Introduction
Tuberculosis is the commonest of the infections world wide and it can affect almost any part of the body, most commonly the thorax. The spine is affected in 50 % of the cases of skeletal tuberculosis. A tuberculous infection of the spine causes a bony destruction and collapse of the vertebrae, with a gibbus deformity, skip lesions, an intervertebral disc involvement, an epidural abscess, a paravertebral abscess and oedema in the soft tissue planes. Magnetic Resonance Imaging (MRI) is the most valuable investigation in the patients with spinal tuberculosis, as it can clearly demonstrate all of the above findings.
Methods
In this study, the MRI scans of 70 known cases of tuberculosis of the spine, which were done in the Department of Radiodiagnosis, R.D.Gardi Medical College, Ujjain, India, were retrospectively analyzed, to determine the pattern of occurrence of various pathological lesions.
Results
It was found in this study, that Pott’s spine was most commonly observed in the 21-50 years age group, with a male predominance. The dorsal and the lumbar vertebrae are commonly involved and multiple vertebrae were often affected, the L3 vertebra being the commonest. An intervertebral disc involvement and pre and paravertebral collections were commonly seen, with an epidural collection occurring in more than 75 % of the cases. Cord oedema was noted in 10% of the cases.
Conclusion
The MRI scan is highly sensitive in the detection of various pathological processes of Pott’s spine and the patterns of occurrence of these findings were analyzed in this study. Since the incidence and prevalence of tuberculosis are dependent on various epidemiologically sensitive parameters, this study can provide a benchmark, against which the results of studies which will be done in the future can be compared.
Introduction
A tubercular infection of the spine or tubercular spondylitis or Pott’s disease was first described by Percival Pott in 1779 and it has been detected in the ancient mummies in Egypt and Peru. It is caused by the Mycobacterium tuberculosis bacteria, most commonly by a haematogenous spread of the infection [1] via the venous plexus of Batson [2]. According to the World Health Organization, in 2006, nearly 2 billion people, about 1/3rd of the world’s population had tuberculosis and it has been reported that annually 6 million become ill and 2 million die from this disease worldwide. It remains the prime infection which causes high morbidity and mortality in the densely populated developing countries. The incidence of tuberculosis had declined in the western world, but in recent years, it has shown resurgence in these nonendemic populations. There has also been an increase in its global prevalence, particularly in immunocompromised patients, with a rate of increase of approximately 1.1% per year [2]. The main causes of the increase in the occurrence of tuberculosis world wide, appears to be the Human Immunodeficiency Virus (HIV) epidemic, poor nutrition, drug addiction, alcoholism, appearance of the drug-resistant strains of tuberculosis, ineffective tuberculosis control programs, over crowding and increased migration [3,4]. About 10.7 million people worldwide have both the HIV and the tuberculosis infections. The occurrence of tuberculosis is thus closely related to epidemiologically sensitive parameters and these parameters can alter the occurrence of various pathological processes of tuberculosis. The infectious process in the vertebra usually starts in the cancellous bone of the vertebral body, anteriorly under the periostium, later extending to the intervertebral disc and to other parts of vertebrae. The destructive process which involves the bone leads to a collapse of the body of the vertebra, along with its wedging. The infective process generates an immune response which causes the formation of granulation tissue and necrotic material which collect within the lytic areas of the bone and in the surrounding soft tissue planes. The infective process can extend directly into the psoas muscle, leading to an abscess formation. The presenting symptoms of Pott’s spine include low-grade fever with an evening rise, lethargy, malaise, night sweats, anorexia and weight loss, which are common to any tubercular infection [4]. The symptoms which are characteristic of Pott’s spine consist of local tenderness and limitations of the motion and in the late phases of the disease, severe spinal deformity (acute kyphotic angulation). The worst complications of tuberculosis of the spine are para or quadriplegia, hemiplegia or monoplegia, with an incidence of paraparesis which varies from 27-47% [4]. Tuberculosis is the most common cause of non-traumatic paraplegia in the most parts of world [4,5]. The neurological symptoms tend to occur in the acute as well as the late stages of the disease. The neurological symptoms include radicular pain, the severe cauda equina syndrome and spinal cord compression. They may result from oedema, vascular engorgement, vertebral collapse, retropulsed debris, meningomyelitis or subarachnoid collections [3]. The multiplanar images and the better tissue contrast make MR imaging the modality of choice in the evaluation and the follow-up of Pott’s spine [2]. It shows not only a bony involvement but also oedema and soft tissue swelling, with serial scans being used to assess the response to the treatment. Despite its advantages, one of the greatest limitations of MRI is its inability to identify the soft tissue calcification on the routine scans, which is characteristic of the tubercular infection. Moreover, it is difficult to perform a guided percutaneous aspiration and a biopsy on an MRI scanner. The MRI appearance of tuberculosis mimics various other pathological processes and these can be categorized into infectious and neoplastic lesions. The major differential diagnoses include low grade infections like atypical mycobacteria, brucella and fungal infections and neoplasms like lymphomas, multiple myelomas and metastases. These infections generally involve the intervertebral discs while they are spared in the neoplasms [3].
Materials and Methods
The aim of this study was to understand the pattern of occurrence and to analyze the various pathological processes of Pott’s spine by using the MRI scan. This study was done at a tertiary care hospital, R.D.Gardi Medical College, Ujjain, MP, India which is located in the state of Madhya Pradesh in central India, which predominantly caters to the rural population. MRI scans of all the proven cases of Pott’s spine, which were done in the Department of Radiodiagnosis from April 2009 to April 2012, were retrospectively studied by two radiologists separately and the discrepancies were later resolved by reaching a consensus. This study included 70 cases in which tuberculosis was histopathologically proven either before the MRI scan or after a suspicion of Pott’s spine was raised following the scan. The patients of all age groups and both sexes were included in the study, while repeat scans of the same patient were excluded. The scans were done on a 1.5T GE HDE scanner by using T1 Weighted spin echo (T1W) and T2 weighted fast spine echo (T2W) sequences in the sagittal and the axial planes and a fat Suppressed Inversion Recovery (STIR) sequence in the coronal plane. The bony structures were identified and any collapse or destruction which was characterized as a hyperintensity on the T2W sequence and as a hypointensity on the T1W sequence was noted [Table/Fig-1]. The marrow oedema appeared as a low signal on the T1W sequence and as a hyperintensity on the STIR sequence. The posterior margins of the vertebral bodies were viewed in detail for any posterior displacement or a bony fragment in the epidural space [Table/Fig-2]. The CSF column was traced for any collection in the epidural space, which appeared as a hypointensity on the T1W sequence with heterogeneity, suggesting the presence of necrotic material and granulation tissue which caused elevation of the dura and compromise of the subarachnoid space. The extent of this collection was noted and more than a 75% reduction in the thickness of the CSF column was considered as significant and as causing neural compression. A similar collection in the pre/paravertebral planes and in the psoas muscles suggested an abscess and screening of the whole spine was done to identify the skip lesions [Table/Figs-3A and B]. The posterior bony component was meticulously visualized for the presence of marrow oedema or destruction [Table/Fig-3C]. The cord oedema was identified as a hyperintensity on the T2W sequence and an alteration in the curvature was noted [Table/Fig-2]. Tuberculosis is characteristically associated with little or no reactive sclerosis or local periosteal reaction, a feature that helps in distinguishing it from pyogenic infections of the spine [2]. A CT or ultrasound guided aspiration of the paravertebral collection or the pathological bony tissue was obtained and sent for culture. The sample was also histopathologically examined for caseating granulomas, along with the detection of acid fast bacilli on Ziehl Neelsen’s staining [3]. If the above investigations were negative, we excluded such cases from our study. The major limitation of the pathological examination is that in many cases of tuberculosis, the above findings are absent and in such cases, the polymerase chain reaction could be positive.
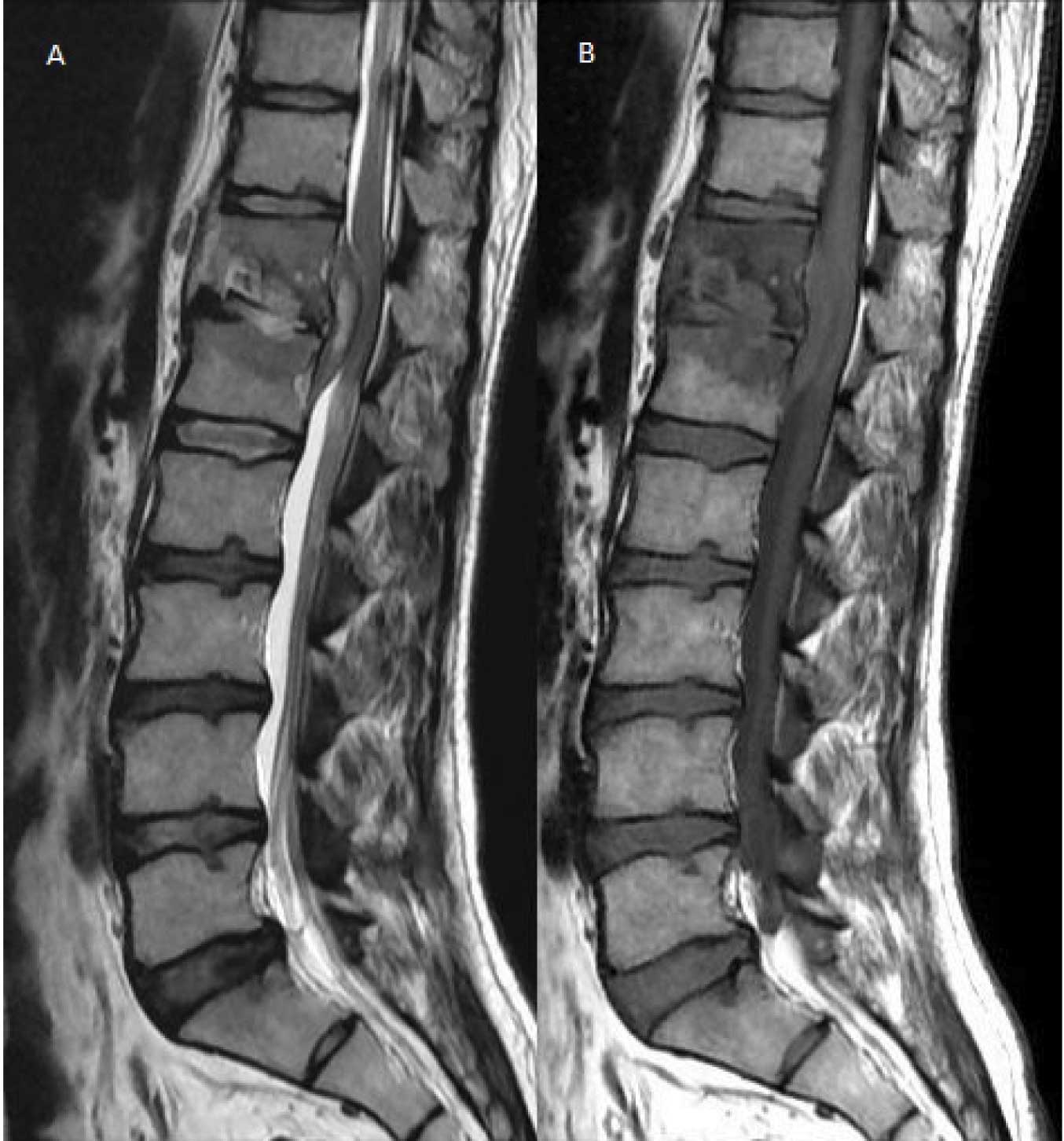
T2W (A) and T1W (B) images in sagittal plane showing destruction and erosion of end plates of D12 and L1 vertebral bodies with involvement of D12-L1 disc, prevertebral and epidural collection causing neural compression. The necrotic component of epidural collection appears as hyperintensity in core of T2W images (A) with isointense peripheral granulation tissue.
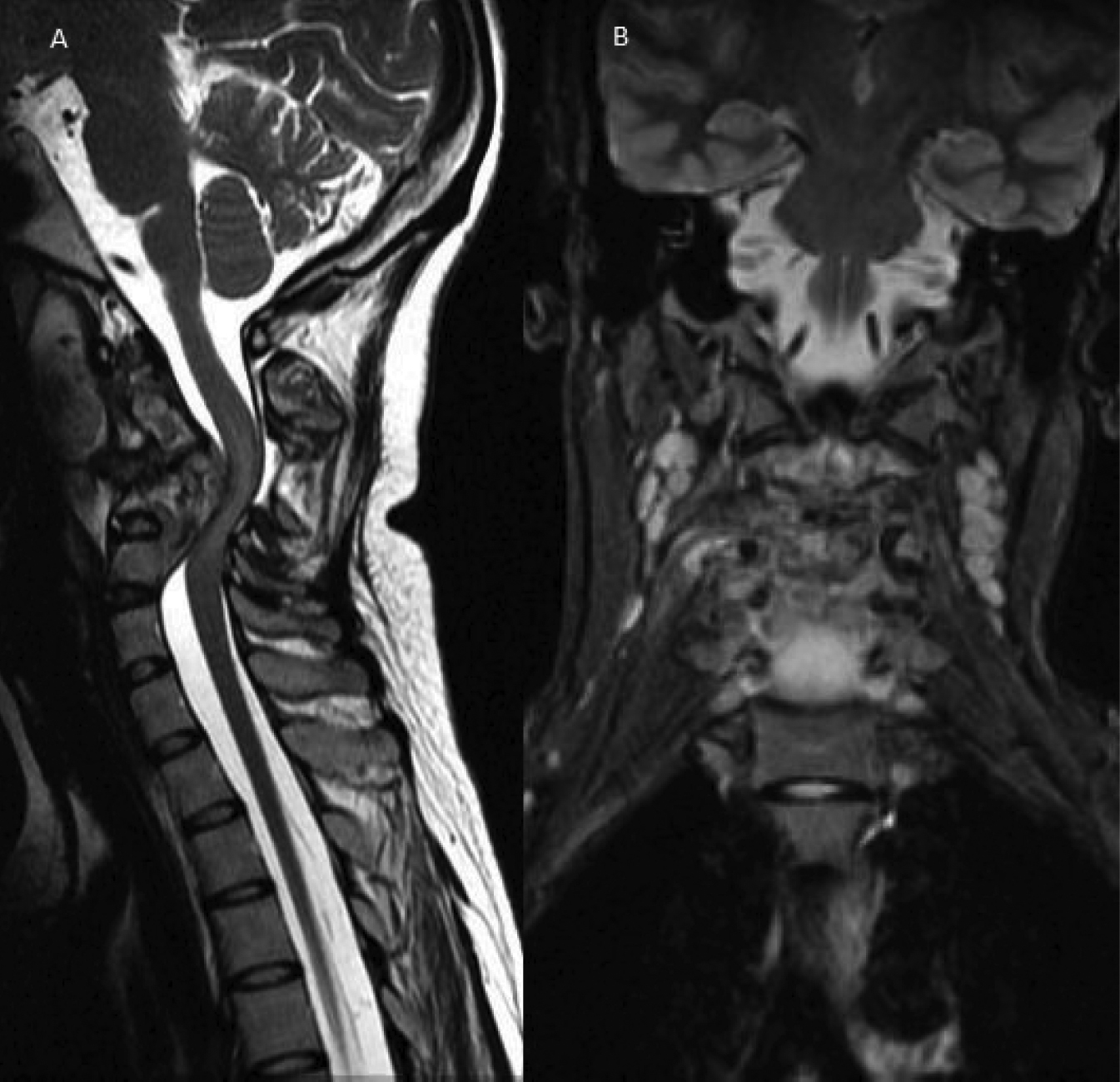
T2W sagittal (A) and STIR coronal (B) images showing destructive lesion in cervical vertebral column with large prevertebral and minimal epidural collection with gibbus causing kinking of cord and cord edema appearing as bright signal in T2W images (A). Lymphadenopathy is noted in planes of neck on STIR image as round bright lesions (B).
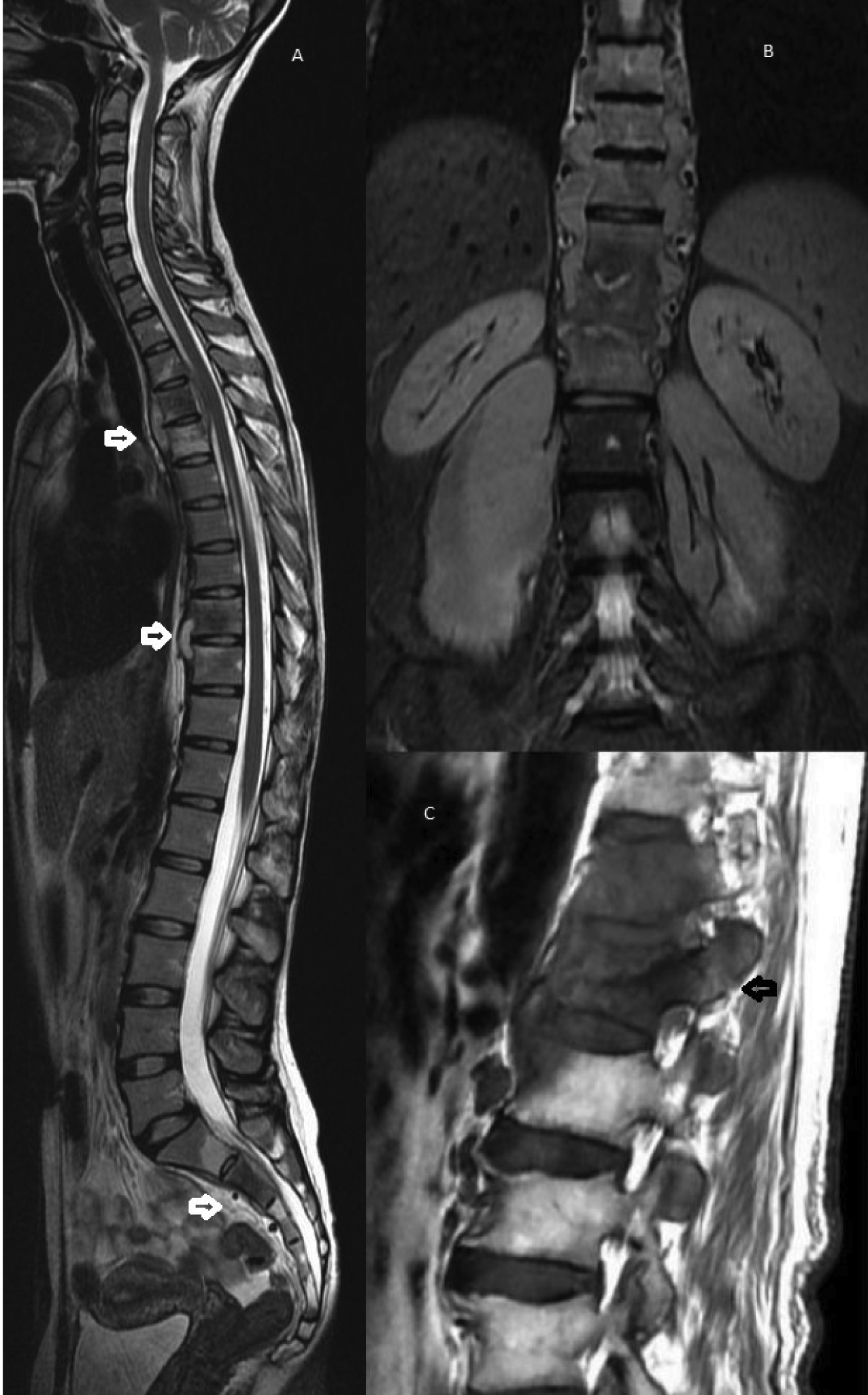
Skip lesions of Pott’s spine in T2W sagittal image (A) in dorsal region and sacrum (arrows). STIR coronal image (B) showing multiple dorso-lumbar vertebrae involvement of paravertebral and psoas abscess bilaterally. T1W parasagittal image (C) showing posterior bony component (arrow) involvement.
Observations and Results
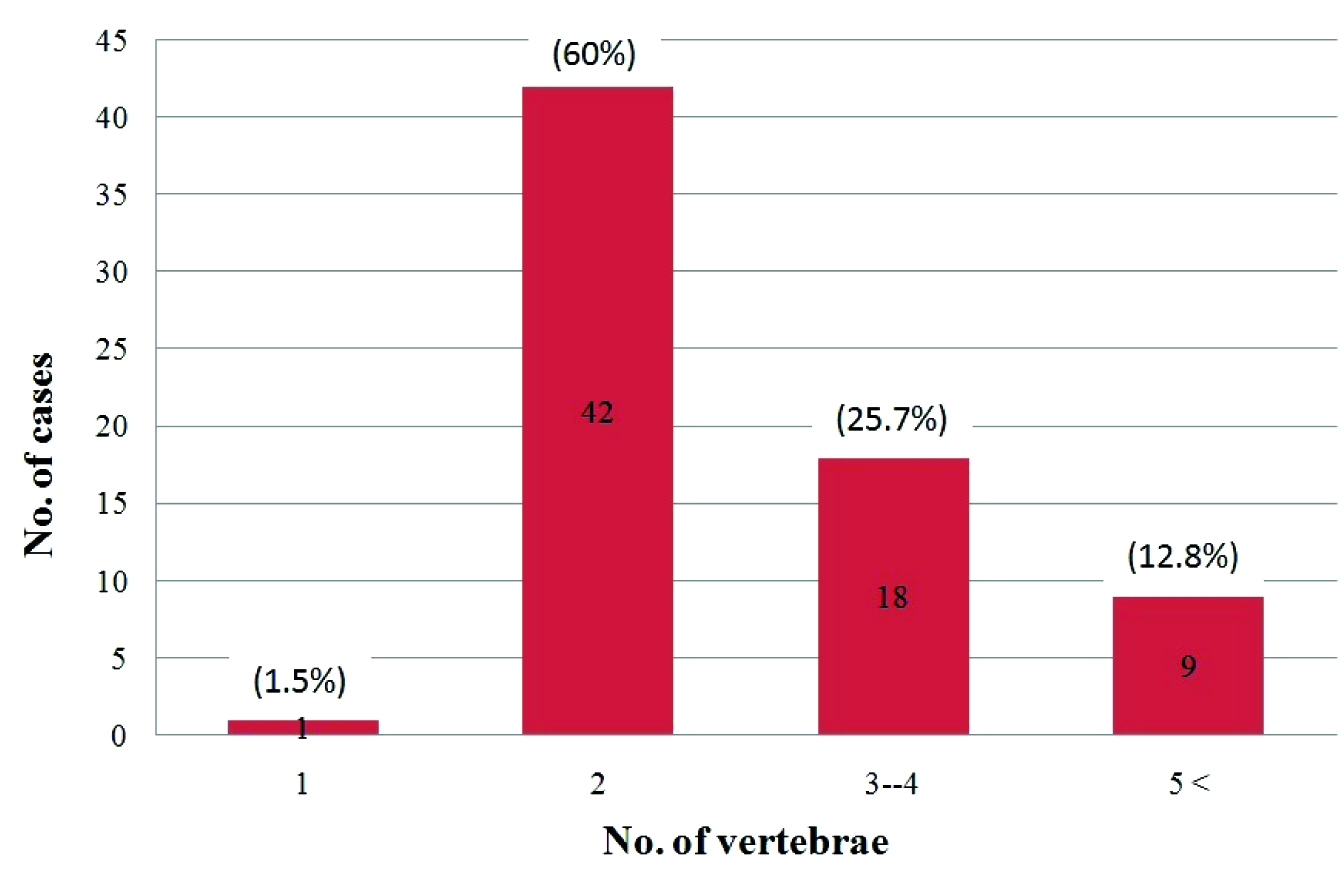
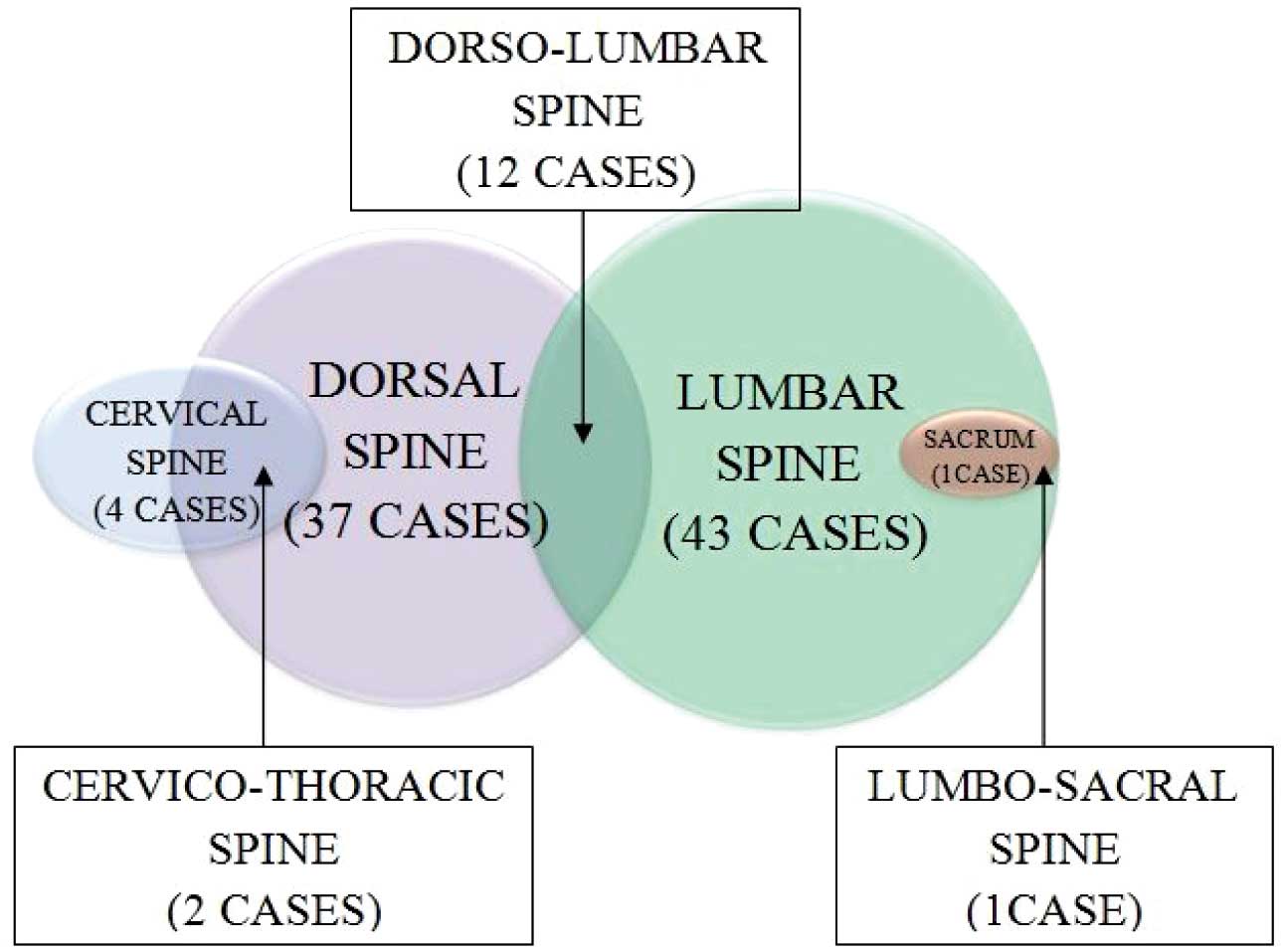
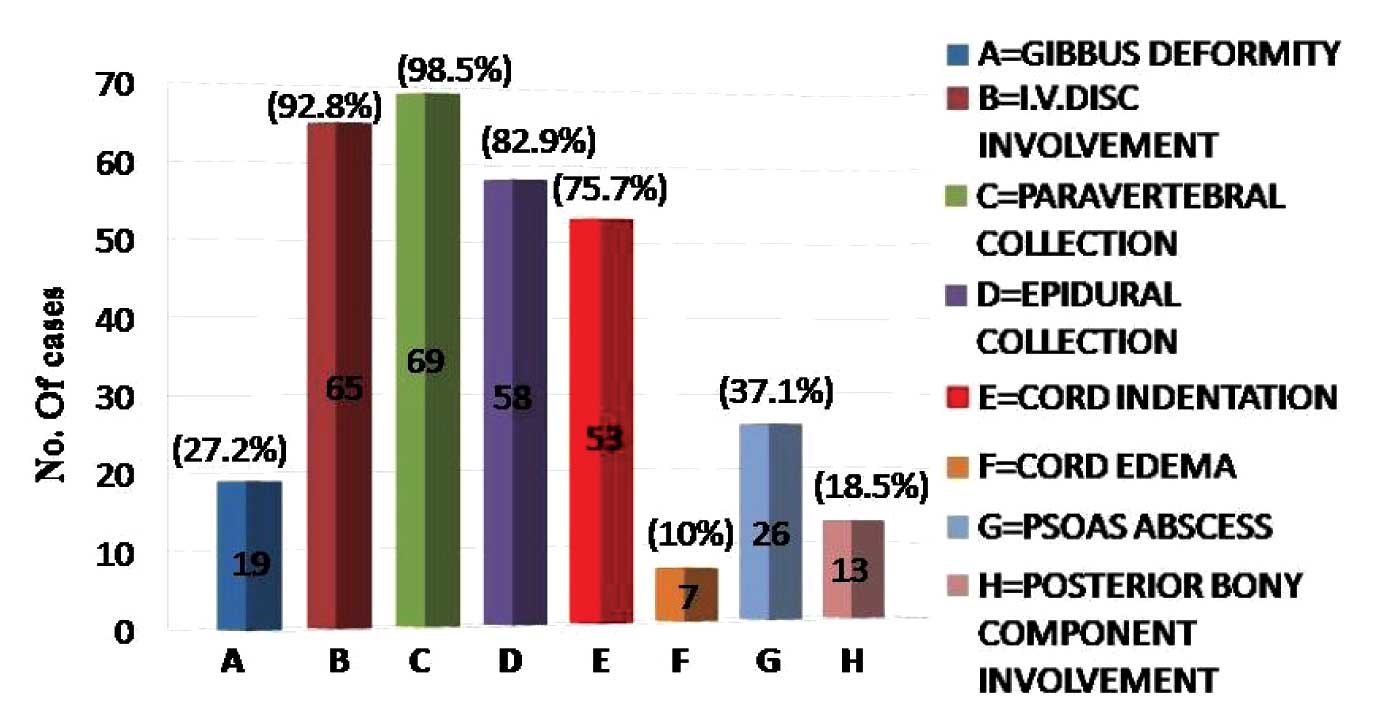
70 cases of Pott’s spine were included in this study, out of which 39 (55.7%) were males. 31 (44.28%) patients were between 21 to 40 years of age with a female predominance, while 45 (64.2%) cases were in the age group of 21 to 50 years [Table/Fig-4]. Multiple vertebrae were affected in most of the cases and a majority had 2 vertebrae involved (60%), followed by the involvement of 3-4 vertebrae (25%), while a single case had a single vertebra involved [Table/Fig-5]. In 37 (52.8%) cases, the dorsal and in 43 (61.4%)cases, the lumbar vertebrae were involved. Among these, 12 cases had both the dorsal and the lumbar vertebrae involvement. Two cases of dorsal vertebrae involvement were in association with the cervical vertebrae and 1 case of the lumbar vertebra was involved in association with the sacrum [Table/Fig-6]. The cervical vertebrae were involved in a total of 4 cases, out of which 3 cases were below 30 years of age. Skip lesions were noted in 2 (2.85%) cases. The vertebral body which was affected in a majority of the cases was L3 (34.2%). An alteration in the curvature, mostly a gibbus deformity which was due to the collapse of the vertebrae was noted in 19 (27.2%) cases. An intervertebral disc involvement was a common finding which was noted in 65 (92.8%) cases. A pre or paravertebral collection was seen in all cases except 1 case (98.5%), while an epidural collection was present in 58 (82.9%) cases [Table/Fig-7]. This epidural collection caused thecal sac indentation in 51 cases, while a cord compromise which was due to a gibbus deformity or posteriorly displaced bony fragments was noted in 16 cases. The combination of the gibbus and the epidural collections caused a neural compromise in 14 patients. The canal dimension was reduced by 75%, which was considered as severe. A neural compromise was noted in 15 (21.4%) cases. Cord oedema was appreciated in 7 (10%) cases. The Psoas abscess was noted in 26 (37.4%) cases, among which a bilateral involvement was seen in 10 patients. Among the 16 cases with the unilateral psoas abscess, 9 were on the right side and 7 were on the left side. The marrow oedema or the destructive process extended into the posterior bony components in 13 (18.6%) cases, among which 11 had involvement of the pedicle.
Age and sex distribution of cases of tubercular spondylitis
| AGE | <10 | 11-20 | 21-30 | 31-40 | 41-50 | 51-60 | 61-70 | 71< | Total |
| Male | 3 | 4 | 6 | 6 | 8 | 4 | 7 | 1 | 39 |
| Female | – | 2 | 12 | 7 | 6 | 2 | – | 2 | 31 |
| Total | 3 | 6 | 18 | 13 | 14 | 6 | 7 | 3 | 70 |
Bar chart showing distribution of number of cases and number of vertebrae infected.
Venn chart showing pattern of involvement of various segments of spine.
Bar chart showing pattern of occurrence of various pathological processes of tubercular spondylitis.
Discussion
Tuberculosis of the skeletal system constitute 1-5 % cases of tuberculosis, out of which 50% have vertebral column involvement [6]. Osborn [7] stated that in the developing countries it was prevalent in the younger age group, while in the western world, it was found in the middle age (mean 40-45 years). Sinan et al., [8] found 43% cases in their study in the age group of 30-49 years. In our study, most of the cases were between the ages of 21-50 years (mean 38.3 + 17.6), which was similar to the finding of Mirsaeidi et al., [9] who found a mean age of 39 + 16 years. Tuberculosis of the spine was found to be more common in males than in females in most of the previously published series [4,8–10], although Osborn [7] suggested no gender predilection. We noted that although overall Pott’s spine was more prevalent in males, in between 21-50 years of age in which Pott’s spine was the most prevalent, there was a female predominance (55%). In the 21-30 year age group, 66.6% of the patients were females [Table/Fig-4]. The mean age of the females in our study was 36.7 + 15.1 years and for the males, it was 40.5 + 19.4 years. This suggested that in the economically productive years of life, Pott’s spine was more prevalent in females. The lower thoracic and the upper lumbar levels were found to be most commonly affected in most of the studies [1,2,8,10]. In our study, the lumbar vertebrae were most commonly affected, followed by the dorsal vertebrae, which was similar to the findings of Sinan et al. [8]. The atypical patterns of the tuberculous involvement of the spine consisted of the infection of a single vertebra or of multiple nonadjacent vertebrae (skip lesions), which in our study were noted in 1.42% and 2.85% of the cases respectively. Stark et al. [1], suggested the presence of skip lesions in 4% of the cases, while in his study, Jain [11] suggested that multiple level tubercular lesions in the spine were observed in 16.3-71.4% of the cases when an MRI study of the whole spine was performed. We found skip lesions only in 2.85% of the cases. This could be because, for screening the whole spine, we had used a T2W sequence which was relatively less sensitive for detecting the minimal marrow oedema in comparison to the STIR and the T1W sequences. Osborn [7] stated that a paraspinal abscess was present in 55-95% of the cases, which in our study was noted in 98.5% of the cases. Sinan et al., [8] in their study, found the involvement of the intervertebral disc in 72% of the cases, while we found it in 92.8% of the cases. Gibbus, which occurs predominantly in the dorsal spine, was observed in 27.2% of the cases, which was reported only in one third of the cases in a study which was done in Africa [10]. Gibbus, which occurs late in the course of the illness in a large number of patients, is caused due to the late referral poor health care system in the rural set up of India, as in Africa. An epidural involvement was observed in over 80% of the cases [11], which was similar to the finding in our study (82.9%). Mirsaeidi et al., [9] found the psoas abscess in 14.3% of the cases, while it was noted in 37.1% of the cases in our study. Contrast enhanced MRI with the use of Gadolinium chelates, improves the delineation of the abscesses which appear as an enhancing periphery and a necrotic core [3]. A smooth enhancement of the wall and a discrete paraspinal enhancement suggests a tubercular infection, with the thick irregular enhancement being generally due to pyogenic infections. An enhancement along the nerve roots is suggestive of early arachnoiditis. In our study, we rarely did contrast enhanced MRI, as it significantly increases the cost and moreover, the information which is obtained does not significantly alter the management. Sequential MRI scans can suggest the response to the treatment of Pott’s spine. The appearance of sclerosis and the fatty changes in the bony tissue, the appearance of ankylosis, a reduction in the quantity of the epidural collection and the fibrosis and the resolution of the paraspinal signal changes highly favour a good response to the treatment. An increase in the quantity of the necrotic collection, an increase in the destructive process in the involved vertebrae with collapse and the appearance of a pathological process in a previously uninvolved vertebra suggests drug resistant tuberculosis and it warrants an alteration in the management protocol.
The cases which were included in this study were positive for tuberculosis on Ziehl Neelsen’s staining or on the culture of the aspirates which were obtained from the pathological tissues. However, in few cases of tuberculosis, these investigations were negative and so, some cases of tuberculosis of the spine were excluded from this study.
Conclusion
MRI is the most valuable method for detecting the early disease and it is the preferred technique for defining the activity and the extent of the infection. It can also act as a guide to the surgical treatment in cases of tuberculosis of the spine. The occurrence of various pathological lesions follow a characteristic pattern and they are likely to change with the variations in the predisposing factors for tuberculosis, like the prevalence of the HIV infection in the society, the nutritional status, etc. Hence, they should be monitored from time to time.
Acknowledgements
We thank the Medical Director of R. D. Gardi Medical College, Dr. V. K. Mahadik and the Dean, Dr J. K. Sharma for giving us permission to perform and to publish this study.
[1]. Stark DD, Bradley WG, Magnetic Resonance ImagingThoracic spine 1999 3rd edMosby:1851-82.Chapter 86 [Google Scholar]
[2]. Burrill J, Williams CJ, Bain G, Conder G, Hine AL, Misra RR, Tuberculosis: A Radiologic ReviewRadiographics 2007 27(5):1255-73.doi: 10.1148/rg.275065176 [Google Scholar]
[3]. De Backer AI, Mortelé KJ, Vanschoubroeck IJ, Deeren D, Vanhoenacker FM, Tuberculosis of the spine: CT and MR imaging featuresJBR–BTR [Internet] 2005 88(2):92-97.[cited 20 Feb 2012] Available from: http://www.rbrs.org/dbfiles/journalarticle_0256.pdf [Google Scholar]
[4]. Garg RK, Tuberculosis of the central nervous systemPostgrad Med J [Internet] 1999 75:133-140.[cited 20 Feb 2012] Available from: http://pmj.bmj.com/content/75/881/133.full [Google Scholar]
[5]. Bajwa GR, Evaluation of the role of MRI in spinal Tuberculosis: A study of 60 casesPak J Med Sci [Internet] 2009 25(6):944-947.[cited 20 Feb 2012] Available from: http://www.pjms.com.pk/issues/octdec209/pdf/14.article13.pdf [Google Scholar]
[6]. Kalita J, Misra UK, Mandal SK, Srivastava M, Prognosis of conservatively treated patients with Pott’s paraplegia: logistic regression analysis.J Neurol Neurosurg Psychiatry [Internet] 2005 76:866-68.[cited 28 Apr 2012] doi: 10.1136/jnnp.2004.037051 [Google Scholar]
[7]. Osborn AG, Diagnostic NeuroradiologyNonneoplastic disorders of the spine and spinal cord 2009 1e edElsevier:820-75.Chapter 20 [Google Scholar]
[8]. Sinan T, Al-Khawari H, Ismail M, Ben-Nakhi A, Sheikh M, Ann Saudi Med [Internet] 2004 [Cited 20 May2012] 24(6):437-41.Available from: http://www.kfshrc.edu.sa/annals/articles/24_6/03-359.pdf [Google Scholar]
[9]. Mirsaeidi SM, Tabarsi P, Amiri MV, Mansoori SD, Bakhshayesh-Karam M, Masjedi MR, Clinical and Radiological Presentation of Adult Tuberculous SpondylitisTanaffos [Internet] 2003 2(6):59-65.[cited 29 Apr 2012] Available from: http://www.nritld.ac.ir/portals/tanaffosjournal/2003;%20vol%202,%20No%206/dr%20mir%20saiedi%206.pdf [Google Scholar]
[10]. Owolabi LF, Nagoda MM, Samaila AA, Aliyu I, Spinal tuberculosis in adults: A study of 87 cases in Northwestern NigeriaNeurology Asia [Internet] 2010 15(3):239-44.[cited 20 Feb 2012] Available from: http://www.neurology-asia.org/articles/neuroasia-2010-15(3)-239.pdf [Google Scholar]
[11]. Jain A K, Tuberculosis of the spine: A fresh look at an old diseaseJ Bone Joint Surg Br 2010 July92-B(70):905-13.doi: 10.1302/0301-620X.92B7.24668 [Google Scholar]