Introduction
When a portion of the blood flow is stopped or disrupted, the brain’s tissue may become deprived of oxygen and nutrients, which can lead to a stroke. Due to the rapid ischaemic changes in brain cells, brain functioning is altered. There are two types of strokes: haemorrhagic (caused by a rupture blood vessel and subsequent brain haemorrhage) and ischaemic (caused by a blockage in a blood vessel) [1]. Sudden weakness or numbness in one or both arms, legs, or face, especially on one side of the body, is a common sign of a stroke. Other symptoms include sudden disorientation, loss of balance or coordination, dizziness, difficulty speaking or understanding, difficulty walking and an abrupt, severe headache with no apparent cause. In India, stroke is a significant public health concern [2]. According to a published article, the age-adjusted prevalence of stroke in India is expected to be 545 per 100,000 persons, with higher rates in rural areas compared to metropolitan ones [3]. The study also highlighted the increasing prevalence of stroke in India, particularly with the rising incidence of risk factors like diabetes, high blood pressure and tobacco use [4].
Following a stroke, the resultant lesion leads to an interhemispheric imbalance involving hyperexcitability of the contralesional hemisphere as well as contralateral motor dysfunction [5]. Bilateral activation of the hemiparetic side is frequently observed during recovery, as neural reorganisation in both the ipsilesional and contralesional hemispheres may occur in an effort to regain motor functioning. There is ongoing debate regarding the role and significance of the contralesional motor cortex, despite the traditional belief that reorganisation of the ipsilesional hemisphere is the most crucial factor for successful recovery [6]. According to several functional magnetic resonance imaging studies, patients who continue to experience hyperexcitability in the contralesional hemisphere after it has been inhibited tend to recover less fully; nonetheless, some patients who achieve a full recovery still exhibit contralesional motor activity and reduced functional ability [7].
One non invasive brain stimulation method that has been studied for its potential to expedite stroke recovery is rTMS [8]. rTMS is used to suppress the contralesional hemisphere or stimulate the ipsilesional motor cortex, which may improve recovery in stroke patients [9]. In the context of stroke, rTMS is commonly used to modify the excitability of the contralesional (opposite side) motor cortex. Low-frequency repetitive motor stimulation applied to the contralesional primary motor cortex has been shown to enhance motor function and decrease interhemispheric asymmetry in patients with subacute stroke [10]. Additionally, it increases the excitability of the ipsilesional (affected) motor cortex.
The effect of rTMS on the side of the brain opposite the stroke lesion, known as the contralesional hemisphere, is a topic of ongoing discussion. Low-frequency rTMS (1 Hz or less) applied to the contralesional hemisphere may enhance motor recovery and reduce its inhibitory effect on the affected hemisphere [11]. Some studies suggest that the contralesional hemisphere may benefit from high-frequency rTMS (more than 5 Hz) to increase its excitatory effect on the affected hemisphere and facilitate motor compensation [12]. The optimal frequency, intensity, duration and timing of rTMS for the contralesional hemisphere may depend on a number of variables, such as the site and severity of the stroke, the patient’s stage of recovery and their individual characteristics.
One study indicates that low-frequency contralesional rTMS can aid in stroke recovery when coupled with intensive physiotherapy [13]. However, the effectiveness of rTMS on the contralesional cortex may vary depending on several factors, including when it is applied in relation to motor training, the type of stroke (chronic or subacute) and the stroke’s location (cortical or subcortical) [10].
Although rTMS shows promise in aiding stroke recovery, particularly when applied to the contralesional cortex, there is a lack of literature regarding the use of the intermittent theta burst stimulation form of rTMS to inhibit the contralesional cortex. Further research is needed to fully understand its effectiveness across different stroke populations and to maximise its potential use in stroke rehabilitation. In this context, this research aims to identify the efficacy of rTMS on the ipsilateral cortex combined with a standard physiotherapy protocol for the contralateral side in improving tone and function in subacute stroke survivors.
This research aims to determine the effectiveness of a standard physiotherapy protocol for the contralateral side combined with rTMS of the ipsilateral (contralesional) cortex in improving muscle tone, function, functional reach (risk of falling) and gait velocity in subacute stroke survivors.
Objectives:
Primary objective: To determine the effectiveness of rTMS on the ipsilateral cortex, along with a standard physiotherapy protocol for the contralateral side, on tone and function in subacute stroke survivors.
Secondary objective: To compare the effectiveness of rTMS on the ipsilateral cortex, along with a standard physiotherapy protocol for the contralateral side, against placebo rTMS and the standard physiotherapy protocol on tone and function in subacute stroke survivors.
Hypothesis:
Null hypothesis (H0): There will be no significant effect of rTMS on the ipsilateral cortex, along with the standard physiotherapy protocol for the contralateral side, on tone and function in subacute stroke survivors.
Alternative hypothesis (Ha): There will be a significant effect of rTMS on the ipsilateral cortex, along with the standard physiotherapy protocol for the contralateral side, on tone and function in subacute stroke survivors.
Review of Literature
Stroke often leads to motor impairments, including abnormalities in muscle tone and reduced function, primarily affecting the contralateral limbs. The standard treatment for improving motor function is physiotherapy, although it may not address the underlying neurological deficits. rTMS has emerged as a promising adjunct therapy that could enhance neural plasticity when applied to the ipsilateral cortex. This randomised controlled trial assesses the combined effect of rTMS on the ipsilateral cortex and standard physiotherapy on the contralateral side in subacute stroke survivors, with a focus on improving muscle tone and functional recovery.
Wu X et al., studied the effects of combined high-frequency rTMS and cervical nerve root magnetic stimulation on upper extremity motor recovery following a stroke. Both rTMS and cervical nerve root magnetic stimulation effectively promoted upper limb motor recovery in stroke patients. The protocol that combines the two techniques has been found to be more beneficial for motor improvement and patients can easily tolerate it [14]. Mann SK and Malhi NK focused on rTMS as a safe, non invasive technique for the cortical modulation of excitability, which is used for a myriad of psychiatric and neurological disorders, including treatment-resistant depression and obsessive-compulsive disorder [8].
Another study by Wu X et al., found that accelerated interrupted transcranial burst stimulation over the left dorsolateral prefrontal cortex significantly improves memory encoding functions in Alzheimer’s disease, with benefits observed for eight weeks and differing efficacies between mild and severe cases [15]. Boddington LJ and Reynolds JNJ suggested that interhemispheric inhibition with neuromodulation can enhance stroke rehabilitation. The extent to which asymmetric interhemispheric inhibition impacts stroke recovery is controversial. They suggested that interhemispheric inhibition is not static following a stroke in terms of the movement phase in which it is aberrantly engaged. Instead, it may dynamically increase in the perilesional areas during early movement, thereby impairing motor initiation. Consequently, its effect on stroke recovery may vary between studies depending on the technique used and the movement phase involved in eliciting the measurement [7].
Finally, authors propose that modulating excitability in the brain through more specific targeting of neural elements underlying interhemispheric inhibition—via stimulation type, location and intensity—may raise the ceiling of recovery following a stroke and enhance functional return [7].
Materials and Methods
This research is a two-arm parallel, single-centric, double-blinded, open-label equivalency randomised controlled trial. This study has been registered with the Clinical Trials Registry India (CTRI) under CTRI No: CTRI/2024/03/064866. The Standard Procedure Items: Recommendations for Interventional Trials (SPIRIT) checklist is adhered to throughout the study procedure [16]. Extended data includes the model consent form and the data collection form. The Consolidated Standards of Reporting Trials (CONSORT) recommendations will also be followed during the conduct of the study [17]. The study procedure is summarised in [Table/Fig-1].
Flowchart depicting the study’s design.
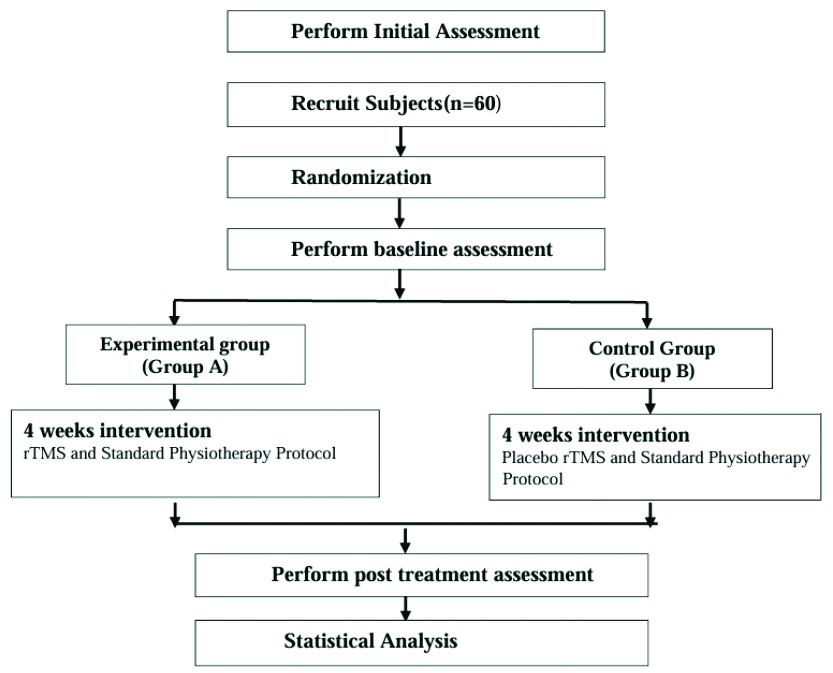
Following approval by the Institutional Ethics Committee (IEC) of the Datta Meghe Institute of Higher Education and Research (Institutional Ethical Clearance was obtained on 31/01/24; IEC No: DMIHER (DU)/IEC/2024/148), participants will be recruited based on the study’s inclusion and exclusion criteria from the neurorehabilitation unit of Acharya Vinoba Bhave Rural Hospital and the Neurophysiotherapy outpatient department of Ravi Nair Physiotherapy College.
Participants diagnosed with subacute stroke will be randomised through simple random sampling and allocated via the SNOSE method into Group A and Group B [Table/Fig-1]. Randomisation and allocation will be conducted by the primary researcher, who is an Assistant Professor in Physiotherapy. Outcome measures will be assessed before the beginning of the study and after its completion by an Assistant Professor with equivalent experience, who is aware of the study but blinded to the intervention.
As part of the inclusion process for the study, subjects will be invited and screened. Participants will receive written informed consent forms and information about the project following the screening. The assessor will use a questionnaire containing basic demographic details and exclusion criteria to conduct the baseline evaluation, which includes assessments of muscle tone and functional ability (MAS, STREAM, FRT, and 10-MWT).
The subjects will be selected in a randomised manner using a computer-generated random number table. Group A (Experimental) and Group B (Control) will be assigned in a 1:1 ratio through randomisation. When recruiting participants, the cut-off values of the baseline parameters will serve as the basis for the inclusion and exclusion criteria. Group A will receive rTMS and standard physiotherapy to assess any marginal differences in improved muscle tone, function, functional reach and gait velocity at the endpoint. The control group will receive placebo rTMS along with standard physiotherapy. Given that the universal method of physiotherapy is well-established, it is most appropriate to compare it with this innovative strategy that still requires validation.
Inclusion criteria: Patients must be experiencing some form of cerebrovascular disease diagnosed by Computed Tomography (CT) or Magnetic Resonance Imaging (MRI), with a unilateral stroke that is either primary or of prior onset but not resulting in continuous neurological deficits. Patients should demonstrate stable consciousness and vital signs, be aged between 45 and 60 years, and belong to either gender. Eligible patients must have a MAS of less than 2 and a Brunnstrom stage of 2-4 [9]. Subjects must be in the subacute phase of stroke, with a disease duration of one week to three months. Additionally, the selected applicant or their relatives must sign the informed consent.
Exclusion criteria: Patients will be excluded if they have a family history of epilepsy or are taking seizure-inducing medications, those experiencing severe declines or failures of respiratory, cardiovascular, urinary, or other major systems and patients with severe communicative or cognitive impairments that would prevent meaningful participation in assessment and treatment. Specific exclusion criteria include: posterior circulation infarction; restrictions related to transcranial magnetic stimulation, such as metal implants, medication delivery systems, insulin pumps and aneurysm clips (excluding those made from a titanium alloy); cervical spinal instability or severe cervical stenosis in the presence of severe cervical spondylosis; recurrent stroke; total internal carotid artery blockage; direct wounds and cranial trauma; flawed stimulation sites; pregnant women; and patients with a history of claustrophobia.
Sample size calculation: The sample size will be determined by comparing two means using Cohen’s effect size, considering a significant effect size difference of 0.8 (large effect size) [18].
Sample size by Cohen’s effect size by comparing two means
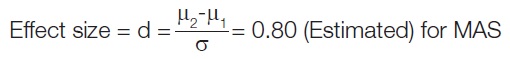
Considering large effect size difference=0.8 (Large effect size)
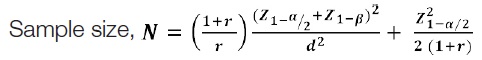
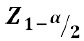 at 5% level of significance=1.96
at 5% level of significance=1.96
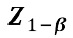 at 80% Power=0.84
at 80% Power=0.84
Ratio allocation (Group-2/Group-1)= 1
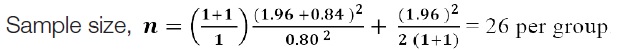
Total 26 samples required per group.
Taking 15% of total dropouts as 4,
Each group must have a total of 30 samples.
Interventions: The experimental group will receive low-frequency rTMS, with a frequency of less than 1 Hz, to the ipsilateral cortex (for 20 minutes), along with a standard physiotherapy protocol for the contralateral side, for one hour each day, five days per week, for four weeks [19]. The control group will receive their standard physiotherapy protocol for the contralateral side for one hour each day, five days per week, for four weeks [20], in addition to placebo rTMS (where the rTMS machine will not be switched on).
rTMS protocol: Patients will be seated during treatment, and 50-Hz rTMS will be administered over the primary motor cortex (M1) on the unaffected side using an rTMS device. The target for stimulation will be the M1 lower-limb motor area contralaterally, with the coil’s midpoint positioned over this motor area. The treatment frequency is 1 Hz, with an amplitude of 90% RMT. Each sequence consists of a 10-second train, with a 5-second inter-train interval, repeated 60 times for a total of 15 minutes or 600 pulses. This protocol was developed based on expert consensus and regarded as current best practice. Treatments will be applied for 15 minutes per session, once daily, five days a week, for four weeks [21].
Standard physiotherapy protocol [20]: Upper limb training involves various exercises to enhance motor function and coordination. This includes reaching for the shoulder in abduction, flexion and extension, as well as performing elbow flexion and extension. Key hand functions such as grasping, releasing and manipulating objects will also be emphasised. Early weight-bearing exercises will involve side-lying and weight-bearing on the elbow. Rolling exercises will target both sides, using Proprioceptive Neuromuscular Facilitation (PNF) in the D1 flexion pattern and pelvic PNF for assistance. In supine-to-sit transitions, assistance from both upper extremities will be provided.
Sitting balance exercises will involve turning the head and trunk while looking over the shoulder, raising the gaze to the ceiling, and extending the paretic hand forward, sideways and backward while seated. “Butt walking,” or scooting while seated, will also be practised. For standing balance, exercises will involve standing with feet apart, looking up at the ceiling and reaching forward, sideways, and backward with various tasks and objects, using one or both hands. Single-leg support will involve practising reaching while standing on one foot on a step and stepping forward with the non paretic limb.
Outcomes: The following outcomes will be assessed at the time of recruitment and after six weeks of treatment.
Modified Ashworth Scale (MAS): The MAS, which is characterised by increased joint resistance to passive movement, is the most commonly used clinical test for assessing the rise in muscle tone following Central Nervous System (CNS) injury. There are five possible scores according to the MAS: 0, 1, 1+, 2, 3, and 4 [22].
Assessment of Movement for Stroke Rehabilitation (STREAM): The STREAM tool measures voluntary movement and mobility after a stroke. It evaluates the patient’s functional mobility, coordination and range of motion. Healthcare practitioners can use this outcome measure to monitor the progression of stroke recovery, as it has been shown to be responsive to changes in acute stroke [23].
Functional Reach Test (FRT): The FRT is a clinical outcome test with a single task for evaluating complex equilibrium. It measures the greatest distance a person can stretch forward while standing still, assessing a patient’s stability. In the most recent version of the FRT, the patient can also be seated in a fixed position [24].
Gait velocity by 10-Metre Walk Test (10-MWT): This test calculates walking speed in meters per second over a brief distance. It records how long it takes to walk 6 metres in total, measured to the nearest tenth of a second. The total walking duration (in seconds) will then be used to convert the distance to metres per second by dividing 6 metres by the walking duration. Walking speed classifications are as follows: 0.40 to 0.80 m/s for household ambulators, 0.40 to 0.80 m/s for limited community ambulators, and ≥0.80 m/s for community ambulators [25].
Study timeline: Participants will be enrolled and assessed pre- and post-therapy, in addition to visits one and two for subject recruitment and screening, baseline assessment and following the conclusion of the four-week period when outcome measures will be measured. The study schedule for enrolment, intervention and assessment is displayed in [Table/Fig-2] (as recommended by the SPIRIT guidelines).
Schedule of enrolment, interventions and assessment from the SPIRIT guidelines.
| Study Period |
|---|
| Enrolment | Allocation | Postallocation |
|---|
| Timepoint | -t1 | 0 | Intervention (t1-4) | Post-test (t4) |
| Enrollment | | | | |
| Eligibility screen | x | | | |
| Informed consent | x | | | |
| Allocation | | x | | |
| Interventions | | | | |
| [Intervention A] | | | x | |
| [Intervention B] | | | x | |
| Assesments | | | | |
| MAS | x | | | x |
| STREAM | x | | | x |
| FRT | x | | | x |
| 10-MWT | x | | | x |
Statistical Analysis
R Studio version 4.3 will be used to analyse the total findings. To examine each participant’s demographics and conduct an analysis, data pertaining to all study participants will be gathered and organised according to their individual records. Quantitative demographic characteristics, such as age, will be displayed with means, standard deviations, medians, maximum, and minimum values. Qualitative or categorical data, such as hand dominance, gender and affected side, will be presented as frequencies and percentages.
The MAS, STREAM, FRT, and 10-MWT will be used to assess each patient before and after therapy. The paired t-test will be employed to evaluate the outcome variables for the pre-post results. The Kolmogorov-Smirnov test will be used to assess the normality of the data. If the data are normally distributed, a parametric test will be performed; if normality is violated, a non parametric test will be utilised, with a significance level set at p-value <0.05.
[1]. Stroke- Symptoms and causes- Mayo Clinic [Internet][Cited 2024 Feb 4]. Available from: https://www.mayoclinic.org/diseases-conditions/stroke/symptoms-causes/syc-20350113 [Google Scholar]
[2]. Donkor ES, Stroke in the 21st Century: A snapshot of the burden, Epidemiology, and quality of lifeStroke Res Treat 2018 2018:3238165[Cited 2022 Jun 12]. Available from: https://www.ncbi.nlm.nih.gov/pmc/articles/10.1155/2018/3238165PMC6288566 [Google Scholar] [CrossRef]
[3]. Kamalakannan S, Gudlavalleti ASV, Gudlavalleti VSM, Goenka S, Kuper H, Incidence & prevalence of stroke in India: A systematic reviewIndian J Med Res 2017 146(2):175-85.[Cited 2021 Jan 24]. Available from: https://www.ijmr.org.in/article.asp?issn=0971-5916;year=2017;volume=146;issue=2;spage=175;epage=185;aulast=Kamalakannan;type=010.4103/ijmr.IJMR_516_1529265018PMC5761027 [Google Scholar] [CrossRef] [PubMed]
[4]. Behera DK, Rahut DB, Mishra S, Analyzing stroke burden and risk factors in India using data from the Global Burden of Disease StudySci Rep 2024 14:22640[Cited 2024 Nov 29]. Available from: https://www.nature.com/articles/s41598-024-72551-410.1038/s41598-024-72551-439349946PMC11443044 [Google Scholar] [CrossRef] [PubMed]
[5]. Dodd KC, Nair VA, Prabhakaran V, Role of the contralesional vs. ipsilesional hemisphere in stroke recoveryFront Hum Neurosci 2017 11:469Available from: https://www.frontiersin.org/articles/10.3389/fnhum.2017.0046910.3389/fnhum.2017.0046928983244PMC5613154 [Google Scholar] [CrossRef] [PubMed]
[6]. Jang SH, Cho MJ, Role of the contra-lesional corticoreticular tract in motor recovery of the paretic leg in stroke: A mini-narrative reviewFront Hum Neurosci 2022 16:89636710.3389/fnhum.2022.89636735721363PMC9204517 [Google Scholar] [CrossRef] [PubMed]
[7]. Boddington LJ, Reynolds JNJ, Targeting interhemispheric inhibition with neuromodulation to enhance stroke rehabilitationBrain Stimul 2017 10(2):214-22.[Cited 2024 Jan 8]. Available from: https://pubmed.ncbi.nlm.nih.gov/28117178/10.1016/j.brs.2017.01.00628117178 [Google Scholar] [CrossRef] [PubMed]
[8]. Mann SK, Malhi NK, Repetitive transcranial magnetic stimulationIn: StatPearls [Internet] 2023 Treasure Island (FL)StatPearls Publishing[Cited 2024 Jan 7]. Available from: http://www.ncbi.nlm.nih.gov/books/NBK568715/ [Google Scholar]
[9]. Chang WH, Kim YH, Bang OY, Kim ST, Park YH, Lee PKW, Long-term effects of rTMS on motor recovery in patients after subacute strokeJ Rehabil Med 2010 42(8):758-64.10.2340/16501977-059020809058 [Google Scholar] [CrossRef] [PubMed]
[10]. Luk KY, Ouyang HX, Pang MYC, Low-frequency rTMS over contralesional M1 increases ipsilesional cortical excitability and motor function with decreased interhemispheric asymmetry in subacute stroke: A randomized controlled studyNeural Plast 2022 2022:3815357[Cited 2024 Feb 4]. Available from: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC8756161/10.1155/2022/381535735035473PMC8756161 [Google Scholar] [CrossRef] [PubMed]
[11]. Ni J, Jiang W, Gong X, Fan Y, Qiu H, Dou J, Effect of rTMS intervention on upper limb motor function after stroke: A study based on fNIRSFront Aging Neurosci 2023 14:1077218[Cited 2024 Feb 4]. Available from: https://pubmed.ncbi.nlm.nih.gov/36711205/10.3389/fnagi.2022.107721836711205PMC9880218 [Google Scholar] [CrossRef] [PubMed]
[12]. Tang Z, Liu T, Han K, Liu Y, Su W, Wang R, The effects of rTMS on motor recovery after stroke: A systematic review of fMRI studiesNeurol Sci 2024 45(3):897-909.[Cited 2024 Jan 26]. Available from: https://doi.org/10.1007/s10072-023-07123-x10.1007/s10072-023-07123-x37880452 [Google Scholar] [CrossRef] [PubMed]
[13]. Sharma H, Vishnu VY, Kumar N, Sreenivas V, Rajeswari MR, Bhatia R, Efficacy of low-frequency repetitive transcranial magnetic stimulation in ischemic stroke: A double-blind randomized controlled trialArch Rehabil Res Clin Transl 2020 2(1):10003910.1016/j.arrct.2020.10003933543068PMC7853333 [Google Scholar] [CrossRef] [PubMed]
[14]. Wu X, Wang R, Wu Q, Liao C, Zhang J, Jiao H, The effects of combined high-frequency repetitive transcranial magnetic stimulation and cervical nerve root magnetic stimulation on upper extremity motor recovery following strokeFront Neurosci 2023 17:1100464Available from: https://www.frontiersin.org/articles/10.3389/fnins.2023.110046410.3389/fnins.2023.110046436845428PMC9951778 [Google Scholar] [CrossRef] [PubMed]
[15]. Wu X, Ji GJ, Geng Z, Wang L, Yan Y, Wu Y, Accelerated intermittent theta-burst stimulation broadly ameliorates symptoms and cognition in Alzheimer’s disease: A randomized controlled trialBrain Stimul 2022 15(1):35-45.Available from: https://www.sciencedirect.com/science/article/pii/S1935861X2100805610.1016/j.brs.2021.11.00734752934 [Google Scholar] [CrossRef] [PubMed]
[16]. Chan AW, Tetzlaff JM, Altman DG, Laupacis A, Gøtzsche PC, Krleža-Jerić K, SPIRIT 2013 Statement: Defining standard protocol items for clinical trialsAnn Intern Med 2013 158:200-207.[Epub 5 February 2013]10.7326/0003-4819-158-3-201302050-0058323295957PMC5114123 [Google Scholar] [CrossRef] [PubMed]
[17]. Turner L, Shamseer L, Altman DG, Weeks L, Peters J, Kober T, Consolidated standards of reporting trials (CONSORT) and the completeness of reporting of randomised controlled trials (RCTs) published in medical journalsCochrane Database Syst Rev [Internet] 2012 2012(11):MR000030Available from: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC7386818/10.1002/14651858.MR000030.pub223152285PMC7386818 [Google Scholar] [CrossRef] [PubMed]
[18]. Serdar CC, Cihan M, Yücel D, Serdar MA, Sample size, power and effect size revisited: simplified and practical approaches in pre-clinical, clinical and laboratory studiesBiochem Med (Zagreb) [Internet] 2021 31(1):010502Available from: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC7745163/10.11613/BM.2021.01050233380887PMC7745163 [Google Scholar] [CrossRef] [PubMed]
[19]. Sung WH, Wang CP, Chou CL, Chen YC, Chang YC, Tsai PY, Efficacy of coupling inhibitory and facilitatory repetitive transcranial magnetic stimulation to enhance motor recovery in hemiplegic stroke patientsStroke 2013 44(5):1375-82.10.1161/STROKEAHA.111.00052223532011 [Google Scholar] [CrossRef] [PubMed]
[20]. Harjpal P, Qureshi MI, Kovela RK, Jain M, Efficacy of bilateral lower-limb training over unilateral lower-limb training to reeducate balance and walking in post-stroke survivors: A randomized clinical trialCureus 2022 14(10):e3074810.7759/cureus.30748 [Google Scholar] [CrossRef]
[21]. Wang L, Wang L, Wang Z, Gao F, Wu J, Tang H, Clinical effect analysis of wearable sensor technology-based gait function analysis in post-transcranial magnetic stimulation stroke patientsSensors (Basel) [Internet] 2024 24(10):3051Available from: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC11125090/10.3390/s2410305138793907PMC11125090 [Google Scholar] [CrossRef] [PubMed]
[22]. Blackburn M, van Vliet P, Mockett SP, Reliability of measurements obtained with the modified ashworth scale in the lower extremities of people with strokePhys Ther 2002 82(1):25-34.Available from: https://academic.oup.com/ptj/article/82/1/25/283697810.1093/ptj/82.1.2511784275 [Google Scholar] [CrossRef] [PubMed]
[23]. Daley K, Mayo N, Wood-Dauphinée S, Reliability of scores on the Stroke Rehabilitation Assessment of Movement (STREAM) measurePhys Ther 1999 79(1):08-19.quiz 20-2310.1093/ptj/79.1.89920188 [Google Scholar] [CrossRef] [PubMed]
[24]. Duncan PW, Weiner DK, Chandler J, Studenski S, Functional reach: A new clinical measure of balanceJ Gerontol 1990 45(6):M192-M197.10.1093/geronj/45.6.M1922229941 [Google Scholar] [CrossRef] [PubMed]
[25]. Cheng DK, Nelson M, Brooks D, Salbach NM, Validation of stroke-specific protocols for the 10-meter walk test and 6-minute walk test conducted using 15-meter and 30-meter walkwaysTop Stroke Rehabil 2020 27(4):251-61.10.1080/10749357.2019.169181531752634 [Google Scholar] [CrossRef] [PubMed]