The Cytological Diagnosis of Extra-Oral Plasmablastic Lymphoma: A Rare Entity
Kanthilatha Pai1, Lakshmi Rao2
1 Professor, Department of Pathology, KMC International Centre, Manipal, India.
2 Professor, Department of Pathology, KMC, Manipal, India.
NAME, ADDRESS, E-MAIL ID OF THE CORRESPONDING AUTHOR: Dr. Kanthilatha Pai, Professor, Department of Pathology, KMC International Centre, Manipal, India.
Phone: 9900405073
E-mail: klpai@yahoo.com
Non-Hodgkin’s Lymphomas (NHLs) which are associated with the Acquired Immunodeficiency Syndrome (AIDS) are heterogeneous. Plasmablastic Lymphoma (PBL) was first recognized as an aggressive, invariably fatal subtype of Non-Hodgkin’s Lymphoma which occurred mostly in patients with AIDS, with distinct histomorphologic and immunophenotypic findings, which affected the jaw and the oral mucosa exclusively. Subsequently, there have been case reports which have described extra-oral plasmablastic lymphomas in the lung, jejunum, caecum, nasal mucosa, etc. We are reporting a case of this rare subtype of diffuse large B-cell lymphoma which presented as a soft tissue mass, which we believe is the first case to be diagnosed by FNAC.
Extra-oral, Plasmablastic lymphoma
INTRODUCTION
PBL is a rare form of Non-Hodgkin’s Lymphoma that is associated with AIDS, and it is commonly encountered in the oral cavity. There are few case reports on PBL which had occurred in extra-oral locations like the gastro-intestinal tract, the nasal mucosa and the soft tissues. We are reporting an unusual case presentation of PBL, which presented as a soft tissue mass clinically and masqueraded as a soft tissue sarcoma. To the best of our knowledge, this is the first case report on PBL which was diagnosed by FNAC.
CASE REPORT
A 35 year old female, who was a known case of retropositive illness on HAART, presented with a swelling in the gluteal region, of 6 months duration, which had rapidly enlarged over the past 2-3 weeks. The patient had difficulty in walking by virtue of its size and also difficulty in lying supine. She gave a history of constipation of 2 weeks duration. She also complained of weight loss and intermittent fever.
On clinical examination, she was found to be poorly built and poorly nourished, with moderate pallor. There was no evidence of lymphadenopathy in the patient.
The local examination revealed a large smooth gluteal swelling which measured approximately 20.0 x 10.0 cm, which involved the sacral region and both glutei. There were dilated veins over the swelling. The swelling was hard in consistency. The systemic examination was unremarkable. A clinical diagnosis of a soft tissue sarcoma was made.
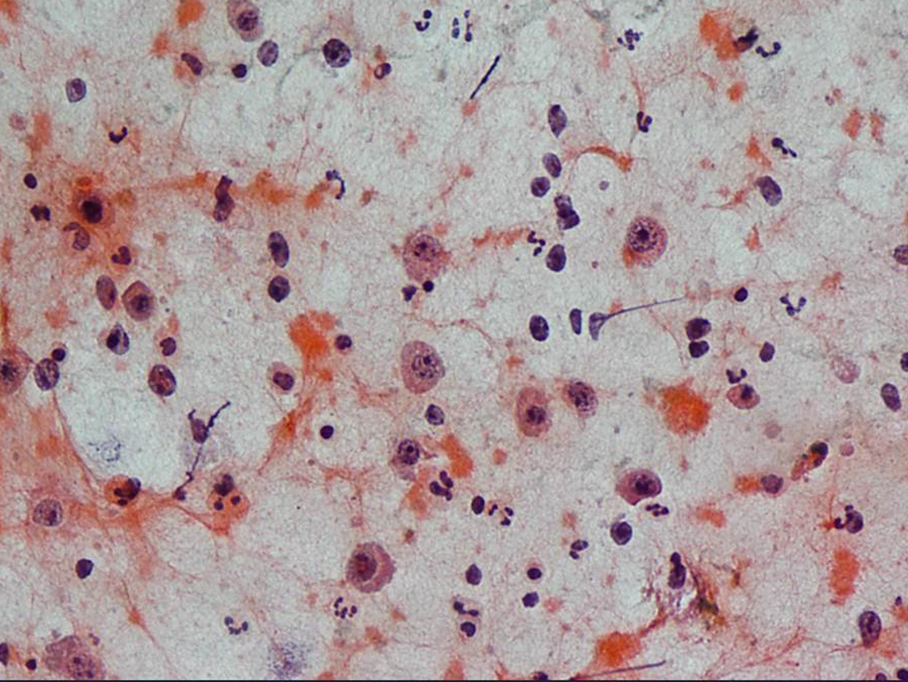
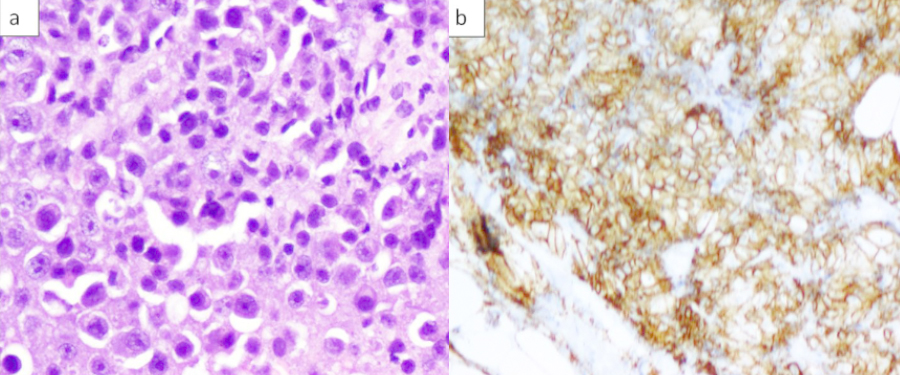
Fine needle aspiration was performed, which revealed a haemorrhagic smear with large plasmacytoid cells which had central to eccentrically placed large nuclei, coarse chromatin and prominent nucleoli with abundant densely eosinophilic cytoplasm which exhibited nuclear pleomorphism, occasional binucleate forms and atypical mitotic figures. Also present were smaller atypical lymphoid cells and mature appearing plasma cells, scattered neutrophils, eosinophils and apoptotic bodies [Table/Fig-1]. Serum and urine cellsprotein electrophoresis were requested, to rule out a plasmablastic myeloma, which revealed the absence of paraprotein or free light chains. A diagnosis of an extra-oral PBL was made. A subsequent biopsy showed a tumour which was composed of sheets of plasmablasts, amidst many mature plasma cells and atypical lymphoid cells [Table/Fig-2a]. The immunohistochemical staining with CD20 was negative, while CD138 revealed a strong positivity among these cells [Table/Fig-2b] Based on the clinical presentation, morphology and immunophenotype, a diagnosis of an extra-oral plasmablastic lymphoma was confirmed.
FNAC smear showing plasmacytoid cells with prominent nuclei in a background of atypical lymphocytes, PAP smear, 200x
Photomicrograph showiig sheets of plasma blasts, mature plasma cells and atypical lymphocytes H & E 200X.b) CD 138 diffuse membranous positivity among the cellsprotein
DISCUSSION
PBL of the oral cavity was first described in 1997 [1]; it has been recognized as a distinct entity, a subtype of a diffuse large B-cell lymphoma, by the WHO classification of lymphoproliferative disorders [2]. Since it was first described, more than 150 patients with this disease have been reported [3]. PBLs have been strongly associated with immunodeficiency, most particularly in patients with the HIV infection [3]. PBL accounts for 2.6% of all the HIVrelated non-Hodgkin’s lymphomas [4].
Recently, the primary sites have been more frequently reported to be outside of the oral cavity or to be concurrent with an oral involvement [5]. Over one-third of all the cases with PBL were first noted at extra-oral locations, and the gastrointestinal tract has been observed to be the most common extra-oral site (10.6%) [3].
The morphologic spectrum ranges from large immature tumour cells (immunoblasts, plasmablasts) to cells with a mature plasmacytic differentiation, which present in a variable and somewhat overlapping maturity range. PBLs usually have a characteristic immunophenotype; they are negative for the typical B-cell antigens (e.g., CD20 and CD45) and positive for the plasma cell markers such as MUM1, EMA, CD38, and CD138 [1,3]. PBLs characteristically display a high rate of mitotic activity by the Ki-67 proliferation index [1].
Although it is not specific, the combination of these histopathological features is characteristic of PBL; therefore, immunophenotypic studies must be performed to confirm the diagnosis. PBL should be differentiated from neoplastic plasma cells and other varieties of NHL.
The main differential diagnoses are extramedullary plasmablastic myeloma and diffuse large B cell lymphoma. It is clinically important and critical to differentiate plasmablastic myelomas and PBLs, as the treatment for these two diseases is significantly different [6]. The distinction between PBLs and plasmablastic myelomas frequently depends on their clinical presentations. The detection of paraproteinaemia in blood and/or excess light chains (Bence-Jones proteins) in urine, lytic bone lesions, and hypercalcaemia or anaemia favours the diagnosis of a plasma cell myeloma over PBL. Immunosuppression, especially HIV-related, is much more frequently associated with PBL. Unlike PBL, a plasmacytoma typically consists of mature plasma cells without a high rate of mitotic activity.
The B-cell lymphomas nearly always express CD20, CD45-RA and CD79a; which is not typical for PBL [1,3].
CONCLUSION
The clinical features of PBL, which include its association with HIV, the male gender and predilection for the oral cavity, may help in the differential diagnosis, however, an extra-oral location in female patients makes it more difficult to suspect it clinically. This case highlights an unusual presentation of PBL which presented as a soft tissue mass. Hence, PBL may be included in the differential diagnosis of soft tissue masses in HIV positive patients.
[1]. Delecluse HJ, Anagnostopoulos I, Dallenbach F, Hummel M, Marafioti T, Schneider U, Plasmablastic lymphomas of the oral cavity: a new entity associated with the human immunodeficiency virus infectionBlood 1997 89:1413-20. [Google Scholar]
[2]. Harris NL, Jaffe ES, Diebold J, Flandrin G, Muller-Hermelink HK, Vardiman J, Lister TA, Bloomfield CD, The World Health Organization classification of neoplasms of the hematopoietic and lymphoid tissues: report of the Clinical Advisory Committee meeting-Airlie House, Virginia,1997Hematol J 2000 1:53-66. [Google Scholar]
[3]. Riedel DJ, Gonzalez-Cuyar LF, Zhao XF, Redfield RR, Gilliam BL, Plasmablastic lymphoma of the oral cavity: a rapidly progressive lymphoma associated with HIV infectionLancet Infect Dis 2008 8:261-67. [Google Scholar]
[4]. Folk GS, Abbondanzo SL, Childers EL, Plasmablastic lymphoma: A clinicopathologic correlationAnn Diagn Pathol 2006 10:8-12. [Google Scholar]
[5]. Lin Y, Rodrigeus GD, Turner JF, Vasef MA, Plasmablastic lymphoma of the lung: report of a unique case and review of the literatureArch Pathol Lab Med 2001 125:282-85. [Google Scholar]
[6]. Teruya-Feldstein J, Diffuse large B-cell lymphomas with plasmablastic differentiationCurr Oncol Rep 2005 7:357-63. [Google Scholar]