The Diagnostic Role of the Marginal Vacuoles in FNAC of Solitary Thyroid Nodules
Anshu Gupta1, Sonia Sharma2, Shivani Kalhan3, Atul Gupta4, Sharmila Dudani5, Amit Devra6
1 Associate Professor, Department of Pathology, Saraswathi Instt of Medical Sciences, Anwarpur, Hapur U.P., India.
2 Assisstant Professor, Department of Pathology, Army College of Medical Sciences, Delhi Cantt, India.
3 Professor, Department of Pathology, Saraswathi Instt of Medical Sciences, Anwarpur, Hapur, U.P., India.
4 Professor & Head, Department of Pathology, S.N.M.C., Agra, U.P., India.
5 Assistant Professor, Department of Pathology, Army College of Medical Sciences, Delhi Cantt. India.
6 Consultant Urologist and Transplant Surgeon Pushpawati Singhania Research, Instt, Sheikh Sarai, New Delhi, India.
NAME, ADDRESS, E-MAIL ID OF THE CORRESPONDING AUTHOR: Dr. Anshu Gupta, Associate Professor, Department of Pathology, R- 13/27, Raj Nagar, Ghaziabad, Pin-201002, U.P., India.
Phone: 09999665416
E-mail: amitanshu75@gmail.com
Context: FNAC thyroid is more sensitive as well as specific in segregating neoplastic and non-neoplastic thyroid lesions. Identification of predominant cell pattern, cell morphology and background details in FNAC smears play an important role in categorisation of various thyroid lesions with accuracy. Marginal vacuoles (MVs) have been described as irregular cytoplasmic vacuoles with largely unstained central area. MVs, although suggestive of thyrotoxicosis, are non-specific, as they are also encountered in non- toxic goitre, Hashimoto’s thyroiditis and follicular carcinoma. This cytologic finding therefore deserves further study to precisely delineate its utility.
Aims & Objective: To investigate whether MVs have a diagnostic role in FNAC of solitary thyroid nodule
Setting & Design: This study was conducted on 40 patients who presented with solitary thyroid nodule.
Methods & Material: Forty patients having solitary thyroid nodule were subjected to FNAC on an outpatient basis. Their cytomorphological features were studied with special attention to presence of MVs. MVs if present were then graded as no/ scant (gradeI ), moderate (gradeII) and abundant (gradeIII).
Results and Conclusion: Majority (77.5%) of nodular goitres had insignificant MVs (gradeI) in their smears. Significant MVs (gradeII+III) were limited to autoimmune thyroiditis presenting as solitary nodule and follicular neoplasms. Abundant MVs/ gradeIII were seen in follicular neoplasm only. MVs were not present in rest of neoplastic goitres. This implies that MVs are a marker of follicular differentiation.
Thyroid, Marginal Vacuoles, Solitary thyroid nodule
INTRODUCTION
FNAC of the thyroid gland is a first line diagnostic modality for the evaluation of goitre and it is the single most effective test for the preoperative diagnosis of the thyroid gland [1,2]. The recent studies have also confirmed that FNAC is more sensitive as well as specific in segregating the neoplastic and the non-neoplastic thyroid lesions [3]. The identification of the predominant cell pattern, the cell morphology and the background details in the FNAC smears play an important role in the categorization of various thyroid lesions with accuracy. Marginal vacuoles (MVs) have been described as irregular cytoplasmic vacuoles which are 1-7 μ in diameter deeply eosinophilic in the periphery, with largely unstained central areas (MGG).They are seen peripherally in a group of follicular epithelial cells and are thus called marginal vacuoles. The term ‘fire-flares’ has also been used because of the MVs’ resemblance to a rising or spreading fire. The MVs stain poorly or not at all with PAS and are not positive for the acid phosphatases. They are not as distinct in wet fixed, haematoxylin-eosin stained smears as in dry fixed MGG stained smears.
Ultra structural studies have suggested that MVs are the dilated cisternae of the endoplasmic reticulum, generated by hyperactivity and vacuolar content as dilated phagolysosomes [4,5]. Nilsson G et al., in 1972, were the first ones to discuss marginal vacuoles in the cytoplasm of the follicular cells in smears from toxic goitres. They reported that MVs were fairly specific signs of a thyroid hyperfunction [6]. However, subsequent studies have shown that MVs, although they are suggestive of thyrotoxicosis, are non-specific, as they are also encountered in non- toxic goitre, Hashimoto’s thyroiditis and follicular carcinomas. It has also been concluded that MVs are not only limited to follicular neoplasms but that they may be even more frequent in the follicular variant of papillary carcinomas [7]. MVs are said to represent the diffusion of the thyroid hormones from the basal aspect of the follicular cells, on their way to the interfollicular capillaries [8]. This cytologic finding therefore deserves further study to precisely delineate its utility.
AIMS AND OBJECTIVES
To evaluate the significance of MVs in the cytodiagnosis of solitary thyroid nodules.
MATERIALS AND METHODS
This study was conducted on 40 cases of solitary thyroid nodules. FNAC was done by using a standard procedure on an out patient basis . Air dried smears were prepared and they were stained with the MGG stain. The adequacy of the samples was assessed by using the Hamburger criteria, that is 6 clusters of cells in at least 2 slides which were prepared from separate aspirates [9]. The cytological diagnoses were arrived at after studying the predominant cell pattern, the background and the presence of MVs. The cases were initially categorized into those of non neoplastic and neoplastic pathologies. Thereafter, they were assigned to specific cytological diagnoses, based on their cytomorphologies. The MVs were studied and graded (loosely based on the criteria which were given by Nilsson G.et al., [10] as per the following criteria:-
No/scanty MVs/ (grade I) – Distinct MVs which exceeded 2μ in diameter were demonstrated in <10% of the cells
Moderate MVs /(grade II) – MVs seen in 10-50% of the cells which were examined
Abundant MVs/ (grade III) – the presence of MVs in >50% of the cells which were examined.
RESULTS
A detailed and a comprehensive study of 40 cases of solitary thyroid nodules was undertaken. The cytomorphological features were studied, with special attention to the presence and the grading of MVs. There were 32 female subjects (80%) and 8 male subjects (10%). The male to female ratio was 1:4. This study revealed a female preponderance in all the cases, ranging from 1:3 in follicular neoplasms to 3:8 in papillary carcinomas. The mean age of the patients in our study was 39.5 ± 13.5 years, with the maximum number of cases belonging to the 4th and 5th decades. The bulk of the solitary nodules in the current study comprised neoplastic aetiologies [29 out of the 40 cases (72.5%)], while non-neoplastic aetiologies accounted for 11 out of the 40 cases (27.5%) [Table/Fig-1].
Correlation of MVs with cytological diagnosis
| Cytological diagnosis | Abundant MVs/grade III | Moderate MVs/gradeII | No/scant MVs/gradeI | Total (%) |
|---|
| Primary hyperplasia | 1 | 0 | 0 | 1 (2.5%) |
| Hashimoto’s thyroiditis | 1 | 1 | 1 | 3 (7.5%) |
| Colloid goitre | 0 | 0 | 5 | 5 (12.5%) |
| Thyroid cyst | 0 | 0 | 1 | 1 (2.5%) |
| Follicular neoplasm | 3 | 2 | 10 | 15 (37.5%) |
| FN/HN | 0 | 0 | 2 | 2 (5%) |
| Papillary carcinoma | 0 | 1 | 10 | 11 (27.5%) |
| SCC | 0 | 0 | 1 | 1 (2.5%) |
| Anaplastic carcinoma | 0 | 0 | 1 | 1 (2.5%) |
| Total | 5 | 4 | 31 | 40 |
| Percentage(%) | 12.5% | 10% | 77.5% | 100% |
Follicular neoplasms accounted for 15 out of the 40 (37.5%) cases, followed by papillary carcinomas which accounted for 11 out of the 40 cases (27%). A majority (77.5%) of the solitary thyroid nodules which were examined, had insignificant (grade I) MVs in their smears. Moderate to abundant (grade II+ grade III) MVs were limited to autoimmune thyroiditis in the non – neoplastic category and to follicular neoplasms amongst the neoplastic group.
Moderate + abundant MVs/grade II +III were observed in 5 out of 15 cases (30%) of the follicular neoplasms [Table/Fig-2]. Coming to papillary carcinomas, only a solitary case (1 out of 11; 9%) was endowed with moderate /grade II MVs [Table/Fig-3]. They were not observed in the rest of the neoplastic pathologies i.e. squamous cell carcinomas and anaplastic carcinomas. Non-neoplastic lesions presenting as nodules accounted for 11 out of the 40(27.5%) cases. Most (5 out of 11 cases;45.5%) of these turned out to be colloid goitres on cytological examination, and all of them were bereft of MVs. Moderate + abundant/ grade II + III MVs were observed in one case of primary hyperplasia and in 2 out of 3 cases of autoimmune thyroiditis only.
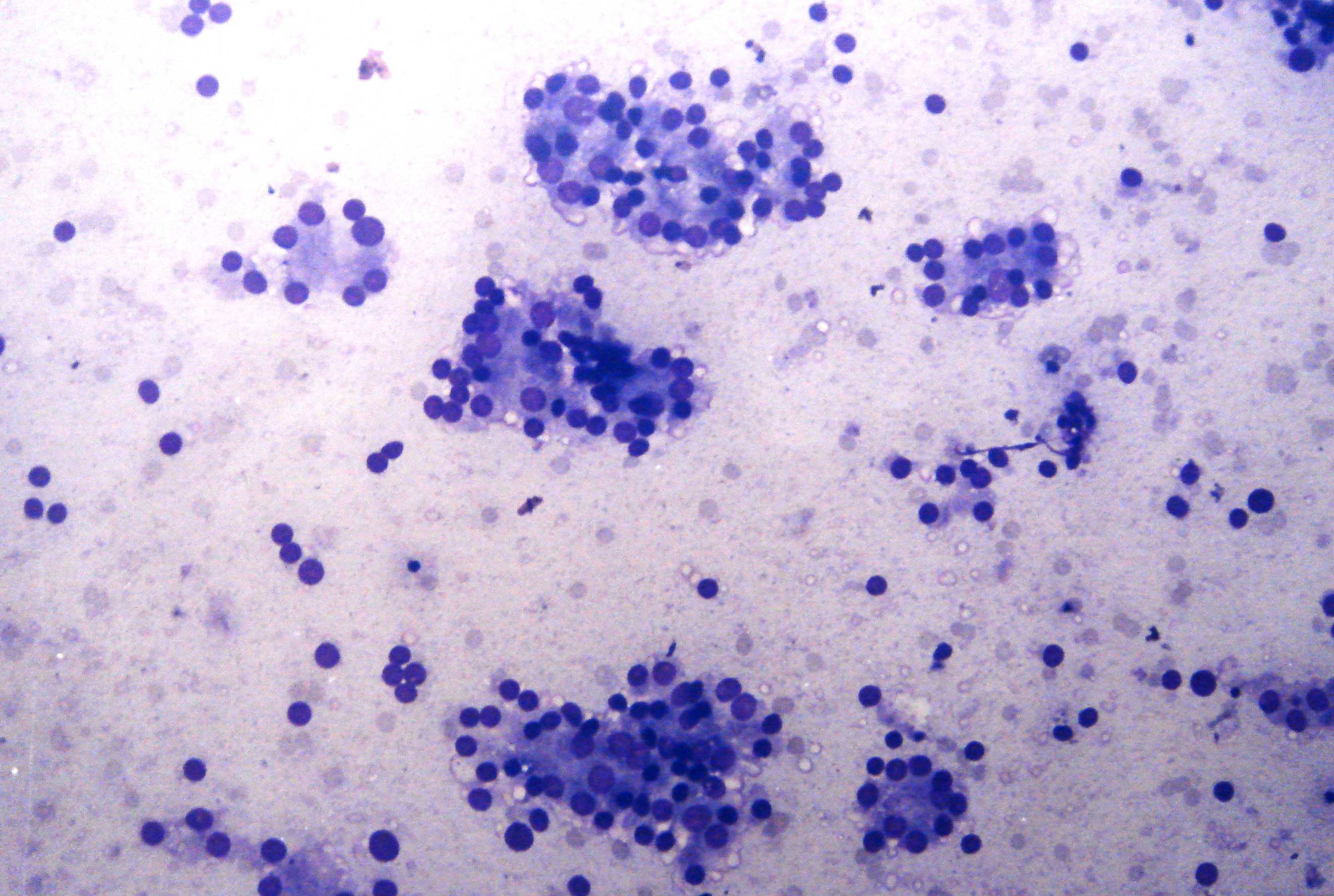
Follicular neoplasm with abundant MVs (MGG,200X)
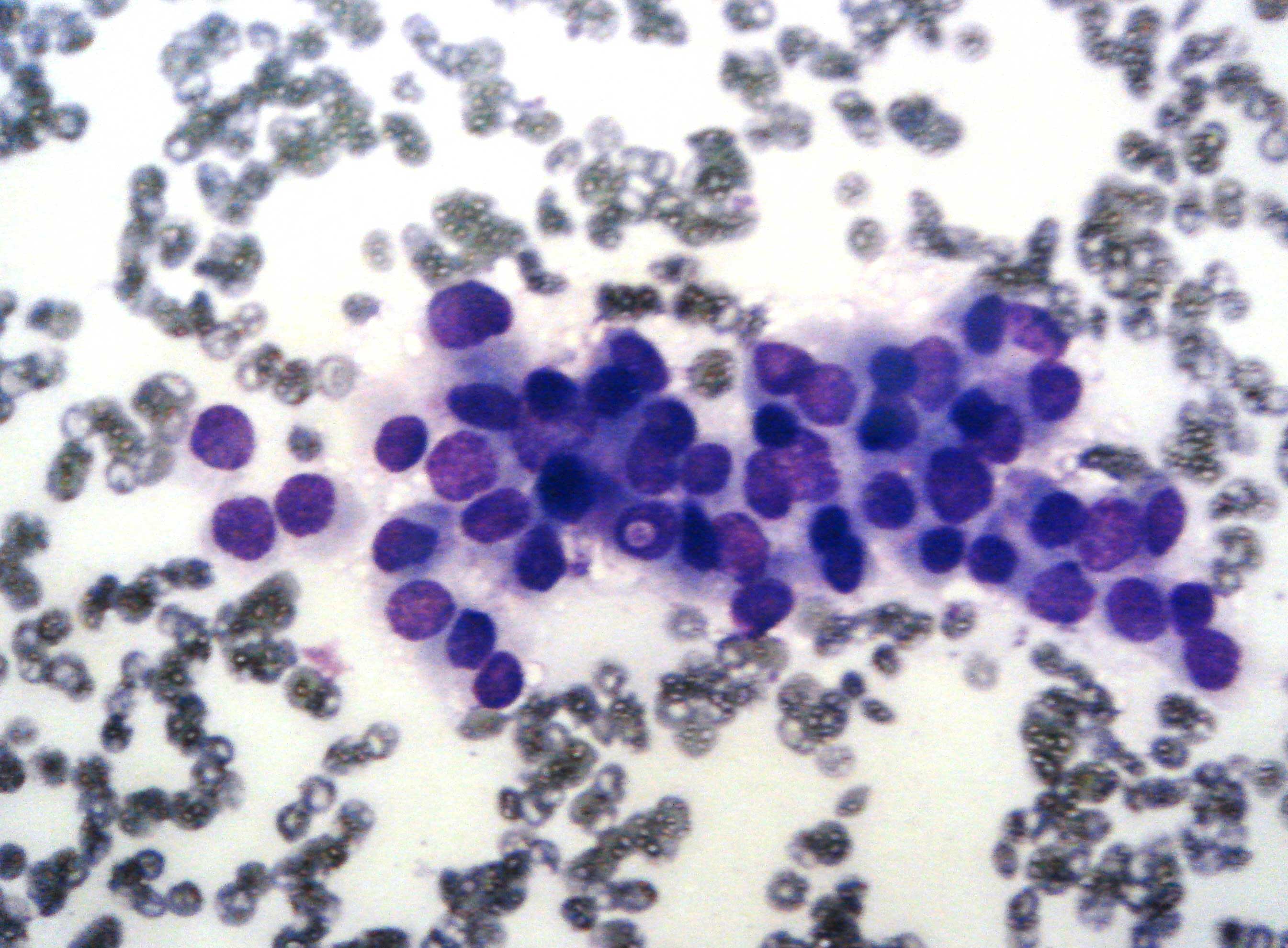
Papillary carcinoma showing intranuclear inclusions and MVs (MGG,400X)
DISCUSSION
A detailed study of 40 cases of solitary thyroid nodules was undertaken. The cytomorphological features were studied with special attention to the presence and the grading of MVs. The presence of prominent MVs, along with a nuclear ring pattern in the follicular wall fragments, are fairly the specific signs of a thyroid hyperfunction [6], but their frequency can also be high in Hashimoto’s thyroiditis, cellular adenomatoid nodules and in follicular neoplasms [7]. It has been suggested that MVs represent the diffusing out of thyroid hormones from the basal aspect of the follicular cells [8]. In the current study, moderate + abundant MVs/ grade II +III were noted in [9] out of the 40 cases(22.5%), whereas, Das et al noted these in 14.7% of the cases [7]. Notably, none of the 5 cases of nodular colloid goitres which presented as solitary nodules had significant MVs in our study. Correspondingly, Das et al., noted the occurrence of MVs in 5.2% cases of nodular colloid goitres [7]. Amongst the inflammatory pathologies which presented as solitary nodules, 66.7% showed moderate + abundant MVs/ grade II +III. The same were observed in 30% of the cases in the literature [7]. Therefore, the frequency of MVs in the cases of inflammatory thyroid pathologies which presented as nodules, was substantially higher in the present study.
5 out of 15 cases (33.3%) of follicular neoplasms showed moderate + abundant MVs/ grade II +III MVs. This figure was comparable to that which was observed by Das et al., (26.7%), but it was lower than that which was observed by Agarwal et al., (57%) [7,11]. We found it difficult to categorize 2 of our cases into specific cytological categories i.e. hyperplastic thyroid nodules versus follicular neoplasms. We observed MVs in one case out of these two. Two cases of a Follicular Variant of Papillary Thyroid Carcinoma (FVPTC) were encountered. A thorough examination of the smears of these two cases showed no MVs [Table/Fig-4]. This was in contrast to the figures which were quoted in the literature (44- 50%) [12,7]. This disparity may have been due to the small sample size of our study.
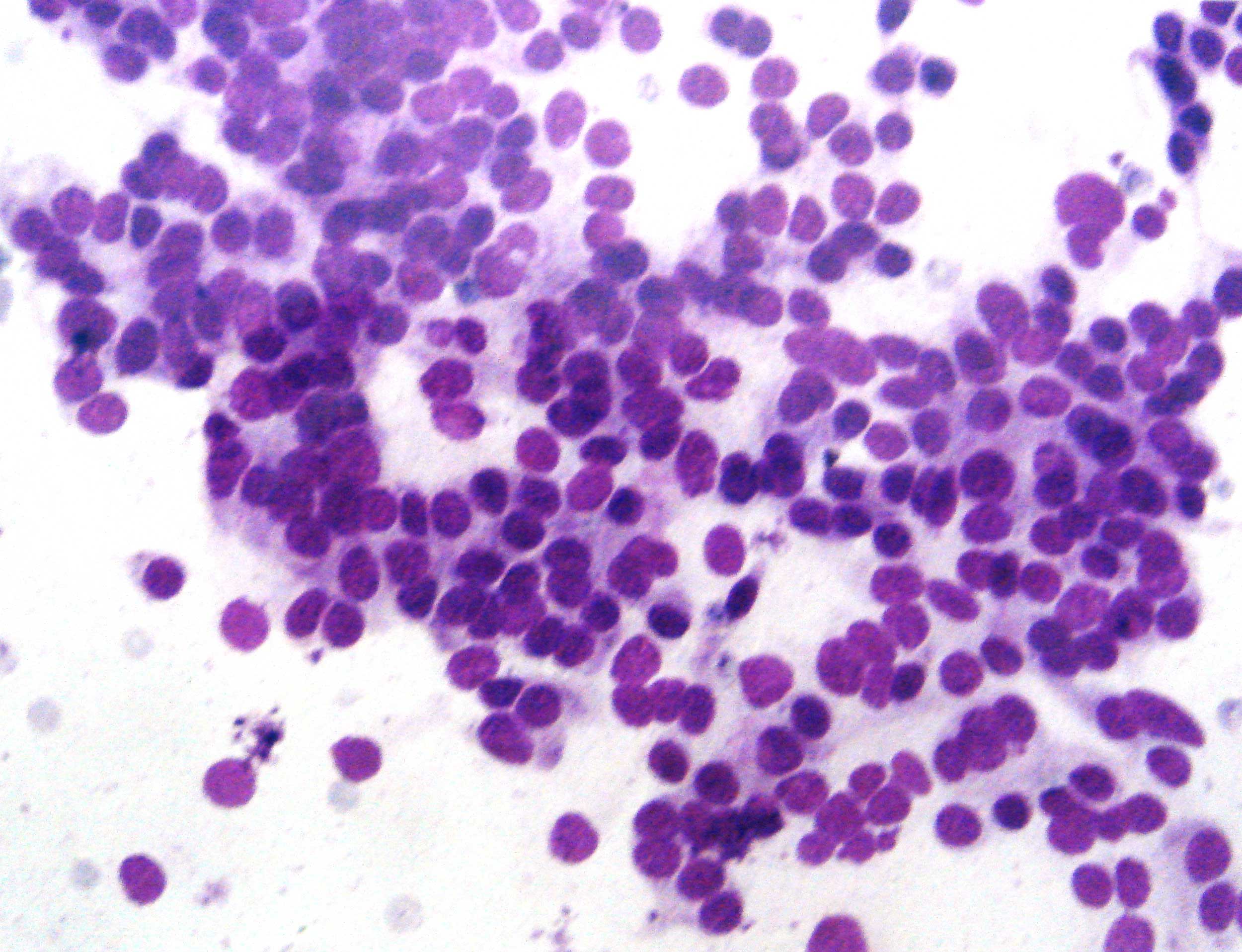
Follicular variant papillary carcinoma without MVs (MGG,400X)
Previous studies have brought forth the fact that MVs are not present in aspiration cytology smears of usual papillary carcinoma. In the present study, however, one out of eleven cases did show moderate/grade II MVs [7]. The fact however remains that most of the cases of usual papillary thyroid carcinomas are bereft of MVs. Also, no MVs were encountered in the 2 cases of anaplastic carcinomas and a single case of squamous cell carcinoma of the thyroid which we came across in our study. These lesions have not been studied by other workers with respect to MVs. Therefore, amongst the neoplastic nodules which were examined in the present study, MVs were limited to the follicular neoplasms only, except for a solitary case of papillary thyroid carcinoma.
SUMMARY AND CONCLUSION
The aspiration smears of forty cases of solitary thyroid nodules were studied, with special attention to the presence of MVs. A majority of the nodules in this study were neoplastic in nature, with follicular neoplasms topping the list (37.5%), out of which 30% had moderate+abundant/grade II+III MVs in their smears. It was therefore concluded that a significant proportion of follicular neoplasms have significant MVs. On the other hand, MVs were found infrequently in papillary carcinomas in this study. MVs were not encountered in the rest of the remaining neoplasms which were encompassed in the present study i.e. anaplastic and squamous carcinomas. This implies that MVs are a marker of the follicular differentiation. However, keeping the limited sample size of the present study in mind, it is recommended that further studies be done to corroborate these conclusions.
[1]. Orell SR, Sterrett GF, Whitaker D, Fine Needle Aspiration Cytology 2005 4thElsevier Churchill Livingstone [Google Scholar]
[2]. Ko HM, Jhu IK, Yang SH, Lee JH, Juhng SW, Choi C, Clinicopathologic analysis of fine needle aspiration cytology of thyroid. A review of 1,613 cases and correlation with histopathologic diagnosisActa Cytol 2003 47:727-32. [Google Scholar]
[3]. Bommanahali BP, Bhat RV, Rupanarayan R, A cell pattern approach to interpretation of fine needle aspiration cytology of thyroid lesions. A cyto histomorphological studyJ Cytol 2010 27:127-32. [Google Scholar]
[4]. Ramzy S, Clinical cytopathology and aspiration biopsy 1990 New YorkAppleton and Lange:273-93. [Google Scholar]
[5]. Davidson HG, Diagnostic problem in thyroid FNADiagn Cytopathol 1997 17:422-701. [Google Scholar]
[6]. Soderstrom N, Nilsson G, Cytologic diagnosis of thyrotoxicosisActa Med Scand 1972 205:263-65. [Google Scholar]
[7]. Das DK, Jain S, Tripathi RP, Parkash S, Khan IU, Rajwanshi Marginal vacuoles in thyroid aspiratesActa Cytol 1998 42:1121-28. [Google Scholar]
[8]. Das DK, Marginal vacuoles (fire-flare appearance) in fine needle aspiration smears of thyroid lesions. Does it represent diffusing out of thyroid hormones at the base of follicular cellsDiagn Cytopathol 2006 34:277-83. [Google Scholar]
[9]. Hamburger JI, Husain M, Semiquantitative criteria for fine needle biopsy diagnosisDiagn Cytopathol l998 4:14-17. [Google Scholar]
[10]. Nilsson G, Marginal Vacuoles in FNAB smears of toxic goitresActa Pathol Microbiol Scand 1972 80:289-93. [Google Scholar]
[11]. Agarwal SK, Jayaram G, Kakar A, Prakash R, Pant CS, Fine needle aspiration cytologic diagnosis of the solitary cold thyroid nodule, comparison with ultrasonography, radionucleotide perfusion study and xeroradiographyActa Cytol 1989 33:41-47. [Google Scholar]
[12]. Gallagher J, Oertel YC, Oertel JE, Follicular variant of papillary carcinoma of thyroidDiagn. Cytopathol 1997 16:207-13. [Google Scholar]