Perforator Plus Fasciocutaneous Flaps in the Reconstruction of Post-Burn Flexion Contractures of the Knee Joint
Madhumita Gupta1, Ashwin A. Pai2, Ravi R. Setty3, Raghavendra Sawarappa4, Bijay Kumar Majumdar5, Tibar Banerjee6, Aditya Kanoi7, Abhimanyu Bhattacharya8
1 Resident, Department of Plastic and Reconstructive Surgery, Ipgme & R, Kolkata, India.
2 Resident, Department of Plastic and Reconstructive Surgery, Ipgme & R, Kolkata, India.
3 Resident, Department of Plastic and Reconstructive Surgery, Ipgme & R, Kolkata, India.
4 Resident, Department of Plastic and Reconstructive Surgery, Ipgme & R, Kolkata, India.
5 Head of the Department, Department of Plastic and Reconstructive Surgery, Ipgme&R, Kolkata, India.
6 Assistant Professor, Department of Plastic and Reconstructive Surgery, Ipgme & R, Kolkata, India.
7 Resident, Department of Plastic and Reconstructive Surgery, Ipgme & R, Kolkata, India.
8 Resident, Department of Anesthesiology, Ipgme & R, Kolkata, India.
NAME, ADRESS, E-MAIL ID OF THE CORRESPONDING AUTHOR: Dr. Madhumita Gupta, Address: 357/1/1, Prince Anwar Shah Road, Kolkata-700068, West Bengal, India.
Phone: +91 9836548574
E-mail: docmadhu82@gmail.com
Background: A post-burn flexion contracture of the knee joint is a disabling condition which interferes with an upright posture and a bipedal locomotion. Islanded perforator flaps have been used to resurface the tissue defect which is produced as a result of the contracture release. Despite their various advantages, they are limited by an increased tendency to undergo venous congestion. Perforator-plus flaps can be used to overcome this limitation, while retaining the merits of the islanded perforator flaps.
Methods: Ninteen patients with post flame burn flexion contractures of the knee joints underwent surgical releases and coverages by various local fasciocutaneous perforator-plus flaps. The patients were followed up for 6 months and the various aspects of the functional and the aesthetic rehabilitations were assessed.
Results: All the local fasciocutaneous perforator-plus flaps resurfaced the tissue defect over popliteal fossa with good colour and texture match and maintenance of the contour. None of the flaps had any significant early or delayed complications (which included venous congestions) which necessitated reoperations. All the patients were satisfied with the functional and aesthetic outcomes.
Conclusion: Local fasciocutaneous perforator-plus flaps can be considered as one of the primary treatment modalities for the surgical release and reconstruction of post burn flexion contractures of the knee.
Perforator-plus flap, Fasciocutaneous flap, Peninsular flap, Post-burn contracture, Knee joint
INTRODUCTION
With a steady decline in the mortality of burn patients over the past six to seven decades, addressing the issue of disabling post burn sequelae has become more vital, for adequately rehabilitating the victim in society [1]. A burn patient who receives the best of treatment, which includes an early physiotherapy, an adequate splintage in the position of function and an early debridement with skin grafting, is expected to heal without any significant contractures. However, post burn contractures are distressingly common in India and other developing nations. The incidence of burn cases has been estimated to range from 60-70 lakhs annually in India [2]. In the emergency setting, most of the cases are treated in ill-equipped peripheral units by inadequately trained staff. This, in turn, gives rise to the enormous burden of post-burn contractures and deformities, which have to be dealt with, in the comparatively few tertiary care centres [2].
The post burn scar contractures over the major joints of the body are characterized by the presence of a single-celled, highly fragile neoepithelium, which may be associated with a long-standing shortening of the underlying musculotendinous units and the neurovascular structures, as well as tightened joint capsules and ligaments. The knee is the largest joint of the human body and it is involved in the maintenance of an upright posture and a bipedal locomotion. An adequate incisional or excisional release of the flexion contracture of this joint leaves large skin defects and it often exposes bowstringed hamstring tendons, major vessels and nerves in the popliteal fossa, which require soft tissue coverage. Those cases which are resurfaced with skin graft need a prolonged splintage and a rigorous physiotherapy for a minimum period of 6 months [3,4]. This leads to a poor patient compliance. Therefore, the rates of recontracture is more, which needs re-operations and this increases the patients’ morbidity and the burden on the already stressed infrastructure .These shortcomings can be overcome by the use of flaps. However, the lower limb has been known to be a site of a less reliable vascularity, poor wound healing and scarcity of flaps. Before the advent of free flaps, only few reconstructive options were present, like conventional local flaps and cross-leg flaps, which required immobilization for weeks [5]. Several muscle or musculocutaneous flaps have been used to repair the soft tissue defects around the knee [6–8]. However, they are bulky, they cause varying degrees of functional loss to an already crippled extremity and they also cause poor aesthesis of the donor site. The random cutaneous and conventional fasciocutaneous flaps are simple and reliable and they provide a durable pliable coverage with a better donor site aesthesis and less morbidity. However, they are restricted by a limited length : breadth ratio, a need of delays and limited mobility and reach. The delicate balance between the flap vascularity and the reach in the lower limb was somewhat achieved with the advent of islanded perforator flaps [9,10]. It was recognized that the skin which overlay the muscles could be reliably transferred separately from the muscles as an islanded flap, based only on the dissected musculocutaneous perforators [9,10]. The donor site has minimal functional compromise and better aesthesis. They provide a thin, pliable coverage with tissues of similar colour and texture and an immense freedom of movement and they maintain the functional integrity of the muscles and the nerves, while safeguarding the main vascular trunks. But an important pitfall of such flaps is venous compromise. This is because the thin walled veins with lower vessel wall elasticities and intraluminal pressures is more sensitive to the torsional forces than the perforator artery during the flap transfer and inset [11,12]. The concept of the perforator-plus flaps aims to combat this pitfall. This new method involves raising of the peninsular fasciocutaneous flaps, thus including and retaining one or more perforators in their base [13,14]. This provides a dual blood supply through the perforator and the subdermal plexus, as well as it reinforces the venous drainage. The pedicle may be narrowed by back cuts to facilitate the mobility and a tension free inset [14]. It has been determined that the perforator-plus fasciocutaneous flaps can be safely used in the knee and the upper and middle thirds of the leg [15,16]. Moreover, most of the cases of post burn contractures of the knee have limited loco-regional unscarred tissues which increase the chance of a flap failure. This is combatted by including a perforator in the flap base [17,18].
The aim of this study was to evaluate the applicability of perforator-plus flaps in the reconstruction of post burn flexion contractures of the knee joint.
METHODS
During the period from August 2010 to March 2012 , 19 patients with post-flame burn flexion contractures of the knee joint were operated in the Department of Plastic and Reconstructive Surgery of our institution. A clearance was obtained from the institutional ethical committee and written informed consents were obtained from all the patients. All the patients were managed by a complete incisional or excisional release of the contracture, followed by coverage with local perforator-plus flaps. The patients were advised to quit smoking at least 2 weeks prior to their surgeries. The degree of contracture was assessed with a goniometer and the extent of the defect which would be produced was approximately estimated by comparing the degree of contracture with the normal side (or limb of another subject of a similar stature and build, in case of a bilateral involvement). The type of fasciocutaneous flap which had to be raised was then planned, after considering the location of the defect and the quality of the surrounding tissue. Any perforator which supplied the proposed flap territory was marked with the help of a hand-held Doppler with an 8 Hz probe. It was considered as preferable but not mandatory to identify the perforator pre-operatively. The approximate dimensions of the flap were determined by planning in reverse . The length of the flap from the identified perforator (which marked the location of the base) was equal to the distance of the distal-most margin of the defect from that perforator, plus one centimetre, to allow a tension-free inset. After the induction of anaesthesia, the contracture was released completely, without any damage to the underlying vital structures, under a tourniquet control. The final planning in reverse was done with a piece of lint and the modified outline was marked out. The flap was then raised by the combination of a sharp and a blunt dissection, thus preserving the supra and the subfascial plexuses. The perforator(s) at the flap base with a strong intra-operative Doppler signal(s) and a visible pulsation(s) was/were preserved. They were dissected, retaining a cuff of the areolar tissue around, in order to ensure the flap mobility and reach. The skin pedicle was narrowed with back cuts if it was needed, to facilitate the reach and inset without significant dog-ears.The flap inset was then accomplished without any tension on the pedicle. The placement of a suction drain under the flap was considered as optional. The donor site was covered by a split-thickness skin graft.The flap was covered by a light dressing and the limb was kept elevated post-operatively. Prophylactic antibiotics were given for 5 days. No special post -operative drugs or monitoring techniques were needed. The dressing over the graft was removed on the 5th post-operative day and the sutures were removed on the 10th day. No post-operative splinting was applied. The limb mobilization was began after 1 week and gradual weight-bearing started two weeks post-operatively . The patient was followed up two weekly in the first month after discharge and monthly thereafter, for 6 months.
RESULTS
A total of 19 patients of post-flame burn flexion contractures of the knee joints were operated between August 2010 and March 2012 [Table/Fig-1 and 2]. Seven cases had involvement of the right side, 11 had involvement of the left side and 1 case had a bilateral affection [Table/Fig-3 and 4]. All the patients had flexion contractures of the knee joints, with a hypertrophic scarring and a hypopigmentation. Non-healing ulcers were present in 9 cases. Of the 19 patients in the study, 8 were males. The age range of the study population was 5-56 years ( mean -29.6 years ). The duration after the burn injury ranged from 6 to 15 months (mean -9.15 months). The degree of the flexion contracture ranged from 10-120 degrees, with an average of 52.75 degrees. All the cases were operated under spinal or general anaesthesia and they underwent incisional or an excisional complete releases in the prone position and coverage with local fasciocutaneous perforator-plus flaps. All the cases included a single perforator at the base. The pre-operative Doppler assessment of the perforator was done in all except 2 cases, where they were identified intra-operatively. The source vessel was the peroneal artery [Table/Fig-5] in 7 cases, it was the superior medial genicular artery [Table/Fig-6] in 5, it was the superior lateral genicular artery in 3, it was the lateral sural [Table/Fig-7] and the medial sural arteries in 1 case each and it was the saphenous/inferior [Table/Fig-3 and 4] medial genicular artery in 3. The flap dimensions ranged from 11.5x6 to 19x11 cm. All the flaps adequately covered the tissue defects over the flexural aspects of the knee joints. They were inset in a tension-free manner, with the maintenance of a good contour, colour and texture match. The operating time spanned from 75 to 105 mins (mean- 88.75 mins). None of the cases needed splintage and all the limbs were mobilized on the 7th post-operative day. The total duration of the hospital stay ranged from 10-17 days (average 12.1 days). Wound infection and dehiscence of the insetting sutures occurred in 2 cases. Only one case of the superior medial genicular artery perforator-plus flap developed marginal tip necrosis. There was no venous congestion in any flap. All the local complications were managed under local anaesthesia with debridement and by secondary suturing or healing by secondary intention. There were minor graft losses over the donor sites in 2 cases, which were treated conservatively. Apart from a single case of a superior medial genicular perforator-plus flap with paresthaesia over the antero-medial thigh, no other case had any significant evidence of a collateral damage like distal limb oedema, sensory loss or muscle weakness. All the patients were followed up for 6 months, except one, who was lost to follow up after 2 months. 8 cases had some evidence of a hyperpigmentation, an induration and/or a hypertrophic scarring over the grafted areas. However, since none of the patients had any grafts over the flexural aspects of the knees, there were no incidences of recontractures and hence, there was no need of reoperations. The flaps provided a long-term stable pliable coverage over the popliteal fossa with a good colour and texture match and maintenance of the contour.The flaps were not bulky and only one patient desired revision surgery for a dog-ear correction, which was accomplished on an outpatients basis. All the patients, at 6 months follow-up, were satisfied with the function and aesthesis of the limbs. They were able to maintain an unassisted symmetrical upright posture and a bipedal locomotion and they could squat without difficulty. Apart from 3 patients, the rest were able to sit cross-legged on the floor without much difficulty.
Patient Profile and Intra-Operative Details
SMGA: superior medial genicular artery, SLGA: superior lateral genicular artery
| Serial No. | Age (Years) | Sex | Side | Duration Since Burn (Month) | Degree Of Contracture | Source Vessel | Flap Dimension (Cm) | Operating Time (Mins) |
|---|
| 1 | 13 | M | left | 9 | 120 | SMGA | 19x11 | 95 |
| 2 | 5 | M | left | 6.5 | 100 | SLGA | 17x7.5 | 80 |
| 3 | 35 | F | right | 8 | 50 | peroneal | 14x6.5 | 80 |
| 4 | 27 | F | right | 6 | 10 | lat. sural | 17.5x8.5 | 75 |
| 5 | 56 | M | left | 8 | 45 | peroneal | 15x7 | 95 |
| 6 | 19 | M | left | 12 | 55 | SLGA | 14x7 | 100 |
| 7 | 26 | F | left | 11 | 85 | SMGA | 18x10.5 | 100 |
| 8 | 34 | F | right | 8 | 70 | SMGA | 17x10 | 80 |
| 9 | 33 | F | left | 15 | 25 | peroneal | 12x6 | 85 |
| 10 | 37 | M | left | 10 | 20 | peroneal | 14x6 | 100 |
| 11 | 31 | F | right | 7 | 90 | peroneal | 16x7 | 95 |
| 12 | 23 | M | left | 9 | 35 | med. sural | 11.5x6 | 85 |
| 13 | 18 | F | left | 10 | 40 | SMGA | 13.5x7 | 90 |
| 14 | 22 | M | left | 11 | 30 | peroneal | 13x7 | 80 |
| 15 | 31 | F | left | 8.5 | 25 | saphenous | 11x6.5 | 75 |
| 16 | 34 | F | right | 8 | 85 | SLGA | 15x8 | 100 |
| 17 | 45 | M | right | 9 | 35 | saphenous | 11x6 | 80 |
| 18 | 37 | F | right | 13 | 70 | peroneal | 14x6.5 | 90 |
| 19 left | 36 | F | left | 7 | 30 | SMGA | 18x10 | 100 |
| 19 right | 36 | F | right | 7 | 35 | saphenous | 15x8 | 90 |
| Serial No. | Hospital Stay (Days) | Follow Up (Months) | Early Complications | Delayed Complications | Aesthetic Acceptability |
|---|
| 1 | 14 | 6 | | hypertrophic scar, dog ear correction | average |
| 2 | 10 | 2 | | | good |
| 3 | 11 | 6 | | | good |
| 4 | 10 | 6 | | | average |
| 5 | 12 | 6 | | hypertrophic scar | average |
| 6 | 11 | 6 | | | good |
| 7 | 17 | 6 | wound infection | hypertrophic scar | average |
| 8 | 11 | 6 | minor graft loss | paresthesia | average |
| 9 | 14 | 6 | | | average |
| 10 | 13 | 6 | | hypertrophic scar | average |
| 11 | 11 | 6 | | hypertrophic scar | average |
| 12 | 10 | 6 | | | good |
| 13 | 14 | 6 | minor graft loss | hypertrophic scar | average |
| 14 | 15 | 6 | wound infection | | good |
| 15 | 10 | 6 | | hypertrophic scar | average |
| 16 | 11 | 6 | | | good |
| 17 | 10 | 6 | | | good |
| 18 | 10 | 6 | | hypertrophic scar | average |
| 19 left | 16 | 6 | | | tip necrosis | average |
| 19 right | 16 | 6 | | | good |
Case no.19 (vide table 1): Bilateral contracture: Left-superior medial genicular artery perforator-plus flap, right-saphenous artery perforator-plus flap
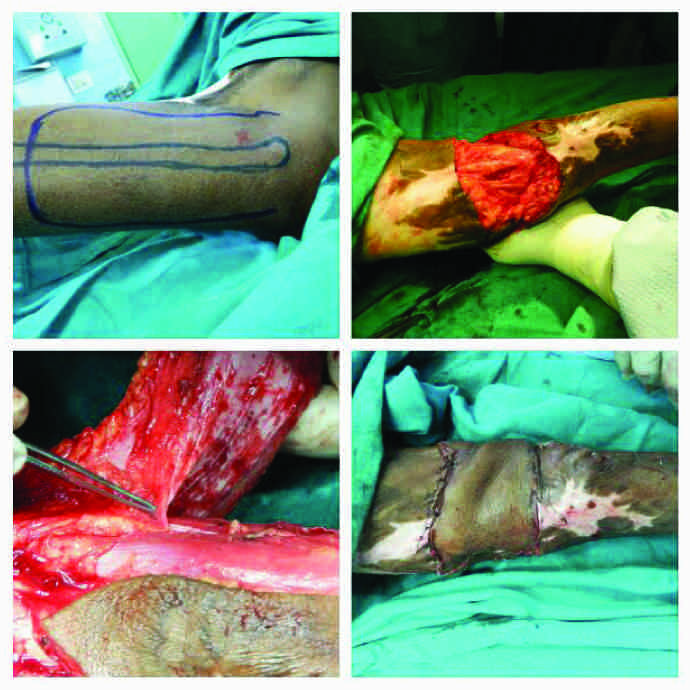
Case no. 19 post-operative day 10
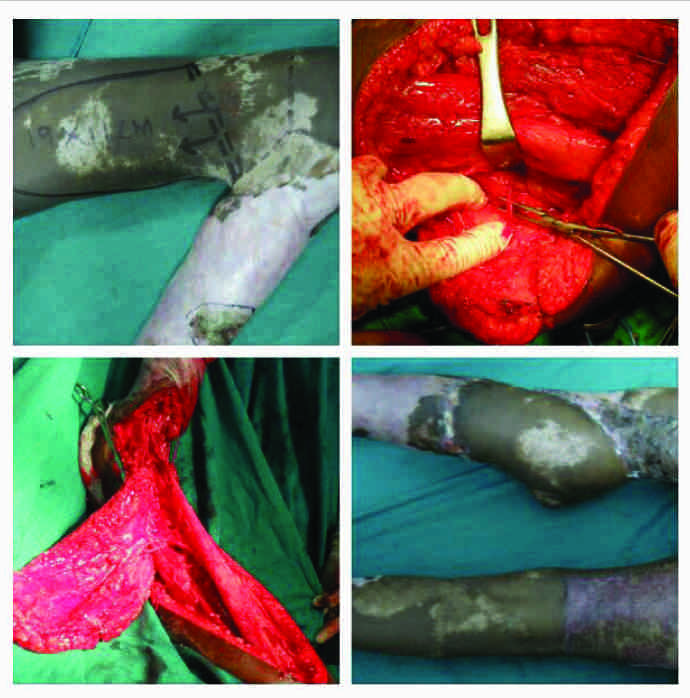
Case no. 3 (vide table1): Peroneal artery perforator-plus flap
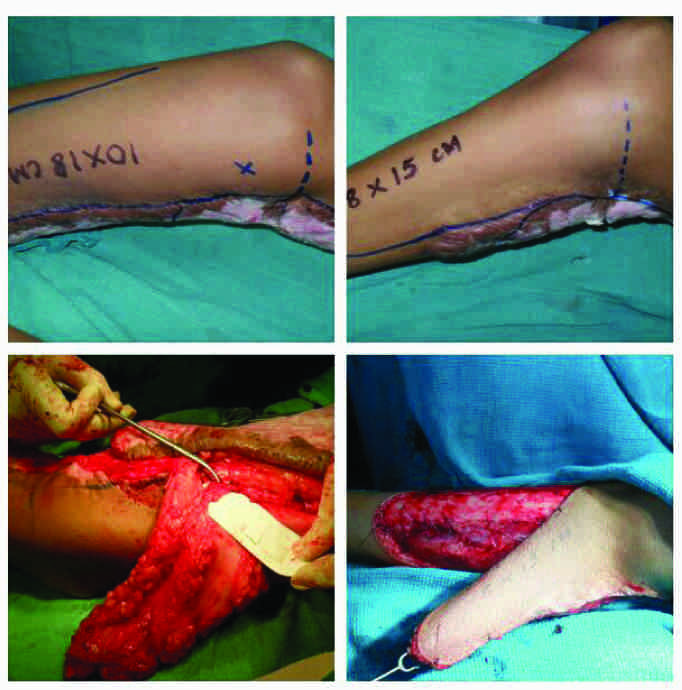
Case no. 1 (vide table 1): Superior medial genicular artery perforator-plus flap
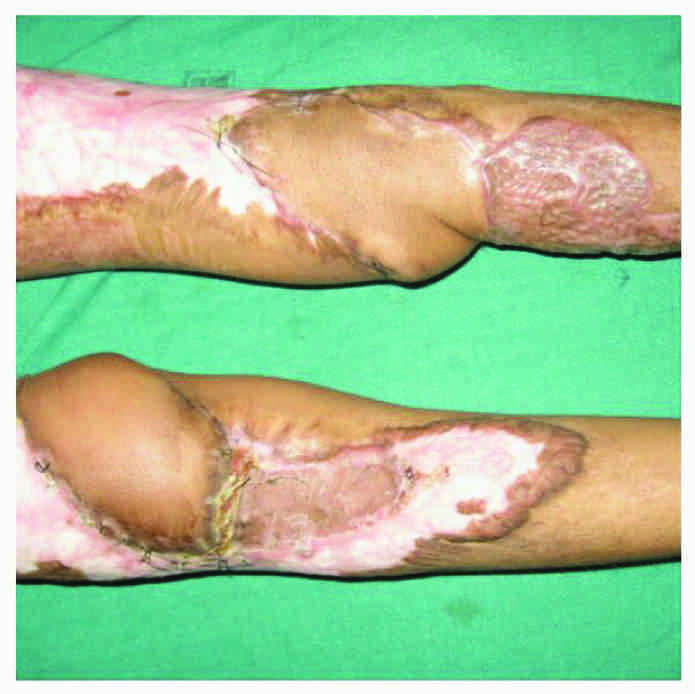
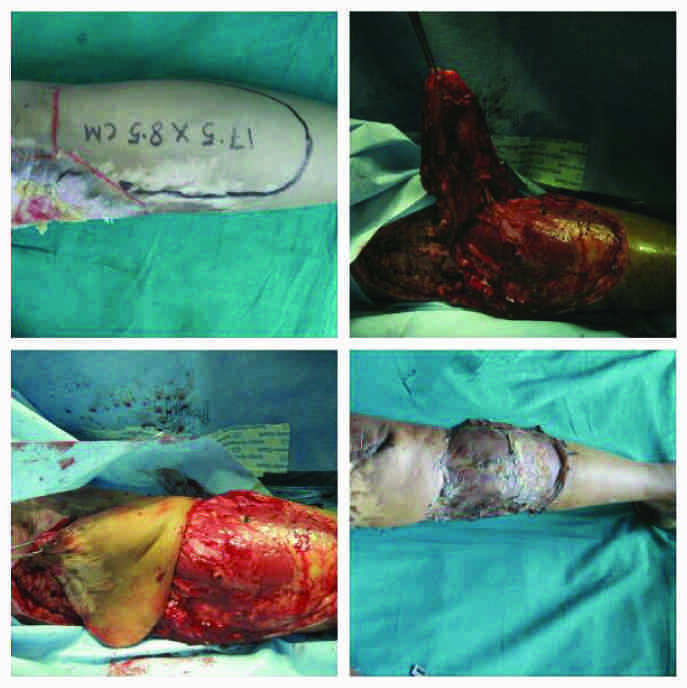
Case 4 (vide table 1): Lateral sural artery perforator-plus flap.
DISCUSSION
The contribution of perforators in the flap circulation was first suggested by Fujino [19]. Perforator flaps were later developed by Koshima [9,10] and Kroll and Rosenfield [20]. According to Taylor and Palmer’s [21] concept of the angiosome (1987), almost all the tissues of an angiosome can be harvested on one adequate perforator. Such perforators originate from one of the main axial vessels, they course through a muscle or septa, pierce the fascia and ramify in the suprafascial level within subcutaneous fat. The perforator-plus flaps were conceptualized and the nomenclature was coined in 2005 [13]. The terms, ‘perforator-plus’ and ‘perforator-sparing flaps’ have been used interchangeably for the same flap design [16]. In the classical rotation design, the moving tip is under tension if the donor defect is closed primarily. This can be prevented by designing oversized flaps or by making a back cut from the existing pivot point into the base. However, this might compromise the vascularity of the conventional flaps. The inclusion of a known or an identified perforator [16] in the base allows the greater freedom of a back cut, which shifts the pivot point closer to the defect and facilitates a tension free inset. This phenomenon is applicable to any design of a peninsular flap and it is the basis of a perforator-flap harvest [22].The peninsular design prevents kinking of the perforator vessels and it improves the venous outflow through the pedicle. The perforator-plus technique is being used in various conditions from lower limb trauma to pressure sores [15]. The evaluation of a series of different perforator-plus flaps for the reconstruction of post burn flexion contractures of the knee could not be found in the published literature till date. There is no conclusive evidence in the literature regarding the safe limit of harvest of the perforator or the perforator-plus flaps. However, it has been noted that there is a six-fold higher chance of an islanded perforator flap failure if the length is more than one-third of the total limb length [23]. It can be postulated that the larger dimensions of the perforator-plus flaps can be raised than the corresponding islanded perforator flaps, due to the dual blood supply. The knee joint has a rich vascular plexus around it, which gives rise to many prominent perforators and connects the femoral vessels which are above, with the popliteal and the tibial vessels which are below. An average of 93 perforators from 21 vascular territories supply the skin of the lower extremity. The mean diameter and the area which are supplied by one perforator are approximately 0.7 mm and 47 cm2 respectively [24]. Perforator-plus flaps may be designed from various aspects of the distal thigh and the proximal leg to resurface the tissue defect over the popliteal fossa which is created by the contracture release. The antero-lateral thigh flap is based on the perforators from the descending branch of the lateral circumflex femoral artery, which are clustered within a 3 cm radius of the midpoint of a line which joins the anterior superior iliac spine and the superolateral patella. The descending branch anastomoses with the superior lateral genicular artery, which allows a distally- based flap to be harvested, with the pivot point 3-10 cm above the knee joint [25]. The anteromedial thigh flap is based on a major perforator from the rectus femoris branch of the descending branch of the lateral circumflex femoral artery, which is present in 51% individuals. It has a musculocutaneous course through the rectus femoris in 34% cases, while the rest are septocutaneous. The perforator is located about 3.2 cm medial to the midpoint of the line which joins the anterior superior iliac spine with the superolateral patella [26]. The cutaneous perforator of the superior lateral genicular artery is located within a triangle which is bounded by the superior border of the lateral femoral condyle, the anterior border of the short head of the biceps femoris and the posterior border of the vastuslateralis [27]. It is located about 7.4 +2.77 cm above the lateral femoral condyle [28]. In 60% of the cases, it traverses the lateral intermuscular septum of the thigh, while in the rest, it passes through the biceps femoris [6]. The saphenous branch of the descending genicularatery originates 13 cm above the medial joint line and it courses along a line which joins the anterior superior iliac spine and the medial tibial condyle. Here, the vessel traverses deep into the sartorius and it gives septocutaneous perforators on either side of the muscle. The terminal branch of this artery anastomoses with the collateral vessels around the knee, like the inferior medial genicular artery, which forms the basis of the reverse flow distally based flaps [29]. The cutaneous perforators of the superior medial genicular artery are located in a small triangle which is bounded by the superior border of the medial femoral condyle, the anterior border of the adductor magnus and the posterior border of the vastusmedialis [30] . The medial and the lateral sural arteries originate from the popliteal artery. 1-5 medial sural artery perforators were detected in 100% cases in an Asian population. But the lateral sural artery perforators were absent in 57% of the cases. These perforators are located between 5 cm above and 17.5 cm below the popliteal crease, within a range of 0.5 to 4.5 cm from the midline raphe of the gastrocnemius [31]. The vascular axis of the posterior tibial artery lies along a line which is ~4.5 cm medial and parallel to the line which joins the tibial tuberosity and the midmalleolar point [32]. The septocutaneous perforators which arise from it are arranged in three clusters, the most proximal of which is 3.6-10.8 cm below the joint line [33]. The peroneal artery perforators [34] are situated along an oblique line, behind the posterior border of the fibula, along the intermuscular septum, between the soleus and the peroneus longus. Proximally, they are 0.25 cm away, while distally, they are 1.7 cm behind the posterior fibular margin. Based on this anatomic knowledge on the perforator distribution around the knee joint and the popliteal fossa, robust and pliable perforator-plus flaps can be harvested to cover the tissue defects, following a surgical release of the knee contracture. With the advent of freestyle perforator flaps [35] the applicability of the perforator-plus flaps can be further broadened with lesser restrictions on the flap axiality and design. However, the maximum safe dimension of the harvest of such flaps has to be ascertained.
[1]. Hawkins HK, Pereira CT, Pathophysiology of the burn scarTotal Burn Care 2007 3rd edPhiladelphiaSaunders Elsevier:608-19. [Google Scholar]
[2]. Hanumadass ML, Some thoughts on organization of delivery of burn care in IndiaIndian J Burns 2003 11:18-20. [Google Scholar]
[3]. Su CW, Alizadeh K, Boddie A, Lee RC, The problem scarClin Plast Surg 1998 25:451-65. [Google Scholar]
[4]. Chavapil M, Koopmann C, Scar formation: Physiology and pathological statesOtolaryngol Clin North Am 1984 17:265-72. [Google Scholar]
[5]. Jayes PH, Cross leg flaps: a review of 60 casesBr J Plast Surg 1950 3:1-5. [Google Scholar]
[6]. Cormack GC, Lamberty MA, The blood supply of thigh skinPlast Reconstr Surg 1985 Mar 75(3):342-54. [Google Scholar]
[7]. Laitung JK, The lower postero-lateral thigh flapBr J Plast Surg 1989 Mar 42(2):133-39. [Google Scholar]
[8]. Maruyama Y, Iwahira Y, Popliteo-posterior thigh fasciocutaneous islanded flap for closure around the kneeBr J Plast Surg 1989 Mar 42(2):140-45. [Google Scholar]
[9]. Koshima I, Soeda S, Inferior epigastric artery skin flaps without rectus abdominis muscleBr J Plast Surg 1989 42:645-48. [Google Scholar]
[10]. Koshima I, Moriguchi T, Soeda S, The gluteal perforator-based flap for repair of sacral pressure soresPlast Reconstr Surg 1993 91:678-83. [Google Scholar]
[11]. Ghale S, Bowman N, The distal medial perforators of the lower leg and their accompanying veinsBr J Plast Surg 2005 58(8):1086-89. [Google Scholar]
[12]. Topalan M, Bilgin SS, Ip WY, Chow SP, Effects of torsion on microarterial anastomosis patencyMicrosurgery 2003 23(1):55-59. [Google Scholar]
[13]. Sharma RK, Mehrotra S, Nanda V, The perforator “plus” flap : a simple nomenclature for locoregional perforator-based flapsPlast Reconstr Surg 2005 116:1838-39. [Google Scholar]
[14]. Mehrotra S, Perforator-plus flaps-optimising results while preserving function and esthesisIndian J Plast Surg 2010 Jul 43(2):141-48. [Google Scholar]
[15]. Wong CH, Tan BK, Perforator-sparing transposition flaps for lower limb defects : anatomic study and clinical applicationAnn Plast Surg 2007 Jun 58(6):614-21. [Google Scholar]
[16]. Wong CH, Tan BK, Perforator-plus flaps or perforator-sparing flaps: Different names, same conceptPlast Reconstr Surg 2007 Nov 120(6):1746-47. [Google Scholar]
[17]. Roberts AHN, Dickson WA, Fasciocutaneous flaps for burn reconstruction. A report of 57 flapsBr J Plast Surg 1988 41:150-53. [Google Scholar]
[18]. Tenenhaus M, Lukacs LA, Ogawa R, A brief historical review of flaps and burn reconstructionJ Wounds 2008 20:7 [Google Scholar]
[19]. Fujino T, Contribution of the axial and perforator vasculature to circulation in flapsPlast Reconstr Surg 1967 39:125-37. [Google Scholar]
[20]. Kroll SS, Rosenfield L, Perforator-based flaps for low posterior midline defectPlast Reconstr Surg 1988 81:561-66. [Google Scholar]
[21]. Taylor GI, Palmer JH, The vascular territories (angiosomes) of the body: experimental study and clinical applicationBr J Plast Surg 1987 40:113-41. [Google Scholar]
[22]. Sharma RK, Perforator-plus flap: Evolution of the concept and its place in plastic surgeon’s repertoireIndian J Plast Surg 2010 43(2):148-50. [Google Scholar]
[23]. Panse NS, Bhatt YC, Tandale MS, What is safe limit of the perforator flap in lower extremity reconstruction? Do we have answers yet?Plast Surg International 2011 article ID 349357, 7 pages [Google Scholar]
[24]. Geddes CR, Tang M, Yang D, Anatomy of the integument of lower extremity. In: Blondeel P, Morris SF, Hallock GG et al editorsPerforator flaps: anatomy, technique and clinical applications 2006 St. Louis (MO)QMP:541-78. [Google Scholar]
[25]. Pan SC, Yu JC, Shieh SJ, Distally based anterolateral thigh flap: an anatomic and clinical studyPlast Reconstr Surg 2004 114:1768-75. [Google Scholar]
[26]. Yu P, Selber J, Perforator patterns of the anteromedial thigh flapPlast Reconstr Surg 2011 Sept 128(3):151e-157e. [Google Scholar]
[27]. Sobotta J, Atlas de anatomiahumana 1993 19th edRio de Janeiroed. Guanabara Koogan:70:71:77:203:364 [Google Scholar]
[28]. Zumiotti AV, Teng HW, Briceno NCQ, Lateral flap of the thigh based upon the lateral superior geniculate artery: an anatomic and histomorphometric study and clinical applicationsActa Ortop Bras 2005 13(1) [Google Scholar]
[29]. Bernard SL. (Feb 24, 2012). Knee reconstruction. Retrieved from: http://emedicine.medscape.com/article/1291548 [Google Scholar]
[30]. Hayashi A, Maruyama Y, The medial genicular artery flapAnn Plast Surg 1990 Sept 25(3):174-80. [Google Scholar]
[31]. Otani M, Okamoto H, Kagami H, Anatomical study on perforators of the medial and lateral sural arteries in AsiansNagoya Med J 2012 52:89-98. [Google Scholar]
[32]. Bhattacharya V, Watts RK, Ipsilateral fasciocutaneous flaps for leg and foot defectsIndian J Plast Surg 2003 36(1):30-35. [Google Scholar]
[33]. Stevanovic G, Djordjevic B, Dakovic M, Fasciocutaneous perforators of the lower leg – anatomic study and clinical significanceVojnosanit Pregl 2010 Feb 67(2):136-44. [Google Scholar]
[34]. Ozalp T, Masquelet AC, Bengue TC, Septocutaneous perforators of the peroneal artery relative to the fibula: anatomical basis of the use of pedicled fasciocutaneous flapSurg Radiol Anat 2006 Mar 28(1):54-58. [Google Scholar]
[35]. Wallace CG, Kao HK, Jeng SF, Wei FC, Free-style flaps: a further step forward for perforator flap surgeryPlast Reconstr Surg 2009 Dec 124(6 suppl):e419-26. [Google Scholar]