INTRODUCTION
Over the past 70 years, a number of astute clinicians in the field of dental medicine have observed and recorded the relationship between periodontopathies and systemic manifestations of disease. The influence of systemic conditions on the oral environment, and especially the periodontium, has long been recognized and supported by scientific evidence. However, an evidence base for the influence of periodontal diseases on the overall systemic health has only recently begun to be established. Some fascinating researches have eroded the tradition-bound concept that oral infections such as periodontitis are simply local entities whose effects are limited to the oral tissues. While the clinical observations of many practitioners have long suggested that periodontal diseases can have widespread systemic effects, only recently has a rigorous scientific investigation supported this concept. Evidence which is emerging, has shed light on the converse side of the relationship between the systemic health and the oral health- that is, the potential effects of periodontal disease on a wide range of organ systems. This field of periodontal medicine addresses pertinent questions such as, ‘whether a bacterial infection of the periodontium has a remote effect from the oral cavity’ and ‘whether a periodontal infection is a risk factor for systemic diseases or conditions that affect the human health’. Recent research which has been done in the area of periodontal medicine marks a resurgence in the concept of focal infections. The subgingival environment in the periodontitis patients provides a persistent gram negative bacterial challenge. These organisms Sectionand their products have access to the circulation via the sulcular epithelium, which is ulcerated in disease. The total surface area of the pocket epithelium which is in contact with the sub gingival bacteria, is approximately the size of the palm of an adult hand and bacteraemias occur frequently during daily functions such as brushing and chewing.
PROTEIN ENERGY MALNUTRITION
PEM is the state of decreased body pools of protein, with or without fat depletion or a state of diminished functional capacity, which is caused at least partly by an inadequate nutrient intake which is relative to the nutrient demand [1]. It is a common phenomenon in maintenance dialysis patients and a risk factor for a poor quality of life and increased morbidity and mortality, including cardiovascular death, in these individuals [2]. Various studies have used different criteria to establish the presence of PEM in the dialysis population. Its reported prevalence varies between 18% and 75% in dialysis patients, according to type of dialysis modality, the nutritional assessment tools, and origin of the patient population [3]. The causes of wasting and PEM in dialysis patients.
Inadequate nutrient intake anorexia which is caused by
Uraemic toxicity
Impaired gastric emptying
Inflammation with/without comorbid conditions
Emotional and/or psychological disorders
Dietary restrictions
Prescribed restrictions: Low-potassium, low-phosphate regimens
Social constraints: Poverty, inadequate dietary support
Physical incapacity: Inability to acquire or prepare food or to eat
Nutrient losses during dialysis
Loss through the haemodialysis membrane into the haemodialysate
Adherence to the haemodialysis membrane or tubing
Loss into the peritoneal dialysate
Hypercatabolism which is caused by
Comorbid illnesses
Cardiovascular diseases
Diabetic complications
Infection and/or sepsis
Other comorbid conditions
Hypercatabolism which is associated with dialysis treatment
Negative protein balance
Negative energy balance
Endocrine disorders of uraemia
Resistance to insulin
Resistance to growth hormones and/or IGF-1
Increased serum levels of or sensitivity to glucagon
Hyperparathyroidism
Other endocrine disorders
Acidaemia with metabolic acidosis
Concurrent nutrient loss with frequent blood losses
Some of these factors can also lead to inflammation [4]. Hence, the known overlap between malnutrition and inflammation in the dialysis patients may have its root at the causal level. The origin of PEM appears to precede the dialysis treatment, and it is engendered progressively as Glomerular Filtration Rate (GFR) decreases of less than 55 mg/min [5]. Hypoalbuminaemia, hypotransferrinaemia, and hypocholesterolaemia have been shown to develop along with the progression of the CKD stages, as has been shown in the Modification of Diet in Renal Disease Study and other studies5. Classically, 3 major lines of inquiries, i.e., dietary intake, biochemical means, and body composition, are used to assess the protein-energy nutritional status. The composite indices that include a combination of assessment measures within these categories also are used, such as the Subjective Global Assessment of Nutrition (SGA) [6] or the Malnutrition-Inflammation Score (MIS) [7]. Some more frequently studied indicators of malnutrition in dialysis patients, that are associated with the clinical outcome, include a decreased dietary protein and energy intake [8]; reduced weight for height [2], Body Mass Index (BMI) [9] and total-body fat percentages, decreased total body nitrogen and total-body potassium levels; reduced mid arm muscle mass and skinfold thicknesses [10]; low serum concentrations of albumin, prealbumin (transthyretin), transferrin (Total Iron-Binding Capacity (TIBC), cholesterol, and creatinine; and a more abnormal score which is obtained with the use some nutritional assessment tools, such as the SGA [6] and the MIS [7]. SGA also may be a marker of the degree of sickness and comorbidity in maintenance dialysis patients. During acute catabolic states, the urea nitrogen appearance may increase transiently, independent of the food intake.
INFLAMMATION-THE SILENT KILLER
Inflammation is defined as a localized protective response which is elicited by the injury to or the destruction of the tissues, that serves to destroy, dilute, or sequester both the injurious agent and the injured tissue [11]. The acute-phase response (or reaction) is a major pathophysiological phenomenon that accompanies inflammation and it is associated with an increased activity of the proinflammatory cytokines [12]. If the inflammation becomes prolonged and persistent in the form of the so-called chronicacute-phase reaction, it may lead to adverse consequences such as a decline in the appetite, an increased rate of protein depletion in the skeletal muscle and other tissues, muscle and fat wasting, hypercatabolism, endothelial damage, and atherosclerosis [13]. It is believed that inflammation may have an important role in the increased prevalence of cardiovascular disease and mortality which are associated with a renal insufficiency. A renal failure may lead to increased inflammatory responses through a number of mechanisms. Periodontitis is a common disease, the clinical signs of which are seen in the early middle age. It is chronic tissue-destructive inflammation, which degrades the attachment apparatus of the teeth, causing tooth loss and in its most severe form, edentulousness.
THE RELATIONSHIP BETWEEN PEM AND INFLAMMATION
The association between PEM and inflammation in patients with CKD and End Stage Renal Disease (ESRD) may be an explanation for the malnutrition-associated mortality [14]. Several investigators have suggested that PEM is a consequence of the chronic inflammatory processes in patients with a renal insufficiency.
Thus, chronic inflammation may be the missing link that causally ties PEM to the morbidity and the mortality in these individuals. The following arguments have been proposed to indicate that the development of PEM is secondary to inflammation.
1. The proinflammatory cytokines such as the tumour necrosis factor-α (TNF-α), not only promote the catabolic processes, engendering both the protein degradation and suppression of the protein synthesis, but they also induce anorexia [15]. A low appetite has been associated with increased levels of inflammatory markers in the haemodialysis patients.
2. The dialysis patients with inflammation are reported to develop weight loss and a negative protein balance, even those who have an intact appetite, because there may be a shift in the protein synthesis from the muscle to the acute phase proteins as the renal function declines [16].
3. In patients with CKD and ESRD, the albumin synthesis is suppressed when the serum CRP level is elevated [17].
4. Inflammation also may lead to hypocholesterolaemia, a strong mortality risk factor in dialysis patients and the marker of a poor nutritional status [18].
THE TARGET POPULATION-CHRONIC KIDNEY DISEASE
The haemodialysis (HD) patients face a 25% annual mortality rate, with 50% of the reported deaths being attributed to cardiovascular disease. All the causes and the cardiovascular mortality correlate with the acute-phase proteins such as the C-reactive protein (CRP). The hepatic CRP synthesis is upregulated by inflamma tion; however, the elevated CRP values are found frequently in the absence of an apparent infection or inflammation. Because destructive periodontal diseases have been associated with elevated CRP levels, it was hypothesised that they could contribute to the elevated CRP values in the HD populations. Between 30 and 50% of the prevalent patients who are on haemodialysis (HD), have elevated serum levels of the inflammatory markers. In some patients, this elevation is chronic, and in some, it is intermittent and it is generally associated with the breakthrough processes. Furthermore, on many occasions, the HD sessions may trigger inflammation in a way that is not always identifiable with the conventional markers. The inflammatory markers are powerful predictors of the mortality after adjustment for the other risk factors. Inflammation is also responsible for the other mortality risk factors such as anaemia, malnutrition, vascular disease, and left ventricular hypertrophy. For lowering the high morbidity/mortality rate in the patients who are on HD, the inflammation must be tackled.
Mics Causes and Consequences
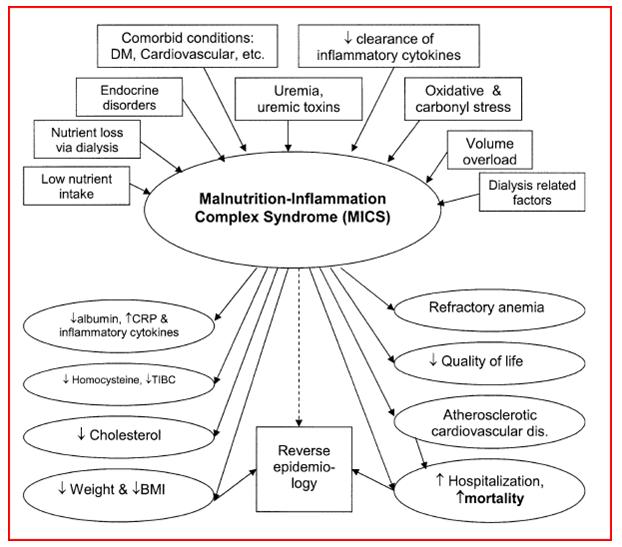
THE MALNUTRITION INFLAMMATION COMPLEX SYNDROME-A NEW PARADIGM
Protein-Energy Malnutrition (PEM) and inflammation are common and they are usually concurrent in maintenance dialysis patients. Many factors that appear to lead to these 2 conditions overlap, as do the assessment tools and the criteria for detecting them, such as hypoalbuminaemia. Both these conditions are related to a poor dialysis outcome. A low appetite and a hypercatabolic state are among the common features. The PEM in dialysis patients has been suggested to be secondary to inflammation; however, the evidence is not conclusive, and an equicausal status or an even opposite causal direction is possible. Hence, Malnutrition-Inflammation Complex Syndrome (MICS) is an appropriate term. The possible causes of MICS include comorbid illnesses, oxidative and carbonyl stress, nutrient loss through dialysis, anorexia and a low nutrient intake, uraemic toxins, a decreased clearance of the inflammatory cytokines, a volume overload, and dialysis-related factors. MICS is believed to be the main cause of erythropoietin hyporesponsiveness, a high rate of cardiovascular atherosclerotic disease, a decreased quality of life, and increased mortality and hospitalization in the dialysis patients. Because MICS leads to a low body mass index, hypocholesterolaemia, hypocreatininaemia, and hypohomocysteinaemia, a “reverse epidemiology” of the cardiovascular risks can occur in the dialysis patients. Therefore, obesity, hypercholesterolaemia, and increased blood levels of creatinine and homocysteine appear to be protective and paradoxically associated with a better outcome. There is no consensus on how to determine the degree of severity of MICS or on how to manage it. Several diagnostic tools and treatment modalities have been discussed. A successful management of MICS may ameliorate the cardiovascular epidemic and the poor outcome in dialysis patients. Clinical trials which focus on MICS and its possible causes and consequences are urgently required to improve the poor clinical outcome in dialysis patients.
THE REVERSE EPIDEMIOLOGY FACTOR
The conventional risk factors of cardiovascular disease and mortality in the general population, such as body mass, serum cholesterol, and blood pressure, are also found to be related to the outcome in maintenance dialysis patients, but often in the opposite direction. Obesity, hypercholesterolaemia, and hypertension appear to be the protective features that are associated with a greater survival among the dialysis patients. A similar protective role has been described for high serum creatinine and possibly the homocysteine levels in the End-Stage Renal Disease (ESRD) patients. These findings are in contrast to the well-known association between over-nutrition and the poor outcome in the general population. The association between under-nutrition and an adverse cardiovascular outcome in dialysis patients, which stands in contrast to that which is seen in the non-ESRD individuals, has been referred to as “reverse epidemiology.” A publication bias may have handicapped or delayed the additional reports of such paradoxical findings in the ESRD patients. The aetiology of this inverse association between the conventional risk factors and the clinical outcome in dialysis patients, is not clear. Several possible causes have been hypothesized. Firstly, a survival bias may play a role, since only a small number of patients with Chronic Kidney Disease (CKD) survive long enough to reach ESRD. Hence, the dialysis patients are probably a distinctively selected population among the CKD patients and they may not represent the risk factor constellations of their CKD predecessors. Secondly, the time discrepancy between the competitive risk factors may play a role. For example, the survival disadvantages of under-nutrition, which is frequently present in dialysis patients, may have a major impact on the mortality in a shorter period of time, and this overwhelms the long-term negative effects of over-nutrition on survival. Thirdly, the presence of the “malnutrition-inflammation Complex Syndrome” (MICS) in dialysis patients may also explain the existence of a reverse epidemiology in the dialysis patients. Both protein-energy malnutrition and inflammation or a combination of these two, are much more common in dialysis patients than in the general population and many elements of MICS, such as a low weight-for-height, hypocholesterolaemia, or hypocreatininaemia, are the known risk factors of a poor outcome in dialysis patients. The existence of a reverse epidemiology may have a bearing on the management of the dialysis patients. It is necessary that new standards or goals for the traditional risk factors such as the body mass, serum cholesterol, and blood pressure, should be considered.
THE MALNUTRITION INFLAMMATION SCORE- A COMPREHENSIVE TOOL
Although many measures of MICS, such as serum albumin or the C-reactive protein (CRP) correlate with the clinical outcome, it is not clear as to which one these blood values has a superior outcome predictability as compared to the others. Moreover, such blood tests generally do not evaluate the clinical condition and the outcome in a combined way for an individual patient, and some of these tests such as CRP or the pro-inflammatory cytokines are not measured routinely and they are too expensive too. A comprehensive scoring system that is reproducible and which is based on the available data can be useful for this purpose, if it is capable of risk stratifying the MHD patients in a quantitative way for an optimal management, while being a practical and an easy tool without cumbersome methods or sophisticated calculations. By using the seven components of the conventional Subjective Global Assessment of Nutrition (SGA), a semi-quantitative scale with three severity levels, and by combining it with three new elements [the Body Mass Index (BMI), serum albumin, and the Total Iron Binding Capacity (TIBC) to represent serum transferrin] in an incremental fashion, the so-called ‘Malnutrition Inflammation Score’ (MIS) with 10 components was created [19]. Each MIS component has four levels of severity from 0 (normal) to 3 (very severe). In a recent prospective study, the MIS was found to be a comprehensive scoring system with significant associations, with a prospective hospitalization and mortality, as well as measures of nutrition, inflammation and anaemia in MHD patients and it was found to be superior to the conventional SGA and to the individual laboratory values, as a predictor of the dialysis outcome.
CONCLUSION
The mortality is markedly elevated in haemodialysis (HD) patients. Between 30 and 50% of the prevalent patients have elevated serum levels of the inflammatory markers such as the C-reactive protein and IL-6. Most of the observational studies have indicated that malnutrition, inflammation, and/or oxidative stress, which are together known as the ‘malnutrition—inflammation complex (or Cachexia) Syndrome’ (MICS), may be a much stronger predictor of the death risk in dialysis patients than the traditional cardiovascular risk factors [20].The presence of inflammation, chronic or episodic, has been found to be associated with an increased mortality risk. The causes of inflammation are multifactorial and they include patient-related factors, such as an underlying disease, comorbidity, oxidative stress, infections, obesity, and genetic or immunologic factors, or on the other side, HD-related factors which mainly depend on the membrane biocompatibility and the dialysate quality. An adequate knowledge on these causes and their prevention or treatment, if possible, may contribute to improving the inflammatory state of the patients who are on HD and possibly their mortality. Periodontitis is a chronic inflammatory disease of the supporting tissues which results from the infection and interaction of selected bacterial species, with components of the host response in disease-susceptible individuals. The recent years have witnessed an emerging evidence which has linked periodontitis to various systemic illnesses and it was confirmed that the periodontal health was poor in haemodialysis patients and that it correlated with the markers of malnutrition and inflammation [21]. The treatment of periodontal diseases reduces the chronic systemic inflammation in maintenance haemodialysis patients [22] by inducing a decline in the systemic inflammatory response [23]. Further studies could evaluate whether the treatment of periodontitis would translate into the amelioration of an inflammatory and protein depletion state and they could confirm a cause-effect relationship which leads to improved outcomes in the haemodialysis population.
[1]. Kalantar Zadeh K, Ikizler TA, Block G, Avram MM, Kopple JD, Malnutrition—inflammation complex syndrome in dialysis patients: causes and consequencesAm J Kidney Dis 2003 42:864-81. [Google Scholar]
[2]. Kopple JD, Zhu X, Lew NL, Lowrie EG, Body weight for-height relationships predict mortality in maintenance hemodialysis patientsKidney Int 1999 56:1136-48. [Google Scholar]
[3]. Kalantar-Zadeh K, Kopple J, Nutritional management of hemodialysis patients, in Massry S (ed)Nutritional Management of Renal Disease 2003 2Philadelphia, PALippincott, Williams and Wilkins:183-98. [Google Scholar]
[4]. Kopple JD, McCollum Award Lecture, 1996. Protein energy malnutrition in maintenance dialysis patientsAm J Clin Nutr 1997 65:1544-57. [Google Scholar]
[5]. Kopple JD, Greene T, Chumlea WC, Relationship between nutritional status and the glomerular filtration rate: Results from the MDRD studyKidney Int 2000 57:1688-03. [Google Scholar]
[6]. Enia G, Sicuso C, Alati G, Zoccali C, Subjective Global Assessment of nutrition in dialysis patientsNephrol Dial Transplant 1993 8:1094-98. [Google Scholar]
[7]. Kalantar-Zadeh K, Kopple JD, Block G, Humphreys MH, A malnutrition-inflammation score is correlated with morbidity and mortality in maintenance hemodialysis patientsAm J Kidney Dis 2001 38:1251-63. [Google Scholar]
[8]. Kalantar-Zadeh K, Kopple JD, Deepak S, Block D, Block G, Food intake characteristics of hemodialysis patients as obtained by food frequency questionnaireJ Ren Nutr 2002 12:17-31. [Google Scholar]
[9]. Port FK, Ashby VB, Dhingra RK, Roys EC, Wolfe RA, Dialysis dose and body mass index are strongly associated with survival in hemodialysis patientsJ Am Soc Nephrol 2002 13:1061-66. [Google Scholar]
[10]. Wolfson M, Strong CJ, Minturn D, Gray DK, Kopple JD, Nutritional status and lymphocyte function in maintenance hemodialysis patientsAm J Clin Nutr 1984 39:547-55. [Google Scholar]
[11]. Newman Dorland W, Anderson D, Dorland’s Illustrated Medical Dictionary 2000 Philadelphia, PASaunders [Google Scholar]
[12]. Kushner I, Acute-phase proteins and other systemic responses to inflammationN Engl J Med 1999 340:448-54. [Google Scholar]
[13]. Suffredini AF, Fantuzzi G, Badolato R, Oppenheim JJ, O’Grady NP, New insights into the biology of the acute phase responseJ Clin Immunol 1999 19:203-14. [Google Scholar]
[14]. Qureshi AR, Alvestrand A, Divino-Filho JC, Inflammation, malnutrition, and cardiac disease as predictors of mortality in hemodialysis patientsJ am Soc Nephrol 2002 13:S28-S36. [Google Scholar]
[15]. McCarthy DO, Tumor necrosis factor alpha and interleukin-6 have differential effects on food intake and gastric emptying in fasted ratsRes Nurs Health 2000 23:222-28. [Google Scholar]
[16]. Kaizu Y, Kimura M, Yoneyama T, Miyaji K, Hibi I, Kumagai H, Interleukin-6 may mediate malnutrition in chronic hemodialysis patientsAm J Kidney Dis 1998 31:93-100. [Google Scholar]
[17]. Kaysen GA, Chertow GM, Adhikarla R, Young B, Ronco C, Levin NW, Inflammation and dietary protein intake exert competing effects on serum albumin and creatinine in hemodialysis patientsKidney Int 2001 60:333-40. [Google Scholar]
[18]. Bologa RM, Levine DM, Parker TS, Cheigh JS, Serur D, Stenzel KH, Interleukin-6 predicts hypoalbuminemia, hypocholesterolemia, and mortality in hemodialysis patientsAm J Kidney Dis 1998 32:107-14. [Google Scholar]
[19]. Kalantar-Zadeh K, Kopple JD, Block G, Humphreys MH, A malnutrition-inflammation score is correlated with morbidity and mortality in maintenance hemodialysis patientsAm J Kidney Dis 2001 38:1251-63. [Google Scholar]
[20]. Kalantar-Zadeh K, Kopple JD, Kamranpour N, Fogelman AM, Navab M, HDL-inflammatory index correlates with poor outcome in hemodialysis patientsKidney International 2007 72:1149-56. [Google Scholar]
[21]. Li-Ping Chen, Chin-Kang Chiang, Chiu-Po Chan, Kuan-Yu Hung, Chiung-Shing Huang, Relationship between periodontal disease and morality in patients treated with maintenance hemodialysisAm J Kidney Disease 2007 57:2:276-82. [Google Scholar]
[22]. Siribamrungwong M, Puangpanngam K, Treatment of periodontal diseases reduces chronic systemic inflammation in maintenance hemodialysis patientsRen Fail 2012 34(2):171-5. [Google Scholar]
[23]. Vilela Eduardo Machado, AmaralBastos Jessica, Fernandes Natalia, Ferreira Ana Paula, Chaoubah Alfredo, Bastos Marcus Gomes, Treatment of chronic periodontitis decreases serum prohepcidin levels in patients with chronic kidney diseaseClinics 2011 66(4):657-62. [Google Scholar]