Introduction
Statistics indicate that 3.4 million individuals around the world have been diagnosed with SLE. In terms of gender, more females are affected by the disease, with nine out of 10 diagnosed being women. While SLE can potentially manifest in individuals of all ages, the demographic group most impacted is women of reproductive age, generally between 15 and 44-year-old [1]. The SLE is a heterogeneous autoimmune disorder whose clinical course and prognosis can vary significantly. The manifestations of SLE may not be obvious or could be pronounced, affecting one or multiple organ systems, and can evolve as the disease progresses, creating challenges for accurate diagnosis. The disease generally manifests in the form of rashes on the skin, such as the malar “butterfly rash,” lupus nephritis, alopecia, arthritis, pleurisy, and serositis [2].
An aspect of the disease that complicates matters for both patients and clinicians is the variable response to treatment and the challenges in predicting how this will transpire. The cause of this clinical heterogeneity is likely the complex immune dysregulation that underlies SLE pathogenesis. At the cellular level, interactions between the innate and adaptive immune systems drive the process, leading to upregulation of cytokines, complement activation, immune complex deposition, and, ultimately, tissue damage and inflammation [2].
Although the disease aetiology is currently not sufficiently understood, research has shown that the interaction between genetic and environmental factors triggers immune responses, causing B cells to produce pathogenic autoantibodies at excessive levels, along with cytokine dysregulation, resulting in damage to both organs and tissues. The presence of antibodies to cytoplasmic and nuclear antigens is a characteristic feature of SLE [3].
The clinical features of the disorder can vary significantly, with some patients experiencing only mild cutaneous involvement, while others suffer severe organ damage, which may include cardiac failure, pulmonary hypertension, and kidney failure. Both laboratory and clinical findings form the basis of SLE diagnosis. The latest and most accurate criteria available for the disease are enhanced classification criteria utilised by the European League Against Rheumatism (EULAR) and the American College of Rheumatology (ACR) [4].
Managing SLE is a complex process that necessitates the involvement of multiple disciplines. The disease severity as well as the specific organs affected, influences the treatment algorithm. While several different systems may be impacted by the disease, individuals can follow differing courses depending on the severity, number of flare-ups, and periods of remission. The specific organ involved can determine whether the patient’s life expectancy is reduced, particularly if it is the heart, lungs, or kidneys. However, if this is not the case and they are closely monitored, the life expectancy of SLE patients will be normal in 80 to 90% of individuals [3].
It has been determined that SLE is a disease in which adaptive immunity is perturbed due to the key pathogenic roles of T and B cells. Through both germinal centre reactions and extrafollicular routes, autoreactive B cells generate distinctive Antinuclear Antibodies (ANAs), such as anti-Ribonucleic Acid (RNA)-associated proteins, anti-RNA, and anti-double-stranded Deoxyribonulceic Acid (DNA). These ANAs form Immune Complexes (ICs), which release nuclear material following excessive or incorrect cell death processes. Once ICs are deposited in tissues, they initiate a pro-inflammatory cycle, attracting various immune cells, releasing cytokines, and causing significant damage to numerous organs [5]. The aforementioned ICs trigger plasmacytoid Dendritic Cells (pDCs) to produce Interferon (IFN)-α via Toll-like Receptor (TLR) -7 or -9. Furthermore, under certain conditions, neutrophils can respond to chromatin to generate IFN-α. The activation of IFN-regulated genes triggers the Janus Kinase (JAK)/Signal Transducer and Activator of Transcription (STAT) pathway when IFN-α binds to its receptor. The majority of SLE patients exhibit increased expression of IFN-inducible genes, sometimes known as “a type I IFN signature,” and/or elevated levels of IFN-α in the blood [6].
Neutrophil granulocytes account for approximately 50 to 70% of all leukocytes in the circulatory system, and among all the cellular components that comprise the human body’s innate immune system, they possess the greatest mobility and abundance [7]. Evidence suggests that neutrophils are involved in a variety of human diseases, such as cancer, inflammatory disorders, chronic autoimmune diseases, several lung diseases, sepsis, and infectious diseases. Nevertheless, the role played by neutrophils can change depending on the type of disease and is impacted by different factors, including disease progression, circadian disruption, ageing, and changes in the microbiome. If neutrophils do not function adequately (e.g., in severe fungal or bacterial infections), it is crucial to enhance the overall activity of the neutrophil compartment. Conversely, in specific inflammatory diseases, neutrophil activation may be excessive, indicating that the neutrophil compartment should be attenuated. In individuals with cancer or sepsis, neutrophils may cease to function normally and shift to an abnormal, pathogenic phenotype, requiring restoration of neutrophil function to normal levels [8].
Neutrophils have various effector functions, including the formation of NETs, oxidative burst, and phagocytosis, all of which are recognised for their roles in defending the host. However, if these functions are abnormal, this may result in the emergence of autoimmune disorders, including Antiphospholipid Syndrome (APS) and SLE [6].
The present review will present a discussion of the particular role neutrophils play in the development of SLE. Additionally, it will explore the potential for these cells to be involved in the diagnosis and treatment of SLE.
Discussion
Neutrophil Phenotypes in Lupus
Neutrophils are more than just a homogenous population of cells; they are a complex type of cell with subtypes that exhibit functional and phenotypical differences [9]. Neutrophils in circulation display natural variations according to their age. In older neutrophils, the expression of the adhesion molecule L-selectin is increased, while that of the activation marker Cluster of Differentiation (CD)11b is decreased. Neutrophils are capable of polarising within tissues according to the specific environment, and, similar to macrophages and monocytes, they have the potential to polarise into a pro-inflammatory {Tumour Necrosis Factor (TNF) α-driven} or anti-inflammatory {Transforming Growth Factor (TGF) β-driven} phenotype [10].
A specific subgroup of granulocytes with lower density and unique features, as compared to granulocytes with normal density, is associated with several chronic inflammatory diseases. These Low-density Granulocytes (LDGs) were initially discovered in relation to SLE in 1986. The Ficoll-Hypaque density gradient centrifugation technique was used to detect these Low-density Neutrophils (LDNs) in the Peripheral Blood Mononuclear Cells (PBMCs) of SLE patients [11]. Based on additional cell function studies, LDNs have been identified as pro-inflammatory neutrophils with increased production of IFN-γ and TNF-α, enhanced cytotoxicity against endothelial cells, and reduced phagocytic capacity. Some cells within the LDNs have nuclei that resemble myelocytes or possess a band structure like immature granulocytes, which differs from typical neutrophils. Other cells feature nuclei that are comprised of several lobes, similar to adult neutrophils [12].
The SLE is associated with two types of low-density neutrophils: LDGs, which are linked with a pro-inflammatory phenotype, and Monocyte-Derived Suppressor Cells (MDSCs), which possess an anti-inflammatory phenotype and are responsible for suppressing T cell function [13].
Specifically, LDGs induce vascular dysfunction and endothelial damage due to their ability to efficiently synthesise and extrude NETs. In relation to SLE, NETs promote the maturation of immune cells, cause tissue damage, and increase the production of pro-inflammatory cytokines and type I IFNs. Apart from other cardiovascular disease risk factors, the quantity of LDGs in SLE is associated with vascular inflammation and in vivo coronary atherosclerosis, as well as increased T cell activation. These results suggest that directly targeting LDGs may prevent certain aspects of SLE-related immune dysregulation, organ damage, and early atherosclerosis [14].
However, two main types of MDSCs have been identified in mice and humans: monocytic MDSCs (M-MDSCs) and Polymorphonuclear MDSCs (PMN-MDSCs). These are categorised based on their origin from either the granulocytic or monocytic cell lineages, respectively [15]. Due to the continuously changing microenvironments during the disease, MDSCs may also depend on inflammatory systems or their various stages of development. MDSCs from early-stage lupus-prone mice have been shown to have the ability to suppress T-cell proliferation. Nevertheless, the regulatory balance between Th17 and T regulatory (Treg) cells eventually takes precedence over the inhibitory capacity of MDSCs [16].
Neutrophils Apoptosis in Lupus
When inflammation is not present, neutrophils circulate in the blood for approximately 24-48 hours before returning to the bone marrow, where they undergo apoptosis. Bcl-2 family proteins are responsible for regulating constitutive neutrophil apoptosis, which includes anti-apoptotic proteins such as MCL1 and A1/BFL1, as well as pro-apoptotic proteins like BAX, BAK, and BID [17]. When MCL1 and BFL1 are lost, BAX and BAK form pores in the mitochondrial membrane; cytochrome c is then released, generating the apoptosome in combination with APAF1, followed by the cleavage of caspases, ultimately leading to apoptosis [18,19].
In SLE, the enhancement of neutrophil apoptosis results in a greater apoptotic burden associated with the development of anti-nuclear autoantibodies. Apoptosis is observed to be increased in SLE neutrophils in vitro, potentially due to dysregulation between pro-apoptotic caspases and apoptosis inhibitors. Serum from SLE patients contains significantly elevated levels of pro-apoptotic Fas Ligand (FasL) and Tumour Necrosis Factor-related Apoptosis-Inducing Ligand (TRAIL), which can induce apoptosis in normal neutrophils. Conversely, SLE serum shows decreased levels of GM-CSF, and supplementation with physiological levels of GM-CSF can restore the ability of SLE serum to promote apoptosis in normal neutrophils [20].
During the apoptosis process, blebs are formed on the cell membrane, which detach from the cell and contain fragmented cellular material, including nuclear antigens. The immune system generally removes this apoptotic debris quickly, making it inaccessible [21]. On average, around one billion neutrophils undergo apoptosis daily [22]. If apoptotic debris containing nucleic acids persists, it can trigger an inflammatory response as nucleic acid recognition receptors, including members of the TLR family, are activated. Apoptotic microparticles in circulation prepare neutrophils for nuclear material, thereby increasing the antigen load. Nucleic acid recognition receptors are capable of controlling endogenous retroviruses, recognising viral pathogens, and defending against intracellular bacteria, and they have a strong association with the production of type I IFN. Evidence now suggests that if these pathways are defective, they play a significant role in the pathogenesis of SLE, as this both increases susceptibility to the disease and can directly cause monogenic forms of SLE [22].
Neutrophils Phagocytosis in Lupus
The phagocytic capacity of neutrophils in SLE is reduced, and the C1q/calreticulin/CD91-mediated apoptotic pathway cannot clear them adequately, while their oxidative activity is elevated [23,24]. A comparison between the phagocytic function of neutrophils in SLE patients and healthy donors revealed that the majority of neutrophils from SLE patients exhibited a reduced ability to phagocytose beads coated in albumin. Furthermore, impaired phagocytosis of beads coated in Immunoglobulin (Ig) was observed in approximately 30% of cases. In certain SLE patients, the ability to phagocytose necrotic cells and degrade chromatin by Polymorphonuclear (PMN) cells was also found to be decreased [25].
One of the factors implicated in the aetiology of autoimmunity is the inability to clear dying cells. In SLE, both apoptotic neutrophils and impaired phagocytosis by macrophages have been observed. When clearance does not occur optimally, there is a possibility for apoptotic cells to transform into Secondary Necrotic Cells (SNECs), leading to the release of danger signals and autoantigens. In healthy individuals, phagocytosis of nuclear remnants does not occur because DNases and C1q rapidly degrade them via the reticuloendothelial system. Both DNase activity impairment and complement protein deficiency are commonly observed in SLE patients. Conversely, nuclear material opsonised by complement and ANAs relies on being removed through phagocytosis by neutrophils, while the recognition of SNECs by antibodies promotes neutrophilic phagocytosis [6,22,26].
In SLE patients, a large number of Damage-Associated Molecular Patterns (DAMPs) accumulate as a result of the more rapid death of cells combined with a reduced ability to clear the resulting debris. Increased oxidative stress and its additional effects cause the Pattern Recognition Receptors (PRRs) on multiple innate immune cells to be activated. The enzyme complex NADPH oxidase (NOX2), found within neutrophils, is primarily responsible for producing Reactive Oxygen Species (ROS) as part of the oxidative/respiratory burst process, which has strong antimicrobial activity. However, one of the factors implicated in the pathogenesis of SLE is the overproduction of ROS, leading to an increase in oxidative stress [27].
Although ROS play an important role in eliminating pathogens, if they are produced at excessive and/or uncontrolled levels, this can result in various oxidative damages to proteins, DNA, lipids, membranes, and other biological molecules, which can result in the emergence of a range of inflammatory and autoimmune diseases. Neutrophils significantly contribute to tissue damage during acute disease processes, including acute injuries to the spinal cord and lungs, as well as chronic disease processes like asthma, atherosclerosis, inflammatory bowel diseases, Chronic Obstructive Pulmonary Disease (COPD), SLE, and rheumatoid arthritis [28].
Additional processes that facilitate the targeting of proteins by ROS include proteolysis, protein aggregation, formation of cross-links, modification of prosthetic groups, and amino acid oxidation. Furthermore, this can lead to the inactivation of key proteins, disrupting essential metabolic processes. Similarly, the radical damage of polyunsaturated fatty acids results in the production of hydroperoxidised lipids and toxic alkyl radicals. Lipid peroxidation in membrane phospholipids can also lead to damage to the cellular membrane [29].
Neutrophil NETosis in Lupus
When neutrophils die, chromatin fibres that resemble ‘spider webs’ are released, known as NETs. Activated neutrophils release NETs through a process called NETosis. This process involves cell death as a reaction to various stimuli, such as oxidative stress and infectious organisms [30]. NETs consist of granules, histones, decondensed chromatin, and components with bactericidal activity. Antimicrobial enzymes, including gelatinase, lactoferrin, cathepsin, proteinase 3, histones, and cathelicidins like LL-37, NE, or MPO, are expelled with the NETs, which can trap, inhibit, and eliminate foreign pathogens in an extracellular manner, as opposed to the recognised phagocytic method [31].
The formation and functioning of NETs involve various enzymes beyond MPO, including Peptidyl Arginine Deiminase 4 (PAD4), Rac2 from the Rho family, and NADPH oxidase. PAD4 is a calcium-dependent enzyme that is restricted to neutrophil nuclei and plays a role in histone citrullination. The action of PAD4 leads to increased citrullination, resulting in a greater generation of autoantigens and the deamination of proteins like histones H2A, H3, and H4. The pathology of SLE is alleviated when PAD4 is pharmacologically inhibited or deleted [32].
When NETs are formed in SLE, the leakage of autoantigens from cell debris that has not been cleared occurs. Follicular Dendritic Cells (DC) within the germinal centres of secondary lymphoid tissues subsequently present B cell neo-epitopes, resulting in the destruction of self-tolerance. When autoreactive B cells are activated and autoantibodies are subsequently produced, ICs are formed, which have the potential to trigger an inflammatory response that can lead to additional tissue damage. Additionally, ICs are capable of attracting phagocytes, which can result in further production of pro-inflammatory cytokines. Such processes may create a vicious cycle in which inflammation is perpetuated [33].
Proteases can remove a significant proportion of NET proteins that are recognised by autoantibodies generated by SLE patients. In both individuals with and without SLE, enhanced NET formation is linked with vascular disease, which can contribute to the accelerated development of coronary plaque and lipoprotein disorders. Additionally, the release of IL-1 and IL-18 can be promoted via the NLRP3 inflammasome or P2X7 purinergic receptors, leading to worsened inflammation in patients with SLE [31].
NETosis plays a role in the type I IFN SLE signature via the stimulation of IFN production via pDCs [31]. This gene signature, known as the IFN signature, is induced when IFNα levels are elevated. Various autoimmune disorders, particularly SLE, feature this signature, and it is regarded as a therapeutic target [34]. Such an effect manifests when TLR9 is activated by DNA and anti-DNA antibodies in complex with NET-derived antimicrobial peptides [35]. Consequently, neutrophils in SLE patients are primed to release NETs by type I IFN, implying the potential existence of a feedback loop. After extrusion from NETs, nuclear antigens are released from this nuclear material, promoting the development of antibodies in SLE [Table/Fig-1] [36].
Role of neutrophils in the pathogenesis of SLE.
Image created via BioRender
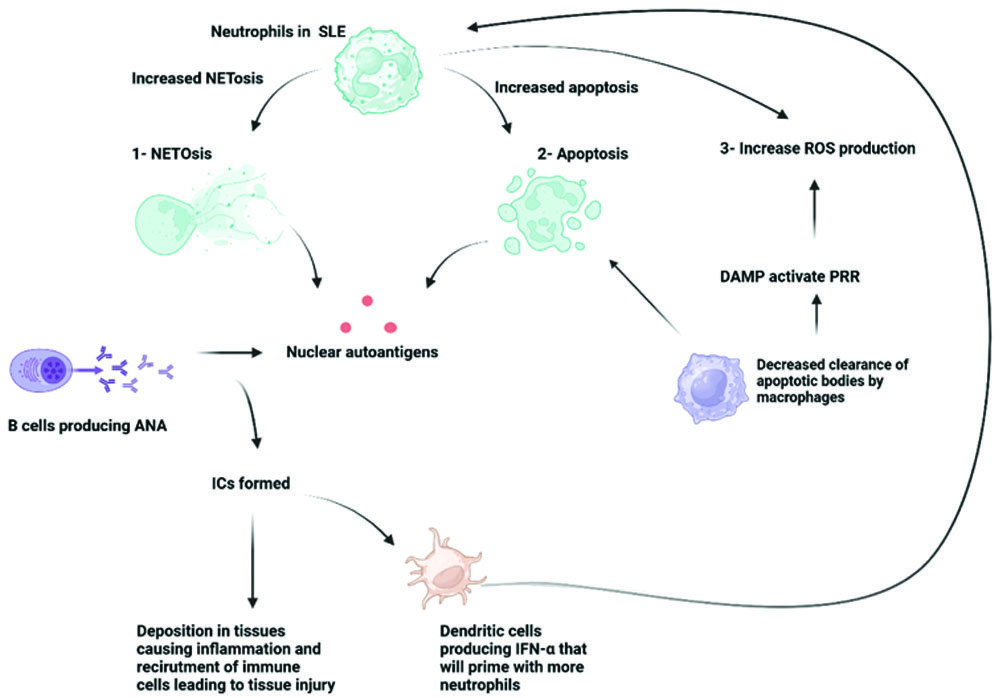
Studies have not only detected neutrophils in nephritic kidneys in cases of systemic lupus but also in ANCA-Associated Vasculitides (AAV), implying that components of NETs could be involved in inducing the severe symptoms of such systemic inflammatory diseases [36].
Neutrophils as a Diagnostic Biomarker in Lupus
The early mortality rates associated with SLE remain high, suggesting that the ability to diagnose and treat the disease faces significant challenges. SLE exhibits a highly heterogeneous clinical presentation, with a growing number of atypical cases, making diagnosis even more difficult [37]. Disease activity in patients with SLE is frequently assessed using the SLE Disease Activity Index 2000 (SLEDAI-2K). Nevertheless, its potential for use in regular clinical practice is limited by its complexity [38]. Additionally, the immunological diagnostic criteria for SLE largely incorporate anti-Smith (anti-SM) antibodies, anti-nuclear antibodies, and anti-double-stranded DNA (anti-dsDNA) antibodies [39]. However, these criteria have reduced sensitivity and specificity and are not particularly effective for diagnosing SLE [40].
Traditional techniques, such as C-Reactive Protein (CRP) and Erythrocyte Sedimentation Rate (ESR), are not fully compatible with SLE activity. It has been demonstrated that IFN-α inhibits IL-6/IL-1β-induced transcription of genes for the production of CRP by hepatocytes. Unlike autoimmune diseases such as rheumatoid arthritis and bacterial infections, SLE is not associated with increased levels of CRP, limiting its use as a marker of inflammation for lupus patients [41]. Thus, there is an increased focus on identifying biomarkers capable of predicting SLE and quantifying disease activity; however, there is a decreased likelihood that any individual biomarker will substitute clinical assessment due to the heterogeneous nature of the disease [42].
Research based on bioinformatics has facilitated the discovery of numerous potential genetic biomarkers. Specifically, five significant genes are of interest: ABCB1, CD247, DSC1, KIR2DL3, and MX2. Evidence from clinical samples taken from patients with SLE supports the good validity and reliability of these genes [41]. Further research suggests the possibility of identifying diagnostic biomarkers (IFI44, IFI44L, EIF2AK2, IFIT3, IFITM3, ZBP1, TRIM22, and PRIC285) for SLE through the integration of bioinformatics techniques, with findings indicating that IFI44 could potentially serve as an ideal biomarker [43].
Numerous researchers have suggested the potential for the Neutrophil-to-Lymphocyte Ratio (NLR) to be utilised as a biomarker, based on the calculation of the basic neutrophil-to-lymphocyte count ratio measured in peripheral blood. This ratio reflects the balance between two components of the immune system: acute/chronic inflammation and adaptive immunity. NLR values typically range between 1 and 2; however, if they exceed 3 or drop below 0.7 in adults, they are considered pathological. The NLRs of lupus patients are higher than those of healthy controls, and studies have demonstrated a correlation between lupus activity according to the SLEDAI and increased NLR values [41]. It is proposed that the NLR may be beneficial for evaluating inflammation levels or potentially sub-clinical inflammation in patients diagnosed with systemic inflammatory disorders. Additionally, another potential avenue could involve testing the NLR as a prognostic biomarker for autoimmune relapse [42].
In a meta-analysis involving eight studies, it was found that there is significant potential for the easily measured NLR to be utilised in the diagnosis of SLE based on its accuracy [44]. Nevertheless, new biomarkers must be identified and defined, as there are still aspects requiring further attention in diagnosing and managing the disease [45].
Neutrophils as a Therapeutic Target in Lupus
Broad-spectrum immunosuppressive therapy is generally applied to treat SLE, which can result in varying levels of efficacy among different patients and can also lead to negative outcomes, especially if administered over an extended period. The varied aetiology of SLE has resulted in different clinical manifestations as well as distinct molecular and cellular bases [37]. There is a lack of therapies specifically designed to target neutrophil subsets or the formation of NETs. Several therapeutic approaches have been developed, such as the depletion or reduction of neutrophil numbers, blocking neutrophil chemotaxis, inhibiting activation, causing neutrophil-derived toxic mediators, and/or preventing NET formation [46].
The LDNs constitute a novel therapeutic target for SLE. If a pathogenic LDN can be identified, this could enable the development of drugs capable of targeting specific populations of neutrophils while preserving important features of host defence mediated by neutrophils. Drugs that can target critical events in the process through which NETs are formed in LDNs or stimulate the clearance of NETs may offer novel therapeutic approaches for SLE patients. This incorporates various drugs currently being used to treat SLE, such as Cyclosporine, Colchicine, and Chloroquine/Hydroxychloroquine (HDQ), as well as novel agents undergoing testing, such as Tofacitinib and N-acetylcysteine (NAC) [5]. For instance, the efficacy of knockout strategies and PAD4 inhibitors has been demonstrated in different mouse models of autoimmunity as well as in human cells in vitro. Proposed medications include antibiotics, vitamin D, recombinant human DNase (rhDNase), NAC, Diphenyleneiodonium (DPI), HDQ, Cl-amidone, among others [47]. However, further human trials must be conducted to determine their safety and efficacy in vivo [46].
Various strategies are being developed aimed at directly reducing NET formation and/or enhancing the degradation process. Examples of such strategies include using diphenyleneiodonium to target Reactive Oxygen Species (ROS), using N-acetylcysteine to target mitochondrial DNA, utilising DNase1 to enhance NET breakdown, enhancing the digestion of DNA in apoptotic microparticles with DNASE1L3, and employing anti-citrullinated protein antibodies to target histones. Tests are being conducted to assess the efficacy of these strategies on lupus in pre-clinical animal models and human studies. Additionally, it is possible to suppress the production of NETs via NADPH oxidase inhibitors [46-48]. This indicates that inhibiting IL-17A or the tissue factor expressed on NETs may represent a novel candidate as a therapeutic target [48].
Conclusion(s)
Neutrophils are crucial cells in both adaptive and innate immunity, playing a vital role in responding to infections through various mechanisms and the secretion of molecules. Their involvement in the development of SLE presents potential opportunities for improving the diagnosis and treatment of this complex autoimmune disorder. Diagnosing SLE is currently challenging due to its varied manifestations, and existing medications only manage symptoms without curing the disease. Further research on neutrophils may lead to the identification of simple diagnostic biomarkers or potential treatment targets that focus on NETosis and its components.
[1]. Hoi A, Igel T, Mok CC, Arnaud L, Systemic lupus erythematosusLancet 2024 403(10441):2326-38.Epub 2024 Apr 17. Erratum in: Lancet. 2024;403(10441)2292. Doi: 10.1016/S0140-6736(24)01044-410.1016/S0140-6736(24)00398-238642569 [Google Scholar] [CrossRef] [PubMed]
[2]. Lazar S, Kahlenberg JM, Systemic lupus erythematosus: New diagnostic and therapeutic approachesAnnu Rev Med 2023 74:339-52.10.1146/annurev-med-043021-03261135804480 [Google Scholar] [CrossRef] [PubMed]
[3]. Ameer MA, Chaudhry H, Mushtaq J, Khan OS, Babar M, Hashim T, An overview of Systemic Lupus Erythematosus (SLE) pathogenesis, classification, and managementCureus 2022 14(10):e3033010.7759/cureus.30330 [Google Scholar] [CrossRef]
[4]. Aringer M, Costenbader K, Daikh D, Brinks R, Mosca M, Ramsey-Goldman R, European League Against Rheumatism/American College of Rheumatology Classification Criteria for Systemic Lupus ErythematosusArthritis Rheumatol 2019 71(9):1400-12.10.1002/art.4093031385462PMC6827566 [Google Scholar] [CrossRef] [PubMed]
[5]. Tay SH, Celhar T, Fairhurst AM, Low-density neutrophils in systemic lupus erythematosusArthritis Rheumatol 2020 72(10):1587-95.10.1002/art.4139532524751PMC7590095 [Google Scholar] [CrossRef] [PubMed]
[6]. Wirestam L, Arve S, Linge P, Bengtsson AA, Neutrophils- important communicators in systemic lupus erythematosus and antiphospholipid syndromeFront Immunol 2019 10:273410.3389/fimmu.2019.0273431824510PMC6882868 [Google Scholar] [CrossRef] [PubMed]
[7]. Kraus RF, Gruber MA, Neutrophils- from bone marrow to first-line defense of the innate immune systemFront Immunol 2021 12:76717510.3389/fimmu.2021.76717535003081PMC8732951 [Google Scholar] [CrossRef] [PubMed]
[8]. Németh T, Sperandio M, Mócsai A, Neutrophils as emerging therapeutic targetsNat Rev Drug Discov 2020 19(4):253-75.10.1038/s41573-019-0054-z31969717 [Google Scholar] [CrossRef] [PubMed]
[9]. Bissenova S, Ellis D, Mathieu C, Gysemans C, Neutrophils in autoimmunity: When the hero becomes the villainClin Exp Immunol 2022 210(2):128-40.10.1093/cei/uxac09336208466PMC9750832 [Google Scholar] [CrossRef] [PubMed]
[10]. Ng LG, Ostuni R, Hidalgo A, Heterogeneity of neutrophilsNat Rev Immunol 2019 19(4):255-65.10.1038/s41577-019-0141-830816340 [Google Scholar] [CrossRef] [PubMed]
[11]. Hassani M, Hellebrekers P, Chen N, van Aalst C, Bongers S, Hietbrink F, On the origin of low-density neutrophilsJ Leukoc Biol 2020 107(5):809-18.10.1002/JLB.5HR0120-459R32170882PMC7318192 [Google Scholar] [CrossRef] [PubMed]
[12]. Sun R, Huang J, Yang Y, Liu L, Shao Y, Li L, Dysfunction of low-density neutrophils in peripheral circulation in patients with sepsisSci Rep 2022 12(1):68510.1038/s41598-021-04682-x35027618PMC8758723 [Google Scholar] [CrossRef] [PubMed]
[13]. Hardisty GR, Llanwarne F, Minns D, Gillan JL, Davidson DJ, Gwyer Findlay E, High purity isolation of low density neutrophils casts doubt on their exceptionality in health and diseaseFront Immunol 2021 12:62592210.3389/fimmu.2021.62592234168640PMC8217868 [Google Scholar] [CrossRef] [PubMed]
[14]. Mistry P, Nakabo S, O’Neil L, Goel RR, Jiang K, Carmona-Rivera C, Transcriptomic, epigenetic, and functional analyses implicate neutrophil diversity in the pathogenesis of systemic lupus erythematosusProc Natl Acad Sci U S A 2019 116(50):25222-28.10.1073/pnas.190857611631754025PMC6911190 [Google Scholar] [CrossRef] [PubMed]
[15]. Veglia F, Sanseviero E, Gabrilovich DI, Myeloid-derived suppressor cells in the era of increasing myeloid cell diversityNat Rev Immunol 2021 21(8):485-98.10.1038/s41577-020-00490-y33526920PMC7849958 [Google Scholar] [CrossRef] [PubMed]
[16]. Ji J, Li P, Shen C, Dou H, Wang T, Shi L, MDSCs: Friend or foe in systemic lupus erythematosusCell Mol Immunol 2019 16(12):937-39.10.1038/s41423-019-0271-831431693PMC6884520 [Google Scholar] [CrossRef] [PubMed]
[17]. Vier J, Groth M, Sochalska M, Kirschnek S, The anti-apoptotic Bcl-2 family protein A1/Bfl-1 regulates neutrophil survival and homeostasis and is controlled via PI3K and JAK/STAT signalingCell Death Dis 2016 7(2):e210310.1038/cddis.2016.2326890142PMC5399193 [Google Scholar] [CrossRef] [PubMed]
[18]. Kale J, Osterlund EJ, Andrews DW, BCL-2 family proteins: Changing partners in the dance towards deathCell Death Differ 2018 25(1):65-80.10.1038/cdd.2017.18629149100PMC5729540 [Google Scholar] [CrossRef] [PubMed]
[19]. Saddam M, Paul SK, Habib MA, Fahim A, Mimi A, Islam S, Emerging biomarkers and potential therapeutics of the BCL-2 protein family: The apoptotic and anti-apoptotic contextEgypt J Med Hum Genet 2024 25(1):1210.1186/s43042-024-00485-7 [Google Scholar] [CrossRef]
[20]. Fresneda Alarcon M, McLaren Z, Wright HL, Neutrophils in the pathogenesis of rheumatoid arthritis and systemic lupus erythematosus: Same foe different M.OFront Immunol 2021 12:64969310.3389/fimmu.2021.64969333746988PMC7969658 [Google Scholar] [CrossRef] [PubMed]
[21]. Battistelli M, Falcieri E, Apoptotic Bodies: Particular extracellular vesicles involved in intercellular communicationBiology 2020 9(1):2110.3390/biology901002131968627PMC7168913 [Google Scholar] [CrossRef] [PubMed]
[22]. Tsokos GC, Lo MS, Reis PC, Sullivan KE, New insights into the immunopathogenesis of systemic lupus erythematosusNat Rev Rheumatol 2016 12(12):716-30.10.1038/nrrheum.2016.18627872476 [Google Scholar] [CrossRef] [PubMed]
[23]. Herrada AA, Escobedo N, Iruretagoyena M, Valenzuela RA, Burgos PI, Cuitino L, Innate immune cells’ contribution to systemic lupus erythematosusFront Immunol 2019 10:77210.3389/fimmu.2019.0077231037070 [Google Scholar] [CrossRef] [PubMed]
[24]. Pilania RK, Rawat A, Shandilya J, Arora K, Gupta A, Saikia B, Pediatric systemic lupus erythematosus: Phagocytic defect and oxidase activity of neutrophilsPediatr Res 2022 92(6):1535-42.10.1038/s41390-022-02055-235414669 [Google Scholar] [CrossRef] [PubMed]
[25]. Mahajan A, Herrmann M, Muñoz LE, Clearance deficiency and cell death pathways: A model for the pathogenesis of SLEFront Immunol 2016 7:3510.3389/fimmu.2016.0003526904025 [Google Scholar] [CrossRef] [PubMed]
[26]. Wu H, Fu S, Zhao M, Lu L, Lu Q, Dysregulation of cell death and its epigenetic mechanisms in systemic lupus erythematosusMolecules 2017 22:3010.3390/molecules2201003028035990PMC6155917 [Google Scholar] [CrossRef] [PubMed]
[27]. Fatemi A, Alipour R, Khanahmad H, Alsahebfosul F, Andalib A, Pourazar A, The impact of neutrophil extracellular trap from patients with systemic lupus erythematosus on the viability, CD11b expression and oxidative burst of healthy neutrophilsBMC Immunology 2021 22(1):1210.1186/s12865-021-00402-233546594PMC7863477 [Google Scholar] [CrossRef] [PubMed]
[28]. Elloumi N, Ben Mansour R, Marzouk S, Mseddi M, Fakhfakh R, Gargouri B, Differential reactive oxygen species production of neutrophils and their oxidative damage in patients with active and inactive systemic lupus erythematosusImmunol Lett 2017 184:01-06.10.1016/j.imlet.2017.01.01828163154 [Google Scholar] [CrossRef] [PubMed]
[29]. Kannan KP, Smiline Girija AS, Exploring the ROS reduction strategies in chronic lupus managementFront Immunol 2024 15:134665610.3389/fimmu.2024.134665638444846PMC10913789 [Google Scholar] [CrossRef] [PubMed]
[30]. Wang H, Kim SJ, Lei Y, Wang S, Wang H, Huang H, Neutrophil extracellular traps in homeostasis and diseaseSignal Transduct Target Ther 2024 9(1):23510.1038/s41392-024-01933-x39300084PMC11415080 [Google Scholar] [CrossRef] [PubMed]
[31]. Delgado-Rizo V, Martínez-Guzmán MA, Iñiguez-Gutierrez L, García-Orozco A, Alvarado-Navarro A, Fafutis-Morris M, Neutrophil extracellular traps and its implications in inflammation: An overviewFront Immunol 2017 8:8110.3389/fimmu.2017.0008128220120PMC5292617 [Google Scholar] [CrossRef] [PubMed]
[32]. Salemme R, Peralta LN, Meka SH, Pushpanathan N, Alexander JJ, The role of netosis in systemic lupus erythematosusJ Cell Immunol 2019 1(2):33-42.10.33696/immunology.1.008 [Google Scholar] [CrossRef]
[33]. Fu X, Liu H, Huang G, Dai SS, The emerging role of neutrophils in autoimmune-associated disorders: Effector, predictor, and therapeutic targetsMedComm (2020) 2021 2(3):402-13.10.1002/mco2.6934766153PMC8554667 [Google Scholar] [CrossRef] [PubMed]
[34]. van den Hoogen LL, van der Linden M, Meyaard L, Fritsch-Stork RDE, van Roon JA, Radstake TR, Neutrophil extracellular traps and low-density granulocytes are associated with the interferon signature in systemic lupus erythematosus, but not in antiphospholipid syndromeAnn Rheum Dis 2020 79(10):e13510.1136/annrheumdis-2019-21578131177097 [Google Scholar] [CrossRef] [PubMed]
[35]. Kubota T, An emerging role for Anti-DNA antibodies in systemic lupus erythematosusInt J Mol Sci 2023 24(22):1649910.3390/ijms24221649938003689PMC10671047 [Google Scholar] [CrossRef] [PubMed]
[36]. Corsiero E, Nerviani A, Bombardieri M, Pitzalis C, NETosis as source of autoantigens in rheumatoid arthritisFront Immunol 2016 7:43010.3389/fimmu.2016.00485 [Google Scholar] [CrossRef]
[37]. Li H, Zhang X, Shang J, Feng X, Yu L, Fan J, Identification of NETs-related biomarkers and molecular clusters in systemic lupus erythematosusFront Immunol 2023 14:115082810.3389/fimmu.2023.115082837143669PMC10151561 [Google Scholar] [CrossRef] [PubMed]
[38]. Wang L, Wang C, Jia X, Yang M, Yu J, Relationship between neutrophil-to-lymphocyte ratio and systemic lupus erythematosus: A meta-analysisClinics 2020 75:e145010.6061/clinics/2020/e145032321113PMC7153360 [Google Scholar] [CrossRef] [PubMed]
[39]. Yu H, Nagafuchi Y, Fujio K, Clinical and immunological biomarkers for systemic lupus erythematosusBiomolecules 2021 11(7):92810.3390/biom1107092834206696PMC8301935 [Google Scholar] [CrossRef] [PubMed]
[40]. Zhao X, Duan L, Cui D, Xie J, Exploration of biomarkers for systemic lupus erythematosus by machine-learning analysisBMC Immunology 2023 24(1):4410.1186/s12865-023-00581-037950194PMC10638835 [Google Scholar] [CrossRef] [PubMed]
[41]. Kourilovitch M, Galarza–Maldonado C, Could a simple biomarker as neutrophil-to-lymphocyte ratio reflect complex processes orchestrated by neutrophils?J Transl Autoimmun 2023 6:10015910.1016/j.jtauto.2022.10015937396460PMC10313502 [Google Scholar] [CrossRef] [PubMed]
[42]. Yu H, Jiang L, Yao L, Gan C, Han X, Liu R, Predictive value of the neutrophil-to-lymphocyte ratio and hemoglobin insystemic lupus erythematosusExp Ther Med 2018 16(2):1547-53.10.3892/etm.2018.6309 [Google Scholar] [CrossRef]
[43]. Jiang Z, Shao M, Dai X, Pan Z, Liu D, Identification of diagnostic biomarkers in systemic lupus erythematosus based on bioinformatics analysis and machine learningFront Genet 2022 13:86555910.3389/fgene.2022.86555935495164PMC9047905 [Google Scholar] [CrossRef] [PubMed]
[44]. Zinellu A, Paliogiannis P, Mangoni AA, A systematic review and meta-analysis of the diagnostic accuracy of the neutrophil-to-lymphocyte ratio and the platelet-to-lymphocyte ratio in systemic lupus erythematosusClin Exp Med 2024 24(1):17010.1007/s10238-024-01438-539052098PMC11272706 [Google Scholar] [CrossRef] [PubMed]
[45]. Capecchi R, Puxeddu I, Pratesi F, Migliorini P, New biomarkers in SLE: From bench to bedsideRheumatology (Oxford) 2020 59(Suppl 5):v12-18.10.1093/rheumatology/keaa48432911542PMC7719038 [Google Scholar] [CrossRef] [PubMed]
[46]. Nakabo S, Romo-Tena J, Kaplan MJ, Neutrophils as Drivers of immune dysregulation in autoimmune diseases with skin manifestationsJ Invest Dermatol 2022 142(3 Pt B):823-33.10.1016/j.jid.2021.04.01434253374 [Google Scholar] [CrossRef] [PubMed]
[47]. Huang J, Hong W, Wan M, Zheng L, Molecular mechanisms and therapeutic target of NETosis in diseasesMedComm 2022 3(3):e16210.1002/mco2.16236000086PMC9390875 [Google Scholar] [CrossRef] [PubMed]
[48]. Wang M, Ishikawa T, Lai Y, Nallapothula D, Singh RR, Diverse roles of NETosis in the pathogenesis of lupusFront Immunol 2022 13:89521610.3389/fimmu.2022.89521635686129PMC9170953 [Google Scholar] [CrossRef] [PubMed]