The Use of Procalcitonin as a Marker of Sepsis in Children
Bin Mathew1, Dinesh Roy D2, T Vijaya Kumar3
1 Research Scholar, Vinayaka Missions University, Salem.
2 Senior Cytogeneticist, Genetika, Centre for Advanced Genetic Studies, Trivandrum, India.
3 Head Basic Medical Sciences, Educare Institute of Dental Sciences, Malappuram, India.
NAME, ADDRESS, E-MAIL ID OF THE CORRESPONDING AUTHOR: Bin Mathew, Nallanattu House, Mullankuzhy, Collectorate P.O. Kottayam – 686002, Kerala State, India.
E-mail: binmathew@sify.com
Objective: Sepsis is a life-threatening disease which is associated with high rates of morbidity and mortality. The critically ill patients often manifest a Systemic Inflammatory Response Syndrome (SIRS) which is independent of an infection. The early diagnosis of different severities of sepsis is important for an early implementation of the specific therapies. Our objective was to evaluate the diagnostic and the prognostic values of blood Procalcitonin (PCT) in cases of bacterial septicaemia in children.
Methods: The total sample comprised of 150 subjects who were admitted to the ICU with septicaemia and 50 normal, healthy, age and sex matched children. The first sample was collected at the time of admission, before the start of the antibiotic therapy (T0). A second sample was collected at 24 hours (T24) and a final sample was collected at 96 hours (T96). A PCT value of > 0.5ng/ml was accepted as positive.
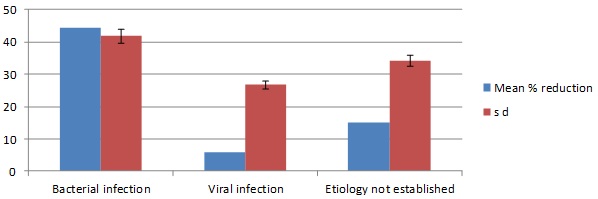
Results: 63% of the children who were diagnosed with a bacterial aetiology showed detectable blood PCT levels with higher concentrations, while in the children who were diagnosed with a viral aetiology, only 22.2 % had detectable PCT levels, but in lower concentrations. The mean percentage reduction in the PCT value among the bacterial infection subjects was 44.39 ± 41.82 as compared to that in the viral infection subjects (5.71 ± 26.68) and in the subjects where the aetiology was not established (5.71 ± 26.68).
Conclusion: The results which were obtained in our study con- firmed that the PCT levels were a better marker for the bacterial infections. The PCT measurements may be used as a guide to the antibiotic therapy in critically ill children with suspected sepsis.
Procalcitonin, Sepsis, SIRS, Bacterial infection
INTRODUCTION
The early diagnosis and the appropriate treatment of sepsis is a daily challenge in the medical intensive care unit and in the emergency room. Therefore, a rapid and an accurate diagnosis is essential for improving the survival rate of the patients, especially children [1, 2]. Sepsis is a potentially deadly medical condition that is characterized by a whole-body inflammatory state which is called the systemic inflammatory response syndrome (SIRS) and the presence of a known or suspected infection [3]. While Procalcitonin (PCT) performs quite well as a diagnostic biomarker, it excels when it is called upon as a monitoring or prognostic bio-marker.
The Present study was undertaken:
To evaluate the serum PCT levels as a marker of the sepsis in the bacterial infections of children.
To evaluate the prognostic application of serum PCT in children with sepsis.
MATERIALS AND METHODS
One hundred and fifty children who were admitted to the neonatal and the paediatric intensive care units of a tertiary referral hospital constituted the test group. Fifty children with no evidence or history of an acute or chronic infection formed the control group. In all the cases, the samples that came to the clinical laboratory for routine investigations were used for the analysis after getting an informed consent from the parents. The first sample was collected from the test group at the time of admission (T0), a second sample was collected at 24 hours (T24) and the final sample was collected 96 hours after the admission (T96). Procalcitonin was estimated and a value of ≥0.5 ng/ml was accepted as pathologically significant. Blood Cultures were performed in all the cases to detect sepsis along with other sepsis monitoring and aetiological confirmation tests.
The data was analyzed by using SPSS, ver. 16.0. The association between the qualitative variables was analyzed by using the Chisquare test and the paired ‘t’ test, the independent sample ‘t’ test and one way ANOVA were used for the comparison of the mean of the quantitative variables.
RESULTS
The subjects who were included in the study were classified into 3 groups based on their ages: Children below 2 years of age, children between the ages of 2 and 5 years and children above 5 years of age [Table/Fig-1]. Those subjects below 2 years showed the highest number of bacterial infections (n=37, 61.7%)
Age Distribution of Test and Control Subjects.
| Age in years | Bacterial infection | Viral infection | Etiology not established | Control | Total |
|---|
| N | % | N | % | N | % | N | % | N | % |
| <2 | 37 | 61.7 | 26 | 72.2 | 33 | 61.1 | 14 | 28.0 | 110 | 55.0 |
| 2-5 | 20 | 33.3 | 10 | 27.8 | 15 | 27.8 | 28 | 56.0 | 73 | 36.5 |
| >5 | 3 | 5.0 | 0 | .0 | 6 | 11.1 | 8 | 16.0 | 17 | 8.5 |
| Total | 60 | 100.0 | 36 | 100.0 | 54 | 100.0 | 50 | 100.0 | 200 | 100.0 |
The values of PCT were divided into four different classes [Table/Fig-2]. The PCT values which were above 0.5 ng.ml were considered to be abnormal and as suggestive of an infection. While the number of subjects with a PCT level below 0.5 ng/ml were analyzed, only 36.7% of the study subjects were found to have bacterial infections, while 77.8% of the subjects were found to have viral infections. In case of the subjects with PCT values above 0.5 ng/ml, the confirmed bacterial infection cases showed a higher percentage as compared to the viral aetiology subjects and the subjects with an aetiology of unknown origin. This showed that PCT could be used as an indicator of bacterial infections.
PCT values obtained in respect to etiology of infection.
| PCT | Bacterial infection | Viral infection | Etiology not established | Control |
|---|
| N | % | N | % | N | % | N | % |
| 0.0-0.5 ng/ml | 22 | 36.7 | 28 | 77.8 | 44 | 81.5 | 49 | 98.0 |
| 0.5-2.0 ng/ml | 13 | 21.7 | 7 | 19.4 | 7 | 13.0 | 1 | 2.0 |
| 2.0-10.0 ng/ml | 11 | 18.3 | 1 | 2.8 | 1 | 1.9 | 0 | .0 |
| >10.0 ng/ml | 14 | 23.3 | 0 | .0 | 2 | 3.7 | 0 | .0 |
| Total | 60 | 100.0 | 36 | 100.0 | 54 | 100.0 | 50 | 100.0 |
(X2 =24.336, p<0.001)
(X2 =69.007, p<0.001)
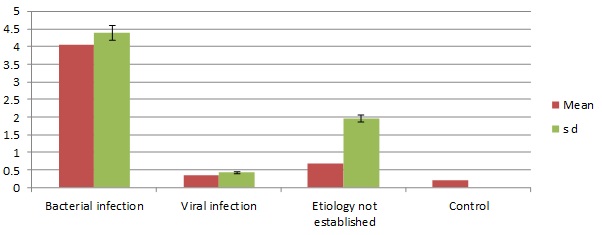
The mean initial PCT value [Table/Fig-3] of the bacterial infection cases was 4.05 ± 4.39 ng/ml, that of the viral infection cases was 0.34 ± 0.42 ng/ml, that of the cases where the aetiology was not established was 0.69 ± 1.95 ng/ml and that of the controls was 0.20 ± 0.00 ng/ml. The mean initial PCT value was significantly higher in the bacterial infection cases as compared by one way ANOVA to that of the other etiological cases (p<0.05).
Comparison of mean initial PCT.
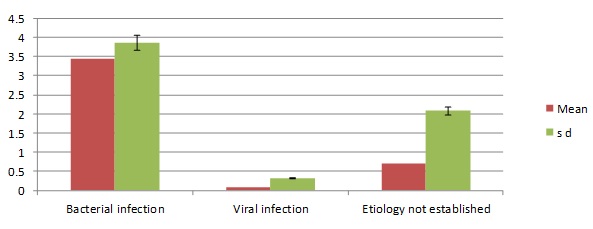
The comparison of the mean PCT values after 24 hours and 96 hours was done by the paired ‘t’ test [Table/Fig-4]. The mean PCT value among the bacterial infection cases after the 24th hour was found to be 4.56±4.68 ng/mL and after the 96th the hour, it had reduced to 1.12±1.80 ng/ml. The observed difference was statistically significant (p<0.05).
Comparison of mean PCT values after 24 hours and 96 hours
| N | PCT | Paired comparison |
|---|
| | After 24 hrs. | After 96 hrs. | t | p |
| Mean | Mean |
| Bacterial infection | 60 | 4.56 | 1.12 | 6.890 | p<0.001 |
| Viral infection | 36 | 0.34 | 0.26 | 1.561 | 0.127 |
| Etiology not established | 54 | 0.94 | 0.21 | 2.551 | 0.014 |
On observing the reduction of the mean PCT value between the 24th hour and the 96th hour among the bacterial infection cases, the viral infection cases and the cases in which the aetiology was not established [Table/Fig-5], the reduction in the mean PCT value was found to be significantly higher in the bacterial infection cases (p<0.05). The mean percentage reduction in the PCT value [Table/Fig-6] among bacterial infection subjects was 44.39±41.82. The bacterial infection cases had a significantly higher reduction than the other cases. No cases of mortality among the study subjects were reported during the period of the study.
Reduction of mean PCT between 24 and 96 hour.
Percentage Reduction in PCT between 24 hr. and 96 hr.
DISCUSSION
Sepsis suffers from a lack of specific clinical signs and symptoms and it may be difficult to distinguish it from other non-infective conditions in critically ill patients [4, 5]. The World Health Organization (WHO) has estimated that globally, there are about 5 million neonatal deaths per year and that ninety eight percent of them occur in the developing countries in the first week of life [6, 7]. In this study, there was a decrease in the number of bacterial infection cases, which was observed with an increase in age. Many studies had shown the efficiency of PCT in determining sepsis. In the present study also, PCT showed a statistical significance in its relationship with sepsis (p<0.001). The results which were obtained in our study also showed a marked increase in the PCT levels in the infections with a bacterial aetiology.
The early diagnosis of sepsis and the initiation of an effective antimicrobial therapy have been major predictors of the outcome in patients with sepsis [8, 9]. Thus, an early identification of the causative agent is crucial and it is often informative enough for directing the treatment decisions towards an evidence-based antimicrobial therapy [10]. Various studies which were done on sepsis, showed that the PCT levels fell quickly when an appropriate antibiotic therapy was initiated [11]. The serial measurement of the PCT levels has also been used as a monitoring biomarker to direct and limit the antibiotic usage. The purpose of this application was to reduce both the bacterial antibiotic resistance as well as the patient-centred side effects such as nephrotoxicity and drug reactions. Multiple adult studies which were done by using PCT-guided algorithms have shown substantial reductions of the antibiotic exposure without increases in the adverse outcomes [12]. In the present study, the mean percentage reduction in PCT was statistically higher in the bacterial infection cases than in the viral infection cases or in the infections of an unknown aetiology. On the basis of the results which were obtained, the PCT levels were also found to be useful in monitoring the course of the infection. In all the cases with a confirmed bacterial aetiology in our study, there was a drop in the PCT levels with a response to the antibiotic treatment.
CONCLUSION
This study supports the usefulness of PCT in establishing an early diagnosis of sepsis in children. The PCT measurements may be used as a guide to the antibiotic therapy in critically ill children with suspected sepsis.
[1]. Ferriere F, Procalcitonin, a new marker for bacterial infectionsAnn. Biol. Clin 2000 58:49-59. [Google Scholar]
[2]. Meisner M, Path biochemistry and clinical use of procalcitoninClin. Chem. Acta 2002 323:17-29. [Google Scholar]
[3]. Levy MM, Fink MP, Marshall JC, Abraham E, Angus D, Cook D, SCCM/ESICM/ACCP/ATS/SIS. International Sepsis Definitions ConferenceCrit. Care Med 2003 31:1250-56. [Google Scholar]
[4]. Korczowski B, Procalcitonin – a new acute phase parameterPediatria Polska. LXXIII 1998 2:95-100. [Google Scholar]
[5]. Monneret G, Labaune JM, Isaac C, Bienvenu F, Putet G, Bienvenu J, Procalcitonin and C-Reactive Protein levels in neonatal infectionsActa. Paediatrica 1997 8:857-61. [Google Scholar]
[6]. Bang AT, Bang Ra, Reddy MH, Baitule SB, Deshmukh MD, Oaul VK, Burden of morbidities and unmet need for health care in rural Indian neonateInd. Pediatr 2001 38:952-65. [Google Scholar]
[7]. WHOPerinatal mortalityReport No: WHO/FRH/MSM/967 1996 GenevaWHO [Google Scholar]
[8]. Andrejaitiene J, The diagnostic value of procalcitonin in severe sepsis MedicinaKaunas 2006 42:69-78. [Google Scholar]
[9]. Garey KW, Rege M, Pai MP, Mingo DE, Suda KJ, Time to initiation of fluconazole therapy impacts mortality in patients with candidemia: a multi-institutional studyClin Infect Dis 2006 43:25-31. [Google Scholar]
[10]. Won H, Rothman R, Ramachandran P, Hsieh JH, Kecojevic A, Rapid Identification of Bacterial Pathogens in Positive Blood Culture Bottles by Use of a Broad-Based PCR Assay Coupled with High-Resolution Melt AnalysisJ. Clin. Microbiol 2010 48:3410-13. [Google Scholar]
[11]. Rey C, Los Arcos M, Concha A, Procalcitonin and C-reactive protein as markers of systemic inflammatory response syndrome severity in critically ill childrenIntensive Care Med 2007 33:477-84. [Google Scholar]
[12]. Kopterides P, Siempos II, Tsangaris I, Tsantes A, Armaganidis A, Procalcitonin- guided algorithms of antibiotic therapy in the intensive care unit: a systematic review and meta-analysis of randomized controlled trialsCrit. Care Med 2010 38:2229-41. [Google Scholar]