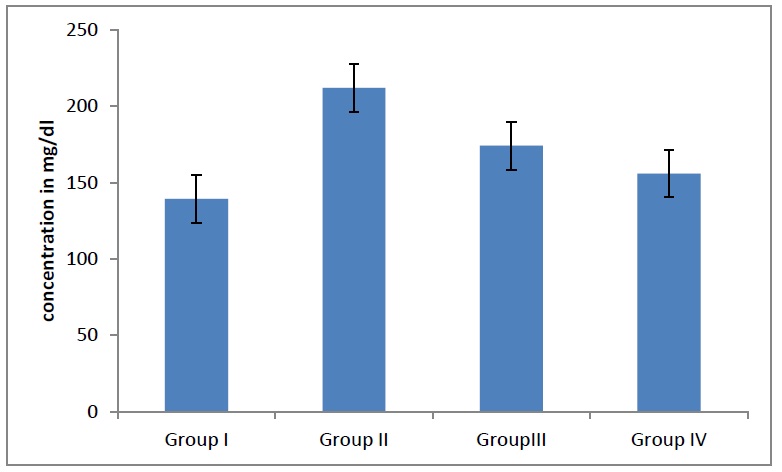
Average triglyceride levels in different groups. The triglyceride levels decreases significantly (p<0.04) in adult males and females as age advanced
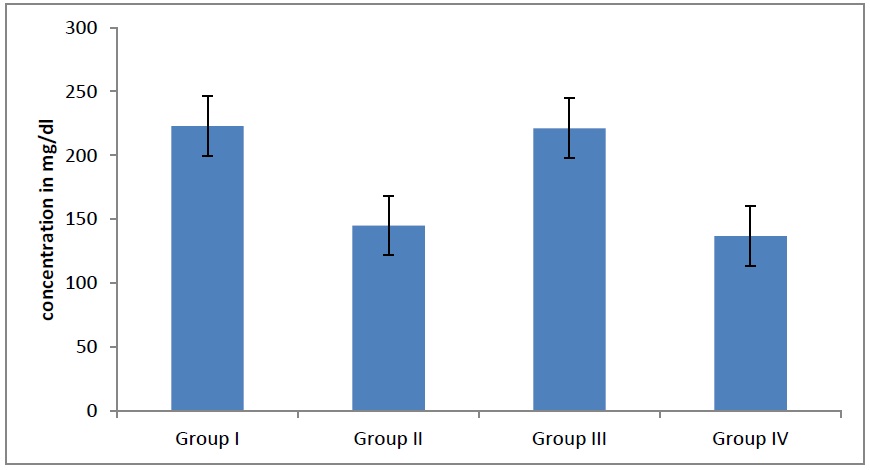
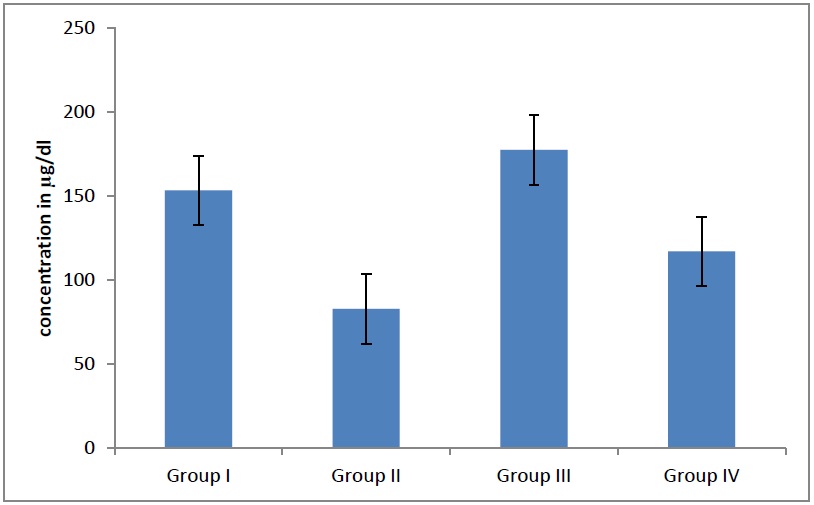
Average HDL levels in different groups. The HDL levels decreases significantly (p<0.05) in adult males and females as age advanced
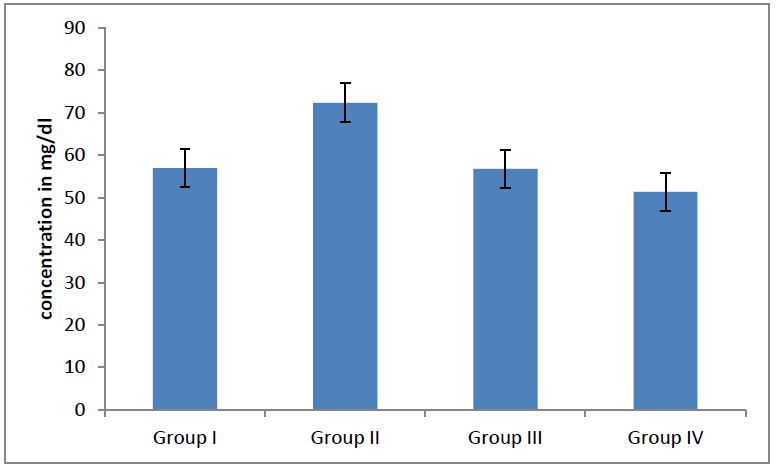
When the LDL levels in the adults were compared, they showed a statistically significantincrease (p=0.04), [Table/Fig- 8] as the age advanced. When a further correlation within the groups was done, the LDL levels were found to be statistically significant. When the VLDL levels in the adults were compared, they showed a statistically significantdecline (p = 0.04), [Table/Fig- 9] as the age advanced. When a further correlation within the groups was done, the VLDL levels were found to be statistically significant.
Average LDL levels in different groups. When LDL levels in adults were compared showed statistically significant (p= 0.04) increase as age advances
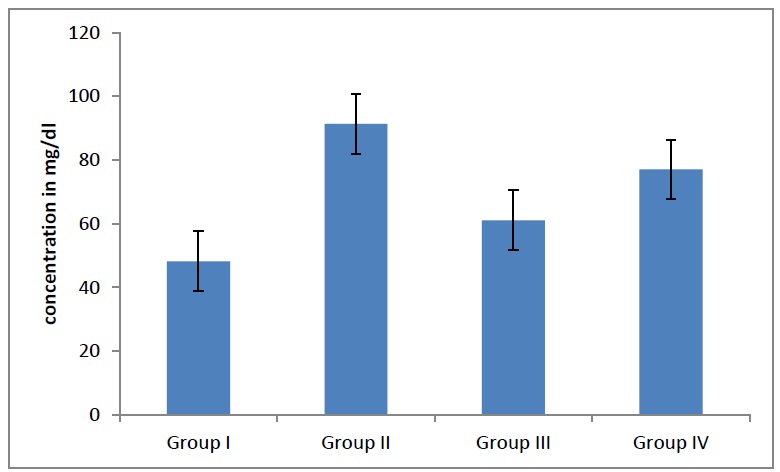
Average VLDL levels in different groups When VLDL levels in adults were compared showed statistically significant decline (p= 0.04) as age advances
Discussion
In the present study, the copper levels in the different groups wereassociated with different parameters like TAS, MDA and the lipid profile. Copper is both prooxidant and antioxidant. Any changes in the oxidative stress can lead toan oxidative damage in the tissues. An increase in the copper levels increases the oxidative stress, whereas a decrease in the copper levels decreases the oxidative stress.
Oxidative stress is directly associated with copper. The association of copper with zinc, serum total cholesterol, HDL-cholesterol and the triglyceride concentrations were studied in adults. The serum copper: zinc ratios were associated with lower HDL-cholesterol concentrations. The higher copper concentrations were associated with higher cholesterol concentrations. The higher serum copper concentrations were associated with higher triglyceride concentrations.
Copper is a main micronutrient that is required by the human body. The association of copper with the zinc levels in group I showed a decrease in the copper levels with an increase in the zinc levels. The Groups II, III and IV showed an increase in the copper levels with a decrease in the zinc levels.A significant direct association between serum copper and zinc was seen. It might be hypothesized that the copper-induced oxidative stress might determine an inflammatory response and that inflammation may represent the result of the copper damage instead of the cause ofthe increase in the copper concentrations.
When the copper levels decreased, the TAS levels also decreased, where as the MDA levels had increased. Hence, the oxidative stress is directly associated with copper and any changes in the copper levels lead to a variation in the oxidative stress. The MDA level is inversely associated with the copper level.
The association of copper with the lipid profile is statistically significant. High levels of zinc can reduce the copper absorption. Taking zinc in high doses can cause significant copper deficiency and anaemia, a condition in which the blood cannot carry enough oxygen. Zinc has a stabilising effect on the membranes, possibly by displacing the bound transition metal ions and thereby preventing the peroxidation of the membrane lipids. Any imbalance in the copper level will lead to its deficiency, because copper is mainly associated with oxidative stress and the metabolic variables. Zinc is required in over 200 enzymes and so its deficiency is likely to affect a number of different systems. Zinc, like copper, is a component of CuZn-superoxide dismutase (CuZnSOD), which is an important anti-oxidant enzyme. The deficiencies of copper rather than zinc [10] can affect the activity of this enzyme [11]. A zinc deficiency has been found to cause oxidative damage to the proteins and thelipids [12].
The antioxidant micronutrients are important, not only for the oxidative and tissue damage, but also in preventing increased cytokine production, which is a result of a prolonged activation of the immune response. It is important to have an adequate antioxidant intake from both the diet and supplementation if needed, and it can be a valuable adjunct in the treatment of chronic inflammatory disorders.
High serum levels of copper are associated with an increased cardiovascular risk, but the significance of these findings is still unclear because of the effect of ceruloplasmin and inflammatory conditions on the serum copper levels. While unbound copper and ceruloplasmin can lead to LDL oxidation, there is little evidence that the increase of copper in the dietary intake could increase the oxidative stress in the human organism [13].
Glucose, total and LDL – cholesterol and TAS decreased, whereas the hs – CRP concentrations increased from the lowest to the highest tertileof the serum copper concentration. The serum copper concentrations were inversely associated with glucose, total cholesterol, LDL – cholesterol, and TAS and they were directly associated with the CRP concentration. A copper deficiency is associated with an unfavourable metabolic pattern, but a copper supplementation might not be recommended in view of its association with inflammation and the markers of oxidative stress [14,15].
Conclusions
There was a significant correlation between micronutrient status and the oxidative stress. Since, there was a significant increase in the serum copper and zinc levels in adults,a corresponding decrease in the total triglyceride levels and an increase in the LDL levels in the present study, we conclude that the micronutrient status will have a direct correlation with the oxidative status as the age advances.